The Vibrant Philly Biotech Scene: Recent Happenings & Deals
Curator: Stephen J. Williams, Ph.D.
As the office and retail commercial real estate market has been drying up since the COVID pandemic, commercial real estate developers in the Philadelphia area have been turning to the health science industry to suit their lab space needs. This includes refurbishing old office space as well as new construction.
Gattuso secures $290M construction loan for life sciences building on Drexel campus

By Ryan Mulligan – Reporter, Philadelphia Business Journal
Dec 19, 2022
Gattuso Development Partners and Vigilant Holdings of New York have secured a $290 million construction loan for a major life sciences building set to be developed on Drexel University’s campus.
The funding comes from Houston-based Corebridge Financial, with an additional equity commitment from Boston-based Baupost Group, which is also a partner on the project. JLL’s Capital Markets group arranged the loan.
Plans for the University City project at 3201 Cuthbert St. carry a price tag of $400 million. The 11-story building will total some 520,000 square feet, making it the largest life sciences research and lab space in the city when it comes online.
The building at 3201 Cuthbert will rise on what had served as a recreation field used by Drexel and is located next to the Armory. Gattuso Development, which will lease the parcel from Drexel, expects to to complete the project by fall 2024. Robert A.M. Stern Architects designed the building.

A rendering of a $400 million lab and research facility Drexel University and Gattuso Development Partners plan to build at 3201 Cuthbert St. in Philadelphia.
The building is 45% leased by Drexel and SmartLabs, an operator of life sciences labs. Drexel plans to occupy about 60,000 square feet, while SmartLabs will lease two floors totaling 117,000 square feet.
“We believe the project validates Philadelphia’s emergence as a global hub for life sciences research, and we are excited to begin construction,” said John Gattuso, the co-founder and president of Philadelphia-based Gattuso Development.
Ryan Ade, Brett Segal and Christopher Peck of JLL arranged the financing.
The project is another play in what amounts to an arms race for life sciences space and tenants in University City. Spark Therapeutics plans to build a $575 million, 500,000-square-foot gene therapy manufacturing plant on Drexel’s campus. One uCity Square, a $280 million, 400,000-square-foot life sciences building, was recently completed at 38th and Market streets. At 3151 Market St., a $307 million, 417,000-square-foot life sciences building is proposed as part of the Schuylkill Yards development.
Tmunity CEO Usman Azam departing to lead ‘stealth’ NYC biotech firm

By John George – Senior Reporter, Philadelphia Business Journal
Feb 7, 2022
The CEO of one of Philadelphia’s oldest cell therapy companies is departing to take a new job in the New York City area.
Usman “Oz” Azam, who has been CEO of Tmunity Therapeutics since 2016, will lead an unnamed biotechnology company currently operating in stealth mode.
In a posting on his LinkedIn page, Azam said, “After a decade immersed in cell therapies and immuno-oncology, I am now turning my attention to a new opportunity, and will be going back to where I started my life sciences career in neurosciences.”
Tmunity, a University of Pennsylvania spinout, is looking to apply CAR T-cell therapy, which has proved to be successful in treating liquid cancers, for the treatment of solid tumors.
Last summer, Tmunity suspended clinical testing of its lead cell therapy candidate targeting prostate cancer after two patients in the study died. Azam, in an interview with the Business Journal in June, said the company, which had grown to about 50 employees since its launch in 2015, laid off an undisclosed number of employees as a result of the setback.
Azam said on LinkedIn he is still a big believer in CAR T-cell therapy, noting Tmunity co-founder Dr. Carl June and his colleagues at Penn just published in Nature the 10-year landmark clinical outcomes study with the first CD19 CAR-T patients and programs.
“It’s just the beginning,” he stated. “I’m excited about the prospect of so many new cell- and gene-based therapies emerging in the next five to 10 years to tackle many solid and liquid tumors, and I hope we all continue to see the remarkable impact this makes on patients and families around the world.”
Azam could not be reached for comment Monday. Tmunity has engaged a search firm to identify his successor.
Tmunity, which is based in Philadelphia, has its own manufacturing operations in East Norriton. Tmunity’s founders include June and fellow Penn cell therapy pioneer Bruce Levine, who led the development of a CAR T-cell therapy now marketed by Novartis as Kymriah, a treatment for certain types of blood cancers.
