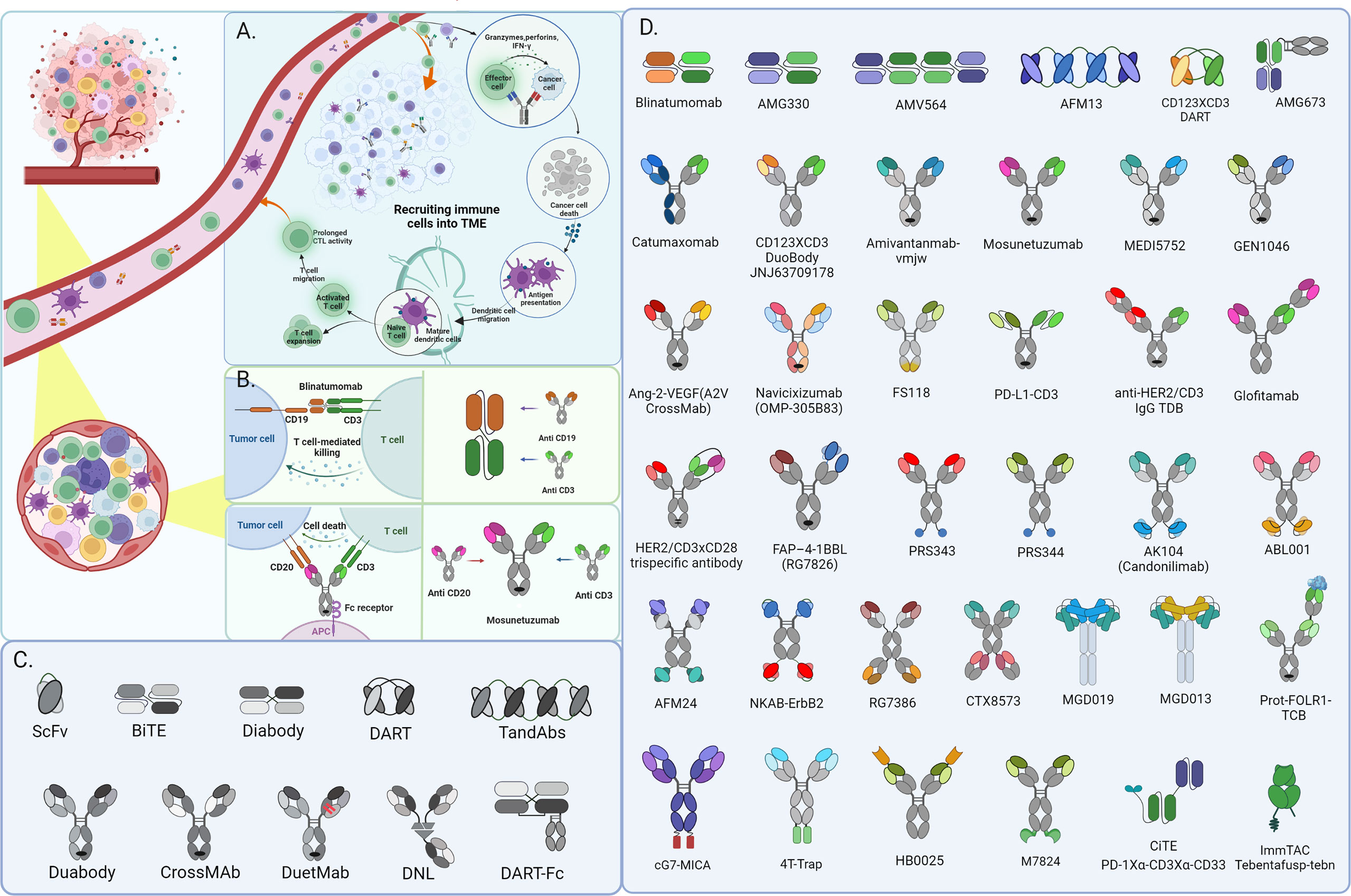
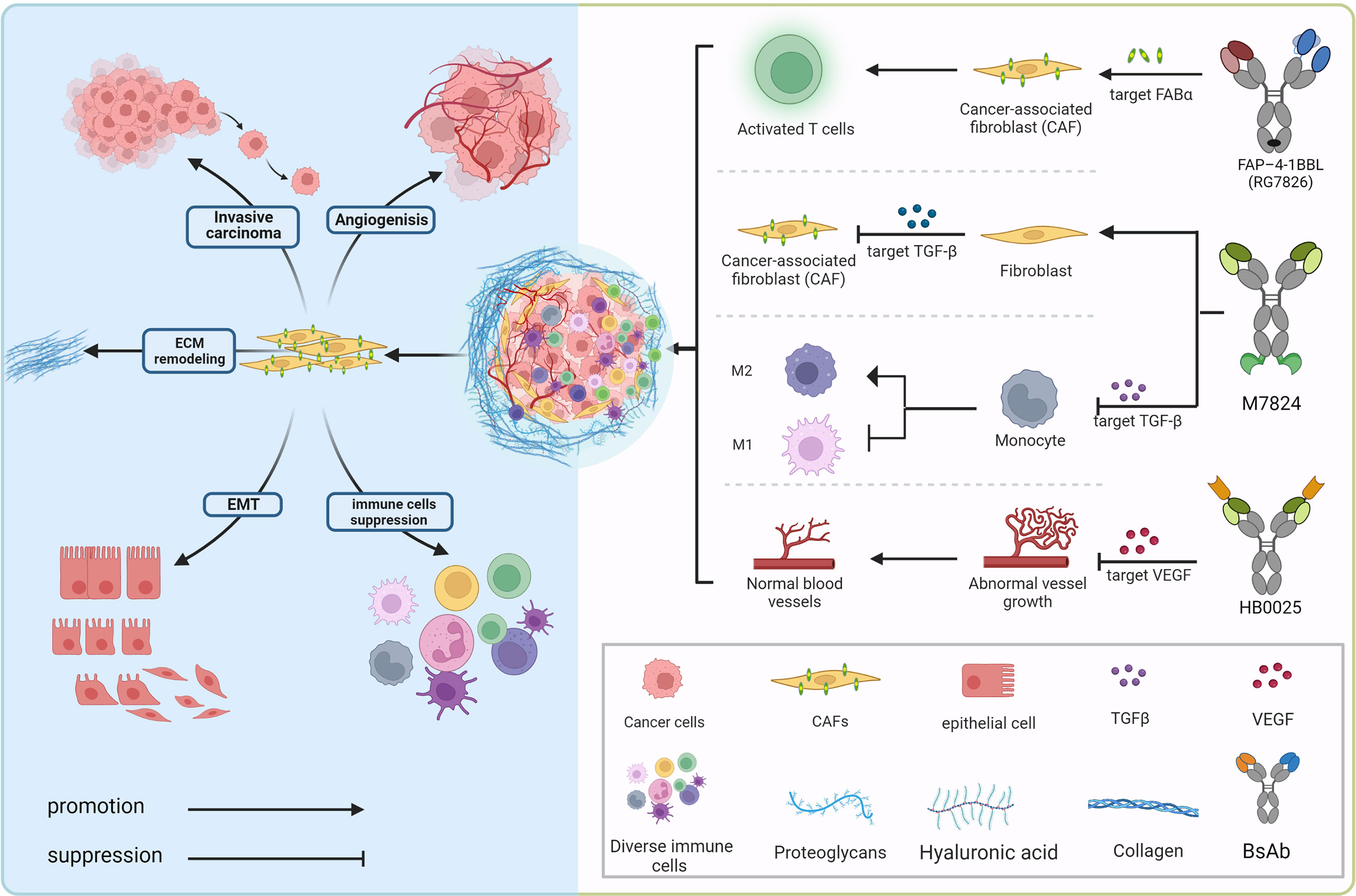
From High-Throughput Assay to Systems Biology: New Tools for Drug Discovery
Curator: Stephen J. Williams, PhD
Marc W. Kirschner*
Department of Systems Biology
Harvard Medical School
Boston, Massachusetts 02115
With the new excitement about systems biology, there is understandable interest in a definition. This has proven somewhat difficult. Scientific fields, like species, arise by descent with modification, so in their earliest forms even the founders of great dynasties are only marginally different than their sister fields and species. It is only in retrospect that we can recognize the significant founding events. Before embarking on a definition of systems biology, it may be worth remembering that confusion and controversy surrounded the introduction of the term “molecular biology,” with claims that it hardly differed from biochemistry. Yet in retrospect molecular biology was new and different. It introduced both new subject matter and new technological approaches, in addition to a new style.