In therapy using CAR-T cells, a patient’s T cells — part of their immune system — are removed and genetically modified in the laboratory. After they are re-injected into a patient, the T cells are better able to attack and destroy tumors. CAR is an acronym for chimeric antigen receptor. Chimeric antigen receptors are receptor proteins that have been engineered to give T cells their improved ability to target tumors.
PIDC names U.S. Department of Treasury veteran, Philadelphia native as next president
The Philadelphia Industrial Development Corp. has tapped U.S. Department of Treasury veteran Jodie Harris to be its next president.
Harris succeeds Anne Bovaird Nevins, who spent 15 years in the organization and took over as president in January 2020 before stepping down at the end of last year. Executive Vice President Sam Rhoads has been interim president.
Harris, a Philadelphia native who currently serves as director of the Community Development Financial Institutions Fund for the Department of Treasury, was picked after a regional and national search and will begin her tenure as president on June 1. She becomes the 12th head of PIDC and the first African-American woman to lead the organization.
PIDC is a public-private economic development corporation founded by the city and the Chamber of Commerce for Greater Philadelphia in 1958. It mainly uses industrial and commercial real estate projects to attract jobs, foster business opportunities and spur overall community growth. The organization has spurred over $18.5 billion in financing across its 65 years.
PIDC has its hand in development projects spanning the city, including master planning roles in expansive campuses like the Philadelphia Navy Yard and the Lower Schuylkill Biotech Campus in Southwest Philadelphia.
In a statement, Harris said that it is “a critical time for Philadelphia’s economy.”
“I’m especially excited for the opportunity to lead such an important and impactful organization in my hometown of Philadelphia,” Harris said. “As head of the CDFI Fund, I know first-hand what it takes to drive meaningful, sustainable, and equitable economic growth, especially in historically underserved communities.”
Harris is a graduate of the University of Maryland and received an MBA and master of public administration from New York University. In the Treasury Department, Harris’ most recent work aligns with PIDC’s economic development mission. At the Community Development Financial Institutions Fund, she oversaw a $331 million budget, mainly comprised of grant and administrative funding for various economic programs. Under Harris’ watch, the fund distributed over $3 billion in pandemic recovery funding, its highest level of appropriated grants ever.
Harris has been a part of the Treasury Department for 15 years, including as director of community and economic development policy.
In addition to government work, Harris has previously spent time in the private, academia and nonprofit sectors. In the beginning of her career, Harris worked at Meridian Bank and Accenture before turning to become a social and education policy researcher at New York University. She also spent two years as president of the Urban Business Assistance Corporation in New York.
Mayor Jim Kenney said that Philadelphia is “poised for long-term growth” and Harris will help drive it.
Source: https://www.bizjournals.com/philadelphia/news/2023/02/23/pidc-names-next-president-treasury.html
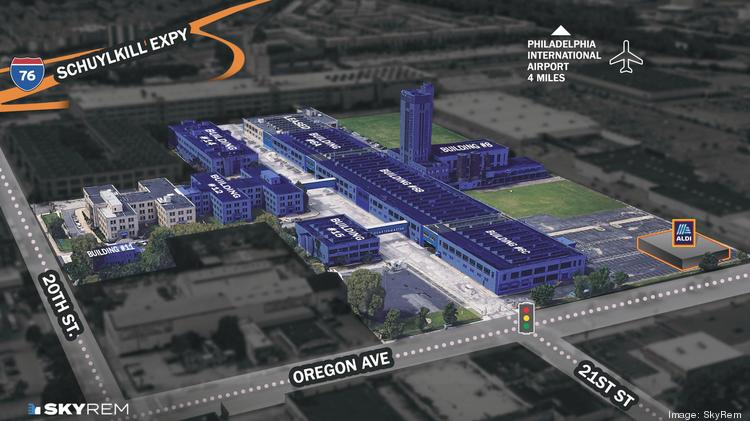
$250M life sciences conversion planned for Philadelphia’s historic Quartermaster site
Listen to this article 3 min
Real estate company SkyREM plans to spend $250 million converting the historic Quartermaster site in South Philadelphia to a life sciences campus with restaurants and a hotel.
The redevelopment would feature wet and dry lab space for research, development and bio-manufacturing.