As a point of departure for systems biology, consider the quintessential experiment in the founding of molecular biology, the one gene one enzyme hypothesis of Beadle and Tatum. This experiment first connected the genotype directly to the phenotype on a molecular level, although efforts in that direction can certainly be found in the work of Archibald Garrod, Sewell Wright, and others. Here a protein (in this case an enzyme) is seen to be a product of a single gene, and a single function; the completion of a specific step in amino acid biosynthesis is the direct result. It took the next 30 years to fill in the gaps in this process. Yet the one gene one enzyme hypothesis looks very different to us today. What is the function of tubulin, of PI-3 kinase or of rac? Could we accurately predict the phenotype of a nonlethal mutation in these genes in a multicellular organism? Although we can connect structure to the gene, we can no longer infer its larger purpose in the cell or in the organism. There are too many purposes; what the protein does is defined by context. The context also includes a history, either developmental or physiological. Thus the behavior of the Wnt signaling pathway depends on the previous lineage, the “where and when” questions of embryonic development. Similarly the behavior of the immune system depends on previous experience in a variable environment. All of these features stress how inadequate an explanation for function we can achieve solely by trying to identify genes (by annotating them!) and characterizing their transcriptional control circuits.
That we are at a crossroads in how to explore biology is not at all clear to many. Biology is hardly in its dotage; the process of discovery seems to have been perfected, accelerated, and made universally applicable to all fields of biology. With the completion of the human genome and the genomes of other species, we have a glimpse of many more genes than we ever had before to study. We are like naturalists discovering a new continent, enthralled with the diversity itself. But we have also at the same time glimpsed the finiteness of this list of genes, a disturbingly small list. We have seen that the diversity of genes cannot approximate the diversity of functions within an organism. In response, we have argued that combinatorial use of small numbers of components can generate all the diversity that is needed. This has had its recent incarnation in the simplistic view that the rules of cis-regulatory control on DNA can directly lead to an understanding of organisms and their evolution. Yet this assumes that the gene products can be linked together in arbitrary combinations, something that is not assured in chemistry. It also downplays the significant regulatory features that involve interactions between gene products, their localization, binding, posttranslational modification, degradation, etc. The big question to understand in biology is not regulatory linkage but the nature of biological systems that allows them to be linked together in many nonlethal and even useful combinations. More and more we come to realize that understanding the conserved genes and their conserved circuits will require an understanding of their special properties that allow them to function together to generate different phenotypes in different tissues of metazoan organisms. These circuits may have certain robustness, but more important they have adaptability and versatility. The ease of putting conserved processes under regulatory control is an inherent design feature of the processes themselves. Among other things it loads the deck in evolutionary variation and makes it more feasible to generate useful phenotypes upon which selection can act.
Systems biology offers an opportunity to study how the phenotype is generated from the genotype and with it a glimpse of how evolution has crafted the phenotype. One aspect of systems biology is the development of techniques to examine broadly the level of protein, RNA, and DNA on a gene by gene basis and even the posttranslational modification and localization of proteins. In a very short time we have witnessed the development of high-throughput biology, forcing us to consider cellular processes in toto. Even though much of the data is noisy and today partially inconsistent and incomplete, this has been a radical shift in the way we tear apart problems one interaction at a time. When coupled with gene deletions by RNAi and classical methods, and with the use of chemical tools tailored to proteins and protein domains, these high-throughput techniques become still more powerful.
High-throughput biology has opened up another important area of systems biology: it has brought us out into the field again or at least made us aware that there is a world outside our laboratories. Our model systems have been chosen intentionally to be of limited genetic diversity and examined in a highly controlled and reproducible environment. The real world of ecology, evolution, and human disease is a very different place. When genetics separated from the rest of biology in the early part of the 20th century, most geneticists sought to understand heredity and chose to study traits in the organism that could be easily scored and could be used to reveal genetic mechanisms. This was later extended to powerful effect to use genetics to study cell biological and developmental mechanisms. Some geneticists, including a large school in Russia in the early 20th century, continued to study the genetics of natural populations, focusing on traits important for survival. That branch of genetics is coming back strongly with the power of phenotypic assays on the RNA and protein level. As human beings we are most concerned not with using our genetic misfortunes to unravel biology’s complexity (important as that is) but with the role of our genetics in our individual survival. The context for understanding this is still not available, even though the data are now coming in torrents, for many of the genes that will contribute to our survival will have small quantitative effects, partially masked or accentuated by other genetic and environmental conditions. To understand the genetic basis of disease will require not just mapping these genes but an understanding of how the phenotype is created in the first place and the messy interactions between genetic variation and environmental variation.
Extracts and explants are relatively accessible to synthetic manipulation. Next there is the explicit reconstruction of circuits within cells or the deliberate modification of those circuits. This has occurred for a while in biology, but the difference is that now we wish to construct or intervene with the explicit purpose of describing the dynamical features of these synthetic or partially synthetic systems. There are more and more tools to intervene and more and more tools to measure. Although these fall short of total descriptions of cells and organisms, the detailed information will give us a sense of the special life-like processes of circuits, proteins, cells in tissues, and whole organisms in their environment. This meso-scale systems biology will help establish the correspondence between molecules and large-scale physiology.
You are probably running out of patience for some definition of systems biology. In any case, I do not think the explicit definition of systems biology should come from me but should await the words of the first great modern systems biologist. She or he is probably among us now. However, if forced to provide some kind of label for systems biology, I would simply say that systems biology is the study of the behavior of complex biological organization and processes in terms of the molecular constituents. It is built on molecular biology in its special concern for information transfer, on physiology for its special concern with adaptive states of the cell and organism, on developmental biology for the importance of defining a succession of physiological states in that process, and on evolutionary biology and ecology for the appreciation that all aspects of the organism are products of selection, a selection we rarely understand on a molecular level. Systems biology attempts all of this through quantitative measurement, modeling, reconstruction, and theory. Systems biology is not a branch of physics but differs from physics in that the primary task is to understand how biology generates variation. No such imperative to create variation exists in the physical world. It is a new principle that Darwin understood and upon which all of life hinges. That sounds different enough for me to justify a new field and a new name. Furthermore, the success of systems biology is essential if we are to understand life; its success is far from assured—a good field for those seeking risk and adventure.
Source: “Meaning of Systems Biology” Cell, Vol. 121, 503–504, May 20, 2005, DOI 10.1016/j.cell.2005.05.005
Old High-throughput Screening, Once the Gold Standard in Drug Development, Gets a Systems Biology Facelift
From Phenotypic Hit to Chemical Probe: Chemical Biology Approaches to Elucidate Small Molecule Action in Complex Biological Systems
Quentin T. L. Pasquer, Ioannis A. Tsakoumagkos and Sascha Hoogendoorn
Molecules 2020, 25(23), 5702; https://doi.org/10.3390/molecules25235702
Abstract
Biologically active small molecules have a central role in drug development, and as chemical probes and tool compounds to perturb and elucidate biological processes. Small molecules can be rationally designed for a given target, or a library of molecules can be screened against a target or phenotype of interest. Especially in the case of phenotypic screening approaches, a major challenge is to translate the compound-induced phenotype into a well-defined cellular target and mode of action of the hit compound. There is no “one size fits all” approach, and recent years have seen an increase in available target deconvolution strategies, rooted in organic chemistry, proteomics, and genetics. This review provides an overview of advances in target identification and mechanism of action studies, describes the strengths and weaknesses of the different approaches, and illustrates the need for chemical biologists to integrate and expand the existing tools to increase the probability of evolving screen hits to robust chemical probes.
5.1.5. Large-Scale Proteomics
While FITExP is based on protein expression regulation during apoptosis, a study of Ruprecht et al. showed that proteomic changes are induced both by cytotoxic and non-cytotoxic compounds, which can be detected by mass spectrometry to give information on a compound’s mechanism of action. They developed a large-scale proteome-wide mass spectrometry analysis platform for MOA studies, profiling five lung cancer cell lines with over 50 drugs. Aggregation analysis over the different cell lines and the different compounds showed that one-quarter of the drugs changed the abundance of their protein target. This approach allowed target confirmation of molecular degraders such as PROTACs or molecular glues. Finally, this method yielded unexpected off-target mechanisms for the MAP2K1/2 inhibitor PD184352 and the ALK inhibitor ceritinib [97]. While such a mapping approach clearly provides a wealth of information, it might not be easily attainable for groups that are not equipped for high-throughput endeavors.
All-in-all, mass spectrometry methods have gained a lot of traction in recent years and have been successfully applied for target deconvolution and MOA studies of small molecules. As with all high-throughput methods, challenges lie in the accessibility of the instruments (both from a time and cost perspective) and data analysis of complex and extensive data sets.
5.2. Genetic Approaches
Both label-based and mass spectrometry proteomic approaches are based on the physical interaction between a small molecule and a protein target, and focus on the proteome for target deconvolution. It has been long realized that genetics provides an alternative avenue to understand a compound’s action, either through precise modification of protein levels, or by inducing protein mutations. First realized in yeast as a genetically tractable organism over 20 years ago, recent advances in genetic manipulation of mammalian cells have opened up important opportunities for target identification and MOA studies through genetic screening in relevant cell types [98]. Genetic approaches can be roughly divided into two main areas, with the first centering on the identification of mutations that confer compound resistance (Figure 3a), and the second on genome-wide perturbation of gene function and the concomitant changes in sensitivity to the compound (Figure 3b). While both methods can be used to identify or confirm drug targets, the latter category often provides many additional insights in the compound’s mode of action.

Figure 3. Genetic methods for target identification and mode of action studies. Schematic representations of (a) resistance cloning, and (b) chemogenetic interaction screens.
5.2.1. Resistance Cloning
The “gold standard” in drug target confirmation is to identify mutations in the presumed target protein that render it insensitive to drug treatment. Conversely, different groups have sought to use this principle as a target identification method based on the concept that cells grown in the presence of a cytotoxic drug will either die or develop mutations that will make them resistant to the compound. With recent advances in deep sequencing it is now possible to then scan the transcriptome [99] or genome [100] of the cells for resistance-inducing mutations. Genes that are mutated are then hypothesized to encode the protein target. For this approach to be successful, there are two initial requirements: (1) the compound needs to be cytotoxic for resistant clones to arise, and (2) the cell line needs to be genetically unstable for mutations to occur in a reasonable timeframe.
In 2012, the Kapoor group demonstrated in a proof-of-concept study that resistance cloning in mammalian cells, coupled to transcriptome sequencing (RNA-seq), yields the known polo-like kinase 1 (PLK1) target of the small molecule BI 2536. For this, they used the cancer cell line HCT-116, which is deficient in mismatch repair and consequently prone to mutations. They generated and sequenced multiple resistant clones, and clustered the clones based on similarity. PLK1 was the only gene that was mutated in multiple groups. Of note, one of the groups did not contain PLK1 mutations, but rather developed resistance through upregulation of ABCBA1, a drug efflux transporter, which is a general and non-specific resistance mechanism [101]. In a following study, they optimized their pipeline “DrugTargetSeqR”, by counter-screening for these types of multidrug resistance mechanisms so that these clones were excluded from further analysis (Figure 3a). Furthermore, they used CRISPR/Cas9-mediated gene editing to determine which mutations were sufficient to confer drug resistance, and as independent validation of the biochemical relevance of the obtained hits [102].
While HCT-116 cells are a useful model cell line for resistance cloning because of their genomic instability, they may not always be the cell line of choice, depending on the compound and process that is studied. Povedana et al. used CRISPR/Cas9 to engineer mismatch repair deficiencies in Ewing sarcoma cells and small cell lung cancer cells. They found that deletion of MSH2 results in hypermutations in these normally mutationally silent cells, resulting in the formation of resistant clones in the presence of bortezomib, MLN4924, and CD437, which are all cytotoxic compounds [103]. Recently, Neggers et al. reasoned that CRISPR/Cas9-induced non-homologous end-joining repair could be a viable strategy to create a wide variety of functional mutants of essential genes through in-frame mutations. Using a tiled sgRNA library targeting 75 target genes of investigational neoplastic drugs in HAP1 and K562 cells, they generated several KPT-9274 (an anticancer agent with unknown target)-resistant clones, and subsequent deep sequencing showed that the resistant clones were enriched in NAMPT sgRNAs. Direct target engagement was confirmed by co-crystallizing the compound with NAMPT [104]. In addition to these genetic mutation strategies, an alternative method is to grow the cells in the presence of a mutagenic chemical to induce higher mutagenesis rates [105,106].
When there is already a hypothesis on the pathway involved in compound action, the resistance cloning methodology can be extended to non-cytotoxic compounds. Sekine et al. developed a fluorescent reporter model for the integrated stress response, and used this cell line for target deconvolution of a small molecule inhibitor towards this pathway (ISRIB). Reporter cells were chemically mutagenized, and ISRIB-resistant clones were isolated by flow cytometry, yielding clones with various mutations in the delta subunit of guanine nucleotide exchange factor eIF2B [107].
While there are certainly successful examples of resistance cloning yielding a compound’s direct target as discussed above, resistance could also be caused by mutations or copy number alterations in downstream components of a signaling pathway. This is illustrated by clinical examples of acquired resistance to small molecules, nature’s way of “resistance cloning”. For example, resistance mechanisms in Hedgehog pathway-driven cancers towards the Smoothened inhibitor vismodegib include compound-resistant mutations in Smoothened, but also copy number changes in downstream activators SUFU and GLI2 [108]. It is, therefore, essential to conduct follow-up studies to confirm a direct interaction between a compound and the hit protein, as well as a lack of interaction with the mutated protein.
5.2.3. “Chemogenomics”: Examples of Gene-Drug Interaction Screens
When genetic perturbations are combined with small molecule drugs in a chemogenetic interaction screen, the effect of a gene’s perturbation on compound action is studied. Gene perturbation can render the cells resistant to the compound (suppressor interaction), or conversely, result in hypersensitivity and enhanced compound potency (synergistic interaction) [5,117,121]. Typically, cells are treated with the compound at a sublethal dose, to ascertain that both types of interactions can be found in the final dataset, and often it is necessary to use a variety of compound doses (i.e., LD20, LD30, LD50) and timepoints to obtain reliable insights (Figure 3b).
An early example of successful coupling of a phenotypic screen and downstream genetic screening for target identification is the study of Matheny et al. They identified STF-118804 as a compound with antileukemic properties. Treatment of MV411 cells, stably transduced with a high complexity, genome-wide shRNA library, with STF-118804 (4 rounds of increasing concentration) or DMSO control resulted in a marked depletion of cells containing shRNAs against nicotinamide phosphoribosyl transferase (NAMPT) [122].
The Bassik lab subsequently directly compared the performance of shRNA-mediated knockdown versus CRISPR/Cas9-knockout screens for the target elucidation of the antiviral drug GSK983. The data coming out of both screens were complementary, with the shRNA screen resulting in hits leading to the direct compound target and the CRISPR screen giving information on cellular mechanisms of action of the compound. A reason for this is likely the level of protein depletion that is reached by these methods: shRNAs lead to decreased protein levels, which is advantageous when studying essential genes. However, knockdown may not result in a phenotype for non-essential genes, in which case a full CRISPR-mediated knockout is necessary to observe effects [123].
Another NAMPT inhibitor was identified in a CRISPR/Cas9 “haplo-insufficiency (HIP)”-like approach [124]. Haploinsuffiency profiling is a well-established system in yeast which is performed in a ~50% protein background by heterozygous deletions [125]. As there is no control over CRISPR-mediated loss of alleles, compound treatment was performed at several timepoints after addition of the sgRNA library to HCT116 cells stably expressing Cas9, in the hope that editing would be incomplete at early timepoints, resulting in residual protein levels. Indeed, NAMPT was found to be the target of phenotypic hit LB-60-OF61, especially at earlier timepoints, confirming the hypothesis that some level of protein needs to be present to identify a compound’s direct target [124]. This approach was confirmed in another study, thereby showing that direct target identification through CRISPR-knockout screens is indeed possible [126].
An alternative strategy was employed by the Weissman lab, where they combined genome-wide CRISPR-interference and -activation screens to identify the target of the phase 3 drug rigosertib. They focused on hits that had opposite action in both screens, as in sensitizing in one but protective in the other, which were related to microtubule stability. In a next step, they created chemical-genetic profiles of a variety of microtubule destabilizing agents, rationalizing that compounds with the same target will have similar drug-gene interactions. For this, they made a focused library of sgRNAs, based on the most high-ranking hits in the rigosertib genome-wide CRISPRi screen, and compared the focused screen results of the different compounds. The profile for rigosertib clustered well with that of ABT-571, and rigorous target validation studies confirmed rigosertib binding to the colchicine binding site of tubulin—the same site as occupied by ABT-571 [127].
From the above examples, it is clear that genetic screens hold a lot of promise for target identification and MOA studies for small molecules. The CRISPR screening field is rapidly evolving, sgRNA libraries are continuously improving and increasingly commercially available, and new tools for data analysis are being developed [128]. The challenge lies in applying these screens to study compounds that are not cytotoxic, where finding the right dosage regimen will not be trivial.