The renamed Quartermaster Science + Technology Park is near the southwest corner of Oregon Avenue and South 20th Street in the city’s Girard Estates neighborhood. It’s east of the Quartermaster Plaza retail center, which sold last year for $100 million.
The 24-acre campus is planned to have six acres of green space, an Aldi grocery store opening by March and already is the headquarters for Indego, the bicycle share program in Philadelphia.
Six buildings totaling 1 million square feet of space would be used for research and development labs. There’s 500,000 square feet of vacant space available for life sciences and high technology companies with availabilities as small as 1,000 square feet up to 250,000 square feet contiguous. There’s also 150,000 square feet of retail space available.
The office park has 200,000 square feet already occupied by tenants. The Philadelphia Job Corps Center and Delaware Valley Intelligence Center are tenants at the site.
A rendering shows part of the Quartermaster Science + Technology Park as a redeveloped mixed-use life science campus.
The campus was previously used by the military as a place to produce clothing, footwear and personal equipment during World War I and II. The clothing factory closed in 1994. The Philadelphia Quartermaster Depot was listed on the National Register of Historic Places in 2010.
“We had a vision to preserve the legacy of this built-to-last historic Philadelphia landmark and transform it to create a vibrant space where the best and brightest want to innovate, collaborate, and work,” SkyREM CEO and Founder Alex Dembitzer said in a statement.
SkyREM, a real estate investor and developer, has corporate offices in New York and Philadelphia. The company acquired the site in 2001.
Vered Nohi, SkyREM’s regional executive director of new business development, called the redevelopment “transformational” for Philadelphia.
SkyREM announced the redevelopment of the Quartermaster campus in South Philadelphia into a life sciences campus with restaurants and a hotel. This rendering looks across Oregon Avenue toward the southwest corner of Oregon and 21st Street.
Quartermaster would join a wave of new life sciences projects being developed in the surrounding area and across the region.
The site is near both interstates 76 and 95 and is about 2 miles north of the Philadelphia Navy Yard, which has undergone a similar transformation from a military hub to a major life sciences and mixed-use redevelopment project. The Philadelphia Industrial Development Corp. is also in the process of selecting a developer to create a massive cell and gene therapy manufacturing complex across two sites totaling about 40 acres on Southwest Philadelphia’s Lower Schuylkill riverfront.
At 34th Street and Grays Ferry Avenue, the University of Pennsylvania is teaming with Longfellow Real Estate Partners on proposed a $365 million, 455,000-square-foot life sciences and biomanufacturing building at Pennovation Works.
A rendering shows part of the future Quartermaster Science and Technology Park in South Philadelphia. The 24-acre campus is planned to have six buildings with 1 million square feet of life science space.
SkyREM is working with Maryland real estate firm Scheer Partners to lease the science and technology space. Philadelphia’s MPN Realty will handle leasing of the retail space. Architecture firm Fifteen is working on the project’s design.
Scheer Partners Senior Vice President Tim Conrey said the Quartermaster conversion will help companies solve for “speed to market” as demand for life science space in the region has been strong.
A diagram shows the buildings that are leased (gray) and the buildings that are available (blue) at the Quartermaster site in South Philadelphia.
Brandywine pauses new spec office development, continues to bet big on life sciences
Brandywine Realty Trust originally planned to redevelop a Radnor medical office into lab and office space, split 50-50 between the two uses.
After changes in demand for lab and office space, Brandywine (NYSE: BDN) recently completed the 168,000-square-foot, four-story building at 250 King of Prussia Road in Radnor fully for life sciences.
“The pipeline is now 100% life sciences, which, while requiring more capital, is also generating longer term leases at a higher return on cost,” Brandywine CEO Jerry Sweeney of the project said during the company’s fourth-quarter earnings call on Thursday.
At the same time, Brandywine is holding off on developing new office buildings unless it has a tenant lined up in advance.
The shift reflects how Philadelphia-based Brandywine continues to lean into — and bet big — on life sciences.
Brandywine is the city’s largest owner of trophy office buildings and has several major development projects in the works. The company is planning to eventually develop 3 million square feet of life sciences space. For now, 800,000 square feet of life sciences space is under development, including a 12-story, 417,000-square-foot life sciences building at 3151 Market St. and a 29-story building with 200,000 square feet of life sciences space at 3025 John F. Kennedy Blvd. Both are part of the multi-phase Schuylkill Yards project underway near 30th Street Station in University City.