SYSTEMS BIOLOGY AND CANCER RESEARCH & DRUG DISCOVERY
Integrative Analysis of Next-Generation Sequencing for Next-Generation Cancer Research toward Artificial Intelligence
Youngjun Park, Dominik Heider and Anne-Christin Hauschild. Cancers 2021, 13(13), 3148; https://doi.org/10.3390/cancers13133148
Abstract
The rapid improvement of next-generation sequencing (NGS) technologies and their application in large-scale cohorts in cancer research led to common challenges of big data. It opened a new research area incorporating systems biology and machine learning. As large-scale NGS data accumulated, sophisticated data analysis methods became indispensable. In addition, NGS data have been integrated with systems biology to build better predictive models to determine the characteristics of tumors and tumor subtypes. Therefore, various machine learning algorithms were introduced to identify underlying biological mechanisms. In this work, we review novel technologies developed for NGS data analysis, and we describe how these computational methodologies integrate systems biology and omics data. Subsequently, we discuss how deep neural networks outperform other approaches, the potential of graph neural networks (GNN) in systems biology, and the limitations in NGS biomedical research. To reflect on the various challenges and corresponding computational solutions, we will discuss the following three topics: (i) molecular characteristics, (ii) tumor heterogeneity, and (iii) drug discovery. We conclude that machine learning and network-based approaches can add valuable insights and build highly accurate models. However, a well-informed choice of learning algorithm and biological network information is crucial for the success of each specific research question
1. Introduction
The development and widespread use of high-throughput technologies founded the era of big data in biology and medicine. In particular, it led to an accumulation of large-scale data sets that opened a vast amount of possible applications for data-driven methodologies. In cancer, these applications range from fundamental research to clinical applications: molecular characteristics of tumors, tumor heterogeneity, drug discovery and potential treatments strategy. Therefore, data-driven bioinformatics research areas have tailored data mining technologies such as systems biology, machine learning, and deep learning, elaborated in this review paper (see Figure 1 and Figure 2). For example, in systems biology, data-driven approaches are applied to identify vital signaling pathways [1]. This pathway-centric analysis is particularly crucial in cancer research to understand the characteristics and heterogeneity of the tumor and tumor subtypes. Consequently, this high-throughput data-based analysis enables us to explore characteristics of cancers with a systems biology and a systems medicine point of view [2].Combining high-throughput techniques, especially next-generation sequencing (NGS), with appropriate analytical tools has allowed researchers to gain a deeper systematic understanding of cancer at various biological levels, most importantly genomics, transcriptomics, and epigenetics [3,4]. Furthermore, more sophisticated analysis tools based on computational modeling are introduced to decipher underlying molecular mechanisms in various cancer types. The increasing size and complexity of the data required the adaptation of bioinformatics processing pipelines for higher efficiency and sophisticated data mining methodologies, particularly for large-scale, NGS datasets [5]. Nowadays, more and more NGS studies integrate a systems biology approach and combine sequencing data with other types of information, for instance, protein family information, pathway, or protein–protein interaction (PPI) networks, in an integrative analysis. Experimentally validated knowledge in systems biology may enhance analysis models and guides them to uncover novel findings. Such integrated analyses have been useful to extract essential information from high-dimensional NGS data [6,7]. In order to deal with the increasing size and complexity, the application of machine learning, and specifically deep learning methodologies, have become state-of-the-art in NGS data analysis.

Figure 1. Next-generation sequencing data can originate from various experimental and technological conditions. Depending on the purpose of the experiment, one or more of the depicted omics types (Genomics, Transcriptomics, Epigenomics, or Single-Cell Omics) are analyzed. These approaches led to an accumulation of large-scale NGS datasets to solve various challenges of cancer research, molecular characterization, tumor heterogeneity, and drug target discovery. For instance, The Cancer Genome Atlas (TCGA) dataset contains multi-omics data from ten-thousands of patients. This dataset facilitates a variety of cancer researches for decades. Additionally, there are also independent tumor datasets, and, frequently, they are analyzed and compared with the TCGA dataset. As the large scale of omics data accumulated, various machine learning techniques are applied, e.g., graph algorithms and deep neural networks, for dimensionality reduction, clustering, or classification. (Created with BioRender.com.)

Figure 2. (a) A multitude of different types of data is produced by next-generation sequencing, for instance, in the fields of genomics, transcriptomics, and epigenomics. (b) Biological networks for biomarker validation: The in vivo or in vitro experiment results are considered ground truth. Statistical analysis on next-generation sequencing data produces candidate genes. Biological networks can validate these candidate genes and highlight the underlying biological mechanisms (Section 2.1). (c) De novo construction of Biological Networks: Machine learning models that aim to reconstruct biological networks can incorporate prior knowledge from different omics data. Subsequently, the model will predict new unknown interactions based on new omics information (Section 2.2). (d) Network-based machine learning: Machine learning models integrating biological networks as prior knowledge to improve predictive performance when applied to different NGS data (Section 2.3). (Created with BioRender.com).
Therefore, a large number of studies integrate NGS data with machine learning and propose a novel data-driven methodology in systems biology [8]. In particular, many network-based machine learning models have been developed to analyze cancer data and help to understand novel mechanisms in cancer development [9,10]. Moreover, deep neural networks (DNN) applied for large-scale data analysis improved the accuracy of computational models for mutation prediction [11,12], molecular subtyping [13,14], and drug repurposing [15,16].
2. Systems Biology in Cancer Research
Genes and their functions have been classified into gene sets based on experimental data. Our understandings of cancer concentrated into cancer hallmarks that define the characteristics of a tumor. This collective knowledge is used for the functional analysis of unseen data.. Furthermore, the regulatory relationships among genes were investigated, and, based on that, a pathway can be composed. In this manner, the accumulation of public high-throughput sequencing data raised many big-data challenges and opened new opportunities and areas of application for computer science. Two of the most vibrantly evolving areas are systems biology and machine learning which tackle different tasks such as understanding the cancer pathways [9], finding crucial genes in pathways [22,53], or predicting functions of unidentified or understudied genes [54]. Essentially, those models include prior knowledge to develop an analysis and enhance interpretability for high-dimensional data [2]. In addition to understanding cancer pathways with in silico analysis, pathway activity analysis incorporating two different types of data, pathways and omics data, is developed to understand heterogeneous characteristics of the tumor and cancer molecular subtyping. Due to its advantage in interpretability, various pathway-oriented methods are introduced and become a useful tool to understand a complex diseases such as cancer [55,56,57].
In this section, we will discuss how two related research fields, namely, systems biology and machine learning, can be integrated with three different approaches (see Figure 2), namely, biological network analysis for biomarker validation, the use of machine learning with systems biology, and network-based models.
2.1. Biological Network Analysis for Biomarker Validation
The detection of potential biomarkers indicative of specific cancer types or subtypes is a frequent goal of NGS data analysis in cancer research. For instance, a variety of bioinformatics tools and machine learning models aim at identify lists of genes that are significantly altered on a genomic, transcriptomic, or epigenomic level in cancer cells. Typically, statistical and machine learning methods are employed to find an optimal set of biomarkers, such as single nucleotide polymorphisms (SNPs), mutations, or differentially expressed genes crucial in cancer progression. Traditionally, resource-intensive in vitro analysis was required to discover or validate those markers. Therefore, systems biology offers in silico solutions to validate such findings using biological pathways or gene ontology information (Figure 2b) [58]. Subsequently, gene set enrichment analysis (GSEA) [50] or gene set analysis (GSA) [59] can be used to evaluate whether these lists of genes are significantly associated with cancer types and their specific characteristics. GSA, for instance, is available via web services like DAVID [60] and g:Profiler [61]. Moreover, other applications use gene ontology directly [62,63]. In addition to gene-set-based analysis, there are other methods that focuse on the topology of biological networks. These approaches evaluate various network structure parameters and analyze the connectivity of two genes or the size and interconnection of their neighbors [64,65]. According to the underlying idea, the mutated gene will show dysfunction and can affect its neighboring genes. Thus, the goal is to find abnormalities in a specific set of genes linked with an edge in a biological network. For instance, KeyPathwayMiner can extract informative network modules in various omics data [66]. In summary, these approaches aim at predicting the effect of dysfunctional genes among neighbors according to their connectivity or distances from specific genes such as hubs [67,68]. During the past few decades, the focus of cancer systems biology extended towards the analysis of cancer-related pathways since those pathways tend to carry more information than a gene set. Such analysis is called Pathway Enrichment Analysis (PEA) [69,70]. The use of PEA incorporates the topology of biological networks. However, simultaneously, the lack of coverage issue in pathway data needs to be considered. Because pathway data does not cover all known genes yet, an integration analysis on omics data can significantly drop in genes when incorporated with pathways. Genes that can not be mapped to any pathway are called ‘pathway orphan.’ In this manner, Rahmati et al. introduced a possible solution to overcome the ‘pathway orphan’ issue [71]. At the bottom line, regardless of whether researchers consider gene-set or pathway-based enrichment analysis, the performance and accuracy of both methods are highly dependent on the quality of the external gene-set and pathway data [72].