Once its existing projects are completed, Brandywine would have 800,000 square feet of life sciences space, making up 8% of its portfolio.Sweeney said the company wants to grow that figure to 21%.
Brandywine is developing a 145,000-square-foot, build-to-suit office building at 155 King of Prussia Road in Radnor for Arkema, a France-based global supplier of specialty materials. The building will be Arkema’s North American headquarters. Construction began in January and is scheduled to be completed in late 2024.
Brandywine reported that since November it raised over $705 million through fourth-quarter asset sales, an unsecured bond transaction and a secured loan. The company has “complete availability” on its $600 million unsecured line of credit, Sweeney said.
Brandywine sold a 95% leased, 86,000-square-foot office building at 200 Barr Harbor Drive in West Conshohocken for $30.5 million. The company also sold its 50% ownership interest in the 1919 Market joint venture for $83.2 million to an undisclosed buyer. 1919 Market St. is a 29-story building with apartments, office and commercial space. Brandywine co-developed the property with LCOR and the California State Teacher’s Retirement System.
Brandywine declined to comment and LCOR could not be reached.
Brandywine’s core portfolio is 91% leased.
The project at 250 King of Prussia Road cost $103.7 million and was recently completed. The renovation included 12-foot high floor-to-ceiling glass on the second floor, a new roof, lobby, elevator core, common area with a skylight and an added structured parking deck.
Located in the Radnor Life Science Center, a new campus with nearly 1 million square feet of lab, research and office space, Sweeney said it’s a “magnet” for biotech companies. Avantor, a global manufacturer and distributor of life sciences products, is headquartered in the complex.
Sweeney said Brandywine is “very confident” demand will stay strong for life sciences in Radnor. The building at 250 King of Prussia Road is projected to be fully leased by early 2024.
“Larger users we’re talking to, they just tend to take a little bit more time than we would like as they go through technical requirements and space planning requirements,” Sweeney said.
Jerry Sweeney, CEO of Brandywine Realty Trust.
While Brandywine is aiming to increase its life sciences footprint, the company is being selective about what it builds next. The company may steer away from developments other than life sciences. The Schuylkill Yards project, for example, features a significant life sciences portion in University City.
“Other than fully leased build-to-suit opportunities, our future development starts are on hold,” Sweeney said, “pending more leasing on the existing joint venture pipeline and more clarity on the cost of debt capital and cap rates.”
Brandywine said about 70% to 75%of suburban tenants have returned to offices while that number has been around 50% in Philadelphia. At this point, though, it hasn’t yet affected demand when leasing space. Some tenants, for example, have moved out of the city while others have moved in.
In the fourth quarter, Brandywine had $55.7 million funds from operations, or 32 cents per share. That’s down from $60.4 million, or 35 cents per share, in the fourth quarter of 2021. Brandywine generated $129 million in revenue in the fourth quarter, up slightly from $125.5 in the year-ago period.
Brandywine stock is up 6.4% since the start of the year to $6.70 per share on Monday afternoon.
Many of Brandywine’s properties are in desirable locations, which have seen demand remain strong despite challenges facing offices, on par with industry trends.
Brandywine’s 12-story, 417,000-square-foot building at 3151 Market St. is on budget for $308 million and on schedule to be completed in the second quarter of 2024. Sweeney said Brandywine anticipates entering a construction loan in the second half of 2023, which would help complete the project. The building, being developed along with a global institutional investor,would be used for life sciences, innovation and office space as part of the larger Schuylkill Yards development in University City.
The company’s 29-story building at 3025 John F. Kennedy Blvd. with 200,000 square feet of life sciences space and 326 luxury apartments, is also on budget, costing $287.3 million, and on time, eyeing completion in the third quarter of this year.

















 As you may already know, starting today, the Biotechnology Industry Organization will become the Biotechnology Innovation Organization. It’s a one-word name change – from industry to innovation – but the implications are substantial.
As you may already know, starting today, the Biotechnology Industry Organization will become the Biotechnology Innovation Organization. It’s a one-word name change – from industry to innovation – but the implications are substantial.