2.2. De Novo Construction of Biological Networks
While the known fraction of existing biological networks barely scratches the surface of the whole system of mechanisms occurring in each organism, machine learning models can improve on known network structures and can guide potential new findings [73,74]. This area of research is called de novo network construction (Figure 2c), and its predictive models can accelerate experimental validation by lowering time costs [75,76]. This interplay between in silico biological networks building and mining contributes to expanding our knowledge in a biological system. For instance, a gene co-expression network helps discover gene modules having similar functions [77]. Because gene co-expression networks are based on expressional changes under specific conditions, commonly, inferring a co-expression network requires many samples. The WGCNA package implements a representative model using weighted correlation for network construction that leads the development of the network biology field [78]. Due to NGS developments, the analysis of gene co-expression networks subsequently moved from microarray-based to RNA-seq based experimental data [79]. However, integration of these two types of data remains tricky. Ballouz et al. compared microarray and NGS-based co-expression networks and found the existence of a bias originating from batch effects between the two technologies [80]. Nevertheless, such approaches are suited to find disease-specific co-expressional gene modules. Thus, various studies based on the TCGA cancer co-expression network discovered characteristics of prognostic genes in the network [81]. Accordingly, a gene co-expression network is a condition-specific network rather than a general network for an organism. Gene regulatory networks can be inferred from the gene co-expression network when various data from different conditions in the same organism are available. Additionally, with various NGS applications, we can obtain multi-modal datasets about regulatory elements and their effects, such as epigenomic mechanisms on transcription and chromatin structure. Consequently, a gene regulatory network can consist of solely protein-coding genes or different regulatory node types such as transcription factors, inhibitors, promoter interactions, DNA methylations, and histone modifications affecting the gene expression system [82,83]. More recently, researchers were able to build networks based on a particular experimental setup. For instance, functional genomics or CRISPR technology enables the high-resolution regulatory networks in an organism [84]. Other than gene co-expression or regulatory networks, drug target, and drug repurposing studies are active research areas focusing on the de novo construction of drug-to-target networks to allow the potential repurposing of drugs [76,85].
2.3. Network Based Machine Learning
A network-based machine learning model directly integrates the insights of biological networks within the algorithm (Figure 2d) to ultimately improve predictive performance concerning cancer subtyping or susceptibility to therapy. Following the establishment of high-quality biological networks based on NGS technologies, these biological networks were suited to be integrated into advanced predictive models. In this manner, Zhang et al., categorized network-based machine learning approaches upon their usage into three groups: (i) model-based integration, (ii) pre-processing integration, and (iii) post-analysis integration [7]. Network-based models map the omics data onto a biological network, and proper algorithms travel the network while considering both values of nodes and edges and network topology. In the pre-processing integration, pathway or other network information is commonly processed based on its topological importance. Meanwhile, in the post-analysis integration, omics data is processed solely before integration with a network. Subsequently, omics data and networks are merged and interpreted. The network-based model has advantages in multi-omics integrative analysis. Due to the different sensitivity and coverage of various omics data types, a multi-omics integrative analysis is challenging. However, focusing on gene-level or protein-level information enables a straightforward integration [86,87]. Consequently, when different machine learning approaches tried to integrate two or more different data types to find novel biological insights, one of the solutions is reducing the search space to gene or protein level and integrated heterogeneous datatypes [25,88].
In summary, using network information opens new possibilities for interpretation. However, as mentioned earlier, several challenges remain, such as the coverage issue. Current databases for biological networks do not cover the entire set of genes, transcripts, and interactions. Therefore, the use of networks can lead to loss of information for gene or transcript orphans. The following section will focus on network-based machine learning models and their application in cancer genomics. We will put network-based machine learning into the perspective of the three main areas of application, namely, molecular characterization, tumor heterogeneity analysis, and cancer drug discovery.
3. Network-Based Learning in Cancer Research
As introduced previously, the integration of machine learning with the insights of biological networks (Figure 2d) ultimately aims at improving predictive performance and interpretability concerning cancer subtyping or treatment susceptibility.
3.1. Molecular Characterization with Network Information
Various network-based algorithms are used in genomics and focus on quantifying the impact of genomic alteration. By employing prior knowledge in biological network algorithms, performance compared to non-network models can be improved. A prominent example is HotNet. The algorithm uses a thermodynamics model on a biological network and identifies driver genes, or prognostic genes, in pan-cancer data [89]. Another study introduced a network-based stratification method to integrate somatic alterations and expression signatures with network information [90]. These approaches use network topology and network-propagation-like algorithms. Network propagation presumes that genomic alterations can affect the function of neighboring genes. Two genes will show an exclusive pattern if two genes complement each other, and the function carried by those two genes is essential to an organism [91]. This unique exclusive pattern among genomic alteration is further investigated in cancer-related pathways. Recently, Ku et al. developed network-centric approaches and tackled robustness issues while studying synthetic lethality [92]. Although synthetic lethality was initially discovered in model organisms of genetics, it helps us to understand cancer-specific mutations and their functions in tumor characteristics [91].
Furthermore, in transcriptome research, network information is used to measure pathway activity and its application in cancer subtyping. For instance, when comparing the data of two or more conditions such as cancer types, GSEA as introduced in Section 2 is a useful approach to get an overview of systematic changes [50]. It is typically used at the beginning of a data evaluation [93]. An experimentally validated gene set can provide information about how different conditions affect molecular systems in an organism. In addition to the gene sets, different approaches integrate complex interaction information into GSEA and build network-based models [70]. In contrast to GSEA, pathway activity analysis considers transcriptome data and other omics data and structural information of a biological network. For example, PARADIGM uses pathway topology and integrates various omics in the analysis to infer a patient-specific status of pathways [94]. A benchmark study with pan-cancer data recently reveals that using network structure can show better performance [57]. In conclusion, while the loss of data is due to the incompleteness of biological networks, their integration improved performance and increased interpretability in many cases.
3.2. Tumor Heterogeneity Study with Network Information
The tumor heterogeneity can originate from two directions, clonal heterogeneity and tumor impurity. Clonal heterogeneity covers genomic alterations within the tumor [95]. While de novo mutations accumulate, the tumor obtains genomic alterations with an exclusive pattern. When these genomic alterations are projected on the pathway, it is possible to observe exclusive relationships among disease-related genes. For instance, the CoMEt and MEMo algorithms examine mutual exclusivity on protein–protein interaction networks [96,97]. Moreover, the relationship between genes can be essential for an organism. Therefore, models analyzing such alterations integrate network-based analysis [98].
In contrast, tumor purity is dependent on the tumor microenvironment, including immune-cell infiltration and stromal cells [99]. In tumor microenvironment studies, network-based models are applied, for instance, to find immune-related gene modules. Although the importance of the interaction between tumors and immune cells is well known, detailed mechanisms are still unclear. Thus, many recent NGS studies employ network-based models to investigate the underlying mechanism in tumor and immune reactions. For example, McGrail et al. identified a relationship between the DNA damage response protein and immune cell infiltration in cancer. The analysis is based on curated interaction pairs in a protein–protein interaction network [100]. Most recently, Darzi et al. discovered a prognostic gene module related to immune cell infiltration by using network-centric approaches [101]. Tu et al. presented a network-centric model for mining subnetworks of genes other than immune cell infiltration by considering tumor purity [102].
3.3. Drug Target Identification with Network Information
In drug target studies, network biology is integrated into pharmacology [103]. For instance, Yamanishi et al. developed novel computational methods to investigate the pharmacological space by integrating a drug-target protein network with genomics and chemical information. The proposed approaches investigated such drug-target network information to identify potential novel drug targets [104]. Since then, the field has continued to develop methods to study drug target and drug response integrating networks with chemical and multi-omic datasets. In a recent survey study by Chen et al., the authors compared 13 computational methods for drug response prediction. It turned out that gene expression profiles are crucial information for drug response prediction [105].
Moreover, drug-target studies are often extended to drug-repurposing studies. In cancer research, drug-repurposing studies aim to find novel interactions between non-cancer drugs and molecular features in cancer. Drug-repurposing (or repositioning) studies apply computational approaches and pathway-based models and aim at discovering potential new cancer drugs with a higher probability than de novo drug design [16,106]. Specifically, drug-repurposing studies can consider various areas of cancer research, such as tumor heterogeneity and synthetic lethality. As an example, Lee et al. found clinically relevant synthetic lethality interactions by integrating multiple screening NGS datasets [107]. This synthetic lethality and related-drug datasets can be integrated for an effective combination of anticancer therapeutic strategy with non-cancer drug repurposing.
4. Deep Learning in Cancer Research
DNN models develop rapidly and become more sophisticated. They have been frequently used in all areas of biomedical research. Initially, its development was facilitated by large-scale imaging and video data. While most data sets in the biomedical field would not typically be considered big data, the rapid data accumulation enabled by NGS made it suitable for the application of DNN models requiring a large amount of training data [108]. For instance, in 2019, Samiei et al. used TCGA-based large-scale cancer data as benchmark datasets for bioinformatics machine learning research such as Image-Net in the computer vision field [109]. Subsequently, large-scale public cancer data sets such as TCGA encouraged the wide usage of DNNs in the cancer domain [110]. Over the last decade, these state-of-the-art machine learning methods have been incorporated in many different biological questions [111].
In addition to public cancer databases such as TCGA, the genetic information of normal tissues is stored in well-curated databases such as GTEx [112] and 1000Genomes [113]. These databases are frequently used as control or baseline training data for deep learning [114]. Moreover, other non-curated large-scale data sources such as GEO (https://www.ncbi.nlm.nih.gov/geo/, accessed on 20 May 2021) can be leveraged to tackle critical aspects in cancer research. They store a large-scale of biological data produced under various experimental setups (Figure 1). Therefore, an integration of GEO data and other data requires careful preprocessing. Overall, an increasing amount of datasets facilitate the development of current deep learning in bioinformatics research [115].
4.1. Challenges for Deep Learning in Cancer Research
Many studies in biology and medicine used NGS and produced large amounts of data during the past few decades, moving the field to the big data era. Nevertheless, researchers still face a lack of data in particular when investigating rare diseases or disease states. Researchers have developed a manifold of potential solutions to overcome this lack of data challenges, such as imputation, augmentation, and transfer learning (Figure 3b). Data imputation aims at handling data sets with missing values [116]. It has been studied on various NGS omics data types to recover missing information [117]. It is known that gene expression levels can be altered by different regulatory elements, such as DNA-binding proteins, epigenomic modifications, and post-transcriptional modifications. Therefore, various models integrating such regulatory schemes have been introduced to impute missing omics data [118,119]. Some DNN-based models aim to predict gene expression changes based on genomics or epigenomics alteration. For instance, TDimpute aims at generating missing RNA-seq data by training a DNN on methylation data. They used TCGA and TARGET (https://ocg.cancer.gov/programs/target/data-matrix, accessed on 20 May 2021) data as proof of concept of the applicability of DNN for data imputation in a multi-omics integration study [120]. Because this integrative model can exploit information in different levels of regulatory mechanisms, it can build a more detailed model and achieve better performance than a model build on a single-omics dataset [117,121]. The generative adversarial network (GAN) is a DNN structure for generating simulated data that is different from the original data but shows the same characteristics [122]. GANs can impute missing omics data from other multi-omics sources. Recently, the GAN algorithm is getting more attention in single-cell transcriptomics because it has been recognized as a complementary technique to overcome the limitation of scRNA-seq [123]. In contrast to data imputation and generation, other machine learning approaches aim to cope with a limited dataset in different ways. Transfer learning or few-shot learning, for instance, aims to reduce the search space with similar but unrelated datasets and guide the model to solve a specific set of problems [124]. These approaches train models with data of similar characteristics and types but different data to the problem set. After pre-training the model, it can be fine-tuned with the dataset of interest [125,126]. Thus, researchers are trying to introduce few-shot learning models and meta-learning approaches to omics and translational medicine. For example, Select-ProtoNet applied the ProtoTypical Network [127] model to TCGA transcriptome data and classified patients into two groups according to their clinical status [128]. AffinityNet predicts kidney and uterus cancer subtypes with gene expression profiles [129].

Figure 3. (a) In various studies, NGS data transformed into different forms. The 2-D transformed form is for the convolution layer. Omics data is transformed into pathway level, GO enrichment score, or Functional spectra. (b) DNN application on different ways to handle lack of data. Imputation for missing data in multi-omics datasets. GAN for data imputation and in silico data simulation. Transfer learning pre-trained the model with other datasets and fine-tune. (c) Various types of information in biology. (d) Graph neural network examples. GCN is applied to aggregate neighbor information. (Created with BioRender.com).
4.2. Molecular Charactization with Network and DNN Model
DNNs have been applied in multiple areas of cancer research. For instance, a DNN model trained on TCGA cancer data can aid molecular characterization by identifying cancer driver genes. At the very early stage, Yuan et al. build DeepGene, a cancer-type classifier. They implemented data sparsity reduction methods and trained the DNN model with somatic point mutations [130]. Lyu et al. [131] and DeepGx [132] embedded a 1-D gene expression profile to a 2-D array by chromosome order to implement the convolution layer (Figure 3a). Other algorithms, such as the deepDriver, use k-nearest neighbors for the convolution layer. A predefined number of neighboring gene mutation profiles was the input for the convolution layer. It employed this convolution layer in a DNN by aggregating mutation information of the k-nearest neighboring genes [11]. Instead of embedding to a 2-D image, DeepCC transformed gene expression data into functional spectra. The resulting model was able to capture molecular characteristics by training cancer subtypes [14].
Another DNN model was trained to infer the origin of tissue from single-nucleotide variant (SNV) information of metastatic tumor. The authors built a model by using the TCGA/ICGC data and analyzed SNV patterns and corresponding pathways to predict the origin of cancer. They discovered that metastatic tumors retained their original cancer’s signature mutation pattern. In this context, their DNN model obtained even better accuracy than a random forest model [133] and, even more important, better accuracy than human pathologists [12].
4.3. Tumor Heterogeneity with Network and DNN Model
As described in Section 4.1, there are several issues because of cancer heterogeneity, e.g., tumor microenvironment. Thus, there are only a few applications of DNN in intratumoral heterogeneity research. For instance, Menden et al. developed ’Scaden’ to deconvolve cell types in bulk-cell sequencing data. ’Scaden’ is a DNN model for the investigation of intratumor heterogeneity. To overcome the lack of training datasets, researchers need to generate in silico simulated bulk-cell sequencing data based on single-cell sequencing data [134]. It is presumed that deconvolving cell types can be achieved by knowing all possible expressional profiles of the cell [36]. However, this information is typically not available. Recently, to tackle this problem, single-cell sequencing-based studies were conducted. Because of technical limitations, we need to handle lots of missing data, noises, and batch effects in single-cell sequencing data [135]. Thus, various machine learning methods were developed to process single-cell sequencing data. They aim at mapping single-cell data onto the latent space. For example, scDeepCluster implemented an autoencoder and trained it on gene-expression levels from single-cell sequencing. During the training phase, the encoder and decoder work as denoiser. At the same time, they can embed high-dimensional gene-expression profiles to lower-dimensional vectors [136]. This autoencoder-based method can produce biologically meaningful feature vectors in various contexts, from tissue cell types [137] to different cancer types [138,139].
4.4. Drug Target Identification with Networks and DNN Models
In addition to NGS datasets, large-scale anticancer drug assays enabled the training train of DNNs. Moreover, non-cancer drug response assay datasets can also be incorporated with cancer genomic data. In cancer research, a multidisciplinary approach was widely applied for repurposing non-oncology drugs to cancer treatment. This drug repurposing is faster than de novo drug discovery. Furthermore, combination therapy with a non-oncology drug can be beneficial to overcome the heterogeneous properties of tumors [85]. The deepDR algorithm integrated ten drug-related networks and trained deep autoencoders. It used a random-walk-based algorithm to represent graph information into feature vectors. This approach integrated network analysis with a DNN model validated with an independent drug-disease dataset [15].
The authors of CDRscan did an integrative analysis of cell-line-based assay datasets and other drug and genomics datasets. It shows that DNN models can enhance the computational model for improved drug sensitivity predictions [140]. Additionally, similar to previous network-based models, the multi-omics application of drug-targeted DNN studies can show higher prediction accuracy than the single-omics method. MOLI integrated genomic data and transcriptomic data to predict the drug responses of TCGA patients [141].
4.5. Graph Neural Network Model
In general, the advantage of using a biological network is that it can produce more comprehensive and interpretable results from high-dimensional omics data. Furthermore, in an integrative multi-omics data analysis, network-based integration can improve interpretability over traditional approaches. Instead of pre-/post-integration of a network, recently developed graph neural networks use biological networks as the base structure for the learning network itself. For instance, various pathways or interactome information can be integrated as a learning structure of a DNN and can be aggregated as heterogeneous information. In a GNN study, a convolution process can be done on the provided network structure of data. Therefore, the convolution on a biological network made it possible for the GNN to focus on the relationship among neighbor genes. In the graph convolution layer, the convolution process integrates information of neighbor genes and learns topological information (Figure 3d). Consequently, this model can aggregate information from far-distant neighbors, and thus can outperform other machine learning models [142].
In the context of the inference problem of gene expression, the main question is whether the gene expression level can be explained by aggregating the neighboring genes. A single gene inference study by Dutil et al. showed that the GNN model outperformed other DNN models [143]. Moreover, in cancer research, such GNN models can identify cancer-related genes with better performance than other network-based models, such as HotNet2 and MutSigCV [144]. A recent GNN study with a multi-omics integrative analysis identified 165 new cancer genes as an interactive partner for known cancer genes [145]. Additionally, in the synthetic lethality area, dual-dropout GNN outperformed previous bioinformatics tools for predicting synthetic lethality in tumors [146]. GNNs were also able to classify cancer subtypes based on pathway activity measures with RNA-seq data. Lee et al. implemented a GNN for cancer subtyping and tested five cancer types. Thus, the informative pathway was selected and used for subtype classification [147]. Furthermore, GNNs are also getting more attention in drug repositioning studies. As described in Section 3.3, drug discovery requires integrating various networks in both chemical and genomic spaces (Figure 3d). Chemical structures, protein structures, pathways, and other multi-omics data were used in drug-target identification and repurposing studies (Figure 3c). Each of the proposed applications has a specialty in the different purposes of drug-related tasks. Sun et al. summarized GNN-based drug discovery studies and categorized them into four classes: molecular property and activity prediction, interaction prediction, synthesis prediction, and de novo drug design. The authors also point out four challenges in the GNN-mediated drug discovery. At first, as we described before, there is a lack of drug-related datasets. Secondly, the current GNN models can not fully represent 3-D structures of chemical molecules and protein structures. The third challenge is integrating heterogeneous network information. Drug discovery usually requires a multi-modal integrative analysis with various networks, and GNNs can improve this integrative analysis. Lastly, although GNNs use graphs, stacked layers still make it hard to interpret the model [148].
4.6. Shortcomings in AI and Revisiting Validity of Biological Networks as Prior Knowledge
The previous sections reviewed a variety of DNN-based approaches that present a good performance on numerous applications. However, it is hardly a panacea for all research questions. In the following, we will discuss potential limitations of the DNN models. In general, DNN models with NGS data have two significant issues: (i) data requirements and (ii) interpretability. Usually, deep learning needs a large proportion of training data for reasonable performance which is more difficult to achieve in biomedical omics data compared to, for instance, image data. Today, there are not many NGS datasets that are well-curated and -annotated for deep learning. This can be an answer to the question of why most DNN studies are in cancer research [110,149]. Moreover, the deep learning models are hard to interpret and are typically considered as black-boxes. Highly stacked layers in the deep learning model make it hard to interpret its decision-making rationale. Although the methodology to understand and interpret deep learning models has been improved, the ambiguity in the DNN models’ decision-making hindered the transition between the deep learning model and translational medicine [149,150].
As described before, biological networks are employed in various computational analyses for cancer research. The studies applying DNNs demonstrated many different approaches to use prior knowledge for systematic analyses. Before discussing GNN application, the validity of biological networks in a DNN model needs to be shown. The LINCS program analyzed data of ’The Connectivity Map (CMap) project’ to understand the regulatory mechanism in gene expression by inferring the whole gene expression profiles from a small set of genes (https://lincsproject.org/, accessed on 20 May 2021) [151,152]. This LINCS program found that the gene expression level is inferrable with only nearly 1000 genes. They called this gene list ’landmark genes’. Subsequently, Chen et al. started with these 978 landmark genes and tried to predict other gene expression levels with DNN models. Integrating public large-scale NGS data showed better performance than the linear regression model. The authors conclude that the performance advantage originates from the DNN’s ability to model non-linear relationships between genes [153].
Following this study, Beltin et al. extensively investigated various biological networks in the same context of the inference of gene expression level. They set up a simplified representation of gene expression status and tried to solve a binary classification task. To show the relevance of a biological network, they compared various gene expression levels inferred from a different set of genes, neighboring genes in PPI, random genes, and all genes. However, in the study incorporating TCGA and GTEx datasets, the random network model outperformed the model build on a known biological network, such as StringDB [154]. While network-based approaches can add valuable insights to analysis, this study shows that it cannot be seen as the panacea, and a careful evaluation is required for each data set and task. In particular, this result may not represent biological complexity because of the oversimplified problem setup, which did not consider the relative gene-expressional changes. Additionally, the incorporated biological networks may not be suitable for inferring gene expression profiles because they consist of expression-regulating interactions, non-expression-regulating interactions, and various in vivo and in vitro interactions.
“ However, although recently sophisticated applications of deep learning showed improved accuracy, it does not reflect a general advancement. Depending on the type of NGS data, the experimental design, and the question to be answered, a proper approach and specific deep learning algorithms need to be considered. Deep learning is not a panacea. In general, to employ machine learning and systems biology methodology for a specific type of NGS data, a certain experimental design, a particular research question, the technology, and network data have to be chosen carefully.”
References
- Janes, K.A.; Yaffe, M.B. Data-driven modelling of signal-transduction networks. Nat. Rev. Mol. Cell Biol. 2006, 7, 820–828. [Google Scholar] [CrossRef] [PubMed]
- Kreeger, P.K.; Lauffenburger, D.A. Cancer systems biology: A network modeling perspective. Carcinogenesis 2010, 31, 2–8. [Google Scholar] [CrossRef] [PubMed]
- Vucic, E.A.; Thu, K.L.; Robison, K.; Rybaczyk, L.A.; Chari, R.; Alvarez, C.E.; Lam, W.L. Translating cancer ‘omics’ to improved outcomes. Genome Res. 2012, 22, 188–195. [Google Scholar] [CrossRef]
- Hoadley, K.A.; Yau, C.; Wolf, D.M.; Cherniack, A.D.; Tamborero, D.; Ng, S.; Leiserson, M.D.; Niu, B.; McLellan, M.D.; Uzunangelov, V.; et al. Multiplatform analysis of 12 cancer types reveals molecular classification within and across tissues of origin. Cell 2014, 158, 929–944. [Google Scholar] [CrossRef] [PubMed]
- Hutter, C.; Zenklusen, J.C. The cancer genome atlas: Creating lasting value beyond its data. Cell 2018, 173, 283–285. [Google Scholar] [CrossRef]
- Chuang, H.Y.; Lee, E.; Liu, Y.T.; Lee, D.; Ideker, T. Network-based classification of breast cancer metastasis. Mol. Syst. Biol. 2007, 3, 140. [Google Scholar] [CrossRef]
- Zhang, W.; Chien, J.; Yong, J.; Kuang, R. Network-based machine learning and graph theory algorithms for precision oncology. NPJ Precis. Oncol. 2017, 1, 25. [Google Scholar] [CrossRef] [PubMed]
- Ngiam, K.Y.; Khor, W. Big data and machine learning algorithms for health-care delivery. Lancet Oncol. 2019, 20, e262–e273. [Google Scholar] [CrossRef]
- Creixell, P.; Reimand, J.; Haider, S.; Wu, G.; Shibata, T.; Vazquez, M.; Mustonen, V.; Gonzalez-Perez, A.; Pearson, J.; Sander, C.; et al. Pathway and network analysis of cancer genomes. Nat. Methods 2015, 12, 615. [Google Scholar]
- Reyna, M.A.; Haan, D.; Paczkowska, M.; Verbeke, L.P.; Vazquez, M.; Kahraman, A.; Pulido-Tamayo, S.; Barenboim, J.; Wadi, L.; Dhingra, P.; et al. Pathway and network analysis of more than 2500 whole cancer genomes. Nat. Commun. 2020, 11, 729. [Google Scholar] [CrossRef]
- Luo, P.; Ding, Y.; Lei, X.; Wu, F.X. deepDriver: Predicting cancer driver genes based on somatic mutations using deep convolutional neural networks. Front. Genet. 2019, 10, 13. [Google Scholar] [CrossRef]
- Jiao, W.; Atwal, G.; Polak, P.; Karlic, R.; Cuppen, E.; Danyi, A.; De Ridder, J.; van Herpen, C.; Lolkema, M.P.; Steeghs, N.; et al. A deep learning system accurately classifies primary and metastatic cancers using passenger mutation patterns. Nat. Commun. 2020, 11, 728. [Google Scholar] [CrossRef]
- Chaudhary, K.; Poirion, O.B.; Lu, L.; Garmire, L.X. Deep learning–based multi-omics integration robustly predicts survival in liver cancer. Clin. Cancer Res. 2018, 24, 1248–1259. [Google Scholar] [CrossRef]
- Gao, F.; Wang, W.; Tan, M.; Zhu, L.; Zhang, Y.; Fessler, E.; Vermeulen, L.; Wang, X. DeepCC: A novel deep learning-based framework for cancer molecular subtype classification. Oncogenesis 2019, 8, 44. [Google Scholar] [CrossRef]
- Zeng, X.; Zhu, S.; Liu, X.; Zhou, Y.; Nussinov, R.; Cheng, F. deepDR: A network-based deep learning approach to in silico drug repositioning. Bioinformatics 2019, 35, 5191–5198. [Google Scholar] [CrossRef]
- Issa, N.T.; Stathias, V.; Schürer, S.; Dakshanamurthy, S. Machine and deep learning approaches for cancer drug repurposing. In Seminars in Cancer Biology; Elsevier: Amsterdam, The Netherlands, 2020. [Google Scholar]
- Weinstein, J.N.; Collisson, E.A.; Mills, G.B.; Shaw, K.R.M.; Ozenberger, B.A.; Ellrott, K.; Shmulevich, I.; Sander, C.; Stuart, J.M.; Network, C.G.A.R.; et al. The cancer genome atlas pan-cancer analysis project. Nat. Genet. 2013, 45, 1113. [Google Scholar] [CrossRef]
- The ICGC/TCGA Pan-Cancer Analysis of Whole Genomes Consortium. Pan-cancer analysis of whole genomes. Nature 2020, 578, 82. [Google Scholar] [CrossRef] [PubMed]
- King, M.C.; Marks, J.H.; Mandell, J.B. Breast and ovarian cancer risks due to inherited mutations in BRCA1 and BRCA2. Science 2003, 302, 643–646. [Google Scholar] [CrossRef] [PubMed]
- Courtney, K.D.; Corcoran, R.B.; Engelman, J.A. The PI3K pathway as drug target in human cancer. J. Clin. Oncol. 2010, 28, 1075. [Google Scholar] [CrossRef] [PubMed]
- Parker, J.S.; Mullins, M.; Cheang, M.C.; Leung, S.; Voduc, D.; Vickery, T.; Davies, S.; Fauron, C.; He, X.; Hu, Z.; et al. Supervised risk predictor of breast cancer based on intrinsic subtypes. J. Clin. Oncol. 2009, 27, 1160. [Google Scholar] [CrossRef]
- Yersal, O.; Barutca, S. Biological subtypes of breast cancer: Prognostic and therapeutic implications. World J. Clin. Oncol. 2014, 5, 412. [Google Scholar] [CrossRef] [PubMed]
- Zhao, L.; Lee, V.H.; Ng, M.K.; Yan, H.; Bijlsma, M.F. Molecular subtyping of cancer: Current status and moving toward clinical applications. Brief. Bioinform. 2019, 20, 572–584. [Google Scholar] [CrossRef] [PubMed]
- Jones, P.A.; Issa, J.P.J.; Baylin, S. Targeting the cancer epigenome for therapy. Nat. Rev. Genet. 2016, 17, 630. [Google Scholar] [CrossRef] [PubMed]
- Huang, S.; Chaudhary, K.; Garmire, L.X. More is better: Recent progress in multi-omics data integration methods. Front. Genet. 2017, 8, 84. [Google Scholar] [CrossRef]
- Chin, L.; Andersen, J.N.; Futreal, P.A. Cancer genomics: From discovery science to personalized medicine. Nat. Med. 2011, 17, 297. [Google Scholar] [CrossRef] [PubMed]
Use of Systems Biology in Anti-Microbial Drug Development
Genomics, Computational Biology and Drug Discovery for Mycobacterial Infections: Fighting the Emergence of Resistance. Asma Munir, Sundeep Chaitanya Vedithi, Amanda K. Chaplin and Tom L. Blundell. Front. Genet., 04 September 2020 | https://doi.org/10.3389/fgene.2020.00965
In an earlier review article (Waman et al., 2019), we discussed various computational approaches and experimental strategies for drug target identification and structure-guided drug discovery. In this review we discuss the impact of the era of precision medicine, where the genome sequences of pathogens can give clues about the choice of existing drugs, and repurposing of others. Our focus is directed toward combatting antimicrobial drug resistance with emphasis on tuberculosis and leprosy. We describe structure-guided approaches to understanding the impacts of mutations that give rise to antimycobacterial resistance and the use of this information in the design of new medicines.
Genome Sequences and Proteomic Structural Databases
In recent years, there have been many focused efforts to define the amino-acid sequences of the M. tuberculosis pan-genome and then to define the three-dimensional structures and functional interactions of these gene products. This work has led to essential genes of the bacteria being revealed and to a better understanding of the genetic diversity in different strains that might lead to a selective advantage (Coll et al., 2018). This will help with our understanding of the mode of antibiotic resistance within these strains and aid structure-guided drug discovery. However, only ∼10% of the ∼4128 proteins have structures determined experimentally.
Several databases have been developed to integrate the genomic and/or structural information linked to drug resistance in Mycobacteria (Table 1). These invaluable resources can contribute to better understanding of molecular mechanisms involved in drug resistance and improvement in the selection of potential drug targets.
There is a dearth of information related to structural aspects of proteins from M. leprae and their oligomeric and hetero-oligomeric organization, which has limited the understanding of physiological processes of the bacillus. The structures of only 12 proteins have been solved and deposited in the protein data bank (PDB). However, the high sequence similarity in protein coding genes between M. leprae and M. tuberculosis allows computational methods to be used for comparative modeling of the proteins of M. leprae. Mainly monomeric models using single template modeling have been defined and deposited in the Swiss Model repository (Bienert et al., 2017), in Modbase (Pieper et al., 2014), and in a collection with other infectious disease agents (Sosa et al., 2018). There is a need for multi-template modeling and building homo- and hetero-oligomeric complexes to better understand the interfaces, druggability and impacts of mutations.
We are now exploiting Vivace, a multi-template modeling pipeline developed in our lab for modeling the proteomes of M. tuberculosis (CHOPIN, see above) and M. abscessus [Mabellini Database (Skwark et al., 2019)], to model the proteome of M. leprae. We emphasize the need for understanding the protein interfaces that are critical to function. An example of this is that of the RNA-polymerase holoenzyme complex from M. leprae. We first modeled the structure of this hetero-hexamer complex and later deciphered the binding patterns of rifampin (Vedithi et al., 2018; Figures 1A,B). Rifampin is a known drug to treat tuberculosis and leprosy. Owing to high rifampin resistance in tuberculosis and emerging resistance in leprosy, we used an approach known as “Computational Saturation Mutagenesis”, to identify sites on the protein that are less impacted by mutations. In this study, we were able to understand the association between predicted impacts of mutations on the structure and phenotypic rifampin-resistance outcomes in leprosy.
FIGURE 2

Figure 2. (A) Stability changes predicted by mCSM for systematic mutations in the ß-subunit of RNA polymerase in M. leprae. The maximum destabilizing effect from among all 19 possible mutations at each residue position is considered as a weighting factor for the color map that gradients from red (high destabilizing effects) to white (neutral to stabilizing effects) (Vedithi et al., 2020). (B) One of the known mutations in the ß-subunit of RNA polymerase, the S437H substitution which resulted in a maximum destabilizing effect [-1.701 kcal/mol (mCSM)] among all 19 possibilities this position. In the mutant, histidine (residue in green) forms hydrogen bonds with S434 and Q438, aromatic interactions with F431, and other ring-ring and π interactions with the surrounding residues which can impact the shape of the rifampin binding pocket and rifampin affinity to the ß-subunit [-0.826 log(affinity fold change) (mCSM-lig)]. Orange dotted lines represent weak hydrogen bond interactions. Ring-ring and intergroup interactions are depicted in cyan. Aromatic interactions are represented in sky-blue and carbonyl interactions in pink dotted lines. Green dotted lines represent hydrophobic interactions (Vedithi et al., 2020).
Examples of Understanding and Combatting Resistance
The availability of whole genome sequences in the present era has greatly enhanced the understanding of emergence of drug resistance in infectious diseases like tuberculosis. The data generated by the whole genome sequencing of clinical isolates can be screened for the presence of drug-resistant mutations. A preliminary in silico analysis of mutations can then be used to prioritize experimental work to identify the nature of these mutations.
FIGURE 3

Figure 3. (A) Mechanism of isoniazid activation and INH-NAD adduct formation. (B) Mutations mapped (Munir et al., 2019) on the structure of KatG (PDB ID:1SJ2; Bertrand et al., 2004).
Other articles related to Computational Biology, Systems Biology, and Bioinformatics on this online journal include:
Quantum Biology And Computational Medicine




 Jing Wei
Jing Wei Yueyao Yang
Yueyao Yang Gang Wang2 and
Gang Wang2 and  Ming Liu
Ming Liu