P13K delta-gamma anticancer agent
Larry H. Bernstein, MD, FCAP, Curator
LPBI
RP 6350, Rhizen Pharmaceuticals S.A. and Novartis tieup for Rhizen’s inhaled dual Pl3K-delta gamma inhibitor
by DR ANTHONY MELVIN CRASTO Ph.D

(A) and (Al) and (A2)
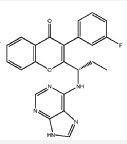
(S)-2-(l-(9H-purin-6-ylamino)propyl)-3-(3-fluorophenyl)-4H-chromen-4-one (Compound A1 is RP 6350).
RP 6350, RP6350, RP-6350
(S)-2-(l-(9H-purin-6-ylamino)propyl)-3-(3-fluorophenyl)-4H-chromen-4-one
mw 415
Rhizen Pharmaceuticals is developing RP-6530, a PI3K delta and gamma dual inhibitor, for the potential oral treatment of cancer and inflammation In November 2013, a phase I trial in patients with hematologic malignancies was initiated in Italy ]\. In September 2015, a phase I/Ib study was initiated in the US, in patients with relapsed and refractory T-cell lymphoma. At that time, the study was expected to complete in December 2016
PATENTS……..WO 11/055215 , WO 12/151525.
- Antineoplastics; Small molecules
- Mechanism of Action Phosphatidylinositol 3 kinase delta inhibitors; Phosphatidylinositol 3 kinase gamma inhibitors
- Phase I Haematological malignancies
- Preclinical Multiple myeloma
| Swaroop K. V. S. Vakkalanka, | |
| COMPANY | Rhizen Pharmaceuticals Sa |
https://clinicaltrials.gov/ct2/show/NCT02017613
PI3K delta/gamma inhibitor RP6530 An orally active, highly selective, small molecule inhibitor of the delta and gamma isoforms of phosphoinositide-3 kinase (PI3K) with potential immunomodulating and antineoplastic activities. Upon administration, PI3K delta/gamma inhibitor RP6530 inhibits the PI3K delta and gamma isoforms and prevents the activation of the PI3K/AKT-mediated signaling pathway. This may lead to a reduction in cellular proliferation in PI3K delta/gamma-expressing tumor cells. In addition, this agent modulates inflammatory responses through various mechanisms, including the inhibition of both the release of reactive oxygen species (ROS) from neutrophils and tumor necrosis factor (TNF)-alpha activity. Unlike other isoforms of PI3K, the delta and gamma isoforms are overexpressed primarily in hematologic malignancies and in inflammatory and autoimmune diseases. By selectively targeting these isoforms, PI3K signaling in normal, non-neoplastic cells is minimally impacted or not affected at all, which minimizes the side effect profile for this agent. Check for active clinical trials using this agent. (NCI Thesaurus)
| Company | Rhizen Pharmaceuticals S.A. |
| Description | Dual phosphoinositide 3-kinase (PI3K) delta and gamma inhibitor |
| Molecular Target | Phosphoinositide 3-kinase (PI3K) delta ; Phosphoinositide 3-kinase (PI3K) gamma |
| Mechanism of Action | Phosphoinositide 3-kinase (PI3K) delta inhibitor; Phosphoinositide 3-kinase (PI3K) gamma inhibitor |
| Therapeutic Modality | Small molecule |
RP6530 is a potent and selective dual PI3Kδ/γ inhibitor that inhibited growth of B-cell lymphoma cell lines with a concomitant reduction in the downstream biomarker, pAKT. Additionally, the compound showed cytotoxicity in a panel of lymphoma primary cells. Findings provide a rationale for future clinical trials in B-cell malignancies.
- Dual PI3K delta/gamma Inhibition By RP6530 Induces Apoptosis and Cytotoxicity
- RP6530, a novel, small molecule PI3K delta/gamma
- Activity and selectivity of RP6530 for PI3K delta and gamma isoforms
Introduction Activation of the PI3K pathway triggers multiple events including cell growth, cell cycle entry, cell survival and motility. While α and β isoforms are ubiquitous in their distribution, expression of δ and γ is restricted to cells of the hematopoietic system. Because these isoforms contribute to the development, maintenance, transformation, and proliferation of immune cells, dual targeting of PI3Kδ and γ represents a promising approach in the treatment of lymphomas. The objective of the experiments was to explore the therapeutic potential of RP6530, a novel, small molecule PI3Kδ/γ inhibitor, in B-cell lymphomas.
Methods Activity and selectivity of RP6530 for PI3Kδ and γ isoforms and subsequent downstream activity was determined in enzyme and cell-based assays. Additionally, RP6530 was tested for potency in viability, apoptosis, and Akt phosphorylation assays using a range of immortalized B-cell lymphoma cell lines (Raji, TOLEDO, KG-1, JEKO, OCI-LY-1, OCI-LY-10, MAVER, and REC-1). Viability was assessed using the colorimetric MTT reagent after incubation of cells for 72 h. Inhibition of pAKT was estimated by Western Blotting and bands were quantified using ImageJ after normalization with Actin. Primary cells from lymphoid tumors [1 chronic lymphocytic leukemia (CLL), 2 diffuse large B-cell lymphomas (DLBCL), 2 mantle cell lymphoma (MCL), 1 splenic marginal zone lymphoma (SMZL), and 1 extranodal MZL (EMZL)] were isolated, incubated with 4 µM RP6530, and analyzed for apoptosis or cytotoxicity by Annexin V/PI staining.
Results RP6530 demonstrated high potency against PI3Kδ (IC50=24.5 nM) and γ (IC50=33.2 nM) enzymes with selectivity over α (>300-fold) and β (>100-fold) isoforms. Cellular potency was confirmed in target-specific assays, namely anti-FcεR1-(EC50=37.8 nM) or fMLP (EC50=39.0 nM) induced CD63 expression in human whole blood basophils, LPS induced CD19+ cell proliferation in human whole blood (EC50=250 nM), and LPS induced CD45R+ cell proliferation in mouse whole blood (EC50=101 nM). RP6530 caused a dose-dependent inhibition (>50% @ 2-7 μM) in growth of immortalized (Raji, TOLEDO, KG-1, JEKO, REC-1) B-cell lymphoma cells. Effect was more pronounced in the DLBCL cell lines, OCI-LY-1 and OCI-LY-10 (>50% inhibition @ 0.1-0.7 μM), and the reduction in viability was accompanied by corresponding inhibition of pAKT with EC50 of 6 & 70 nM respectively. Treatment of patient-derived primary cells with 4 µM RP6530 caused an increase in cell death. Fold-increase in cytotoxicity as evident from PI+ staining was 1.6 for CLL, 1.1 for DLBCL, 1.2 for MCL, 2.2 for SMZL, and 2.3 for EMZL. Cells in early apotosis (Annexin V+/PI-) were not different between the DMSO blank and RP6530 samples.
Conclusions RP6530 is a potent and selective dual PI3Kδ/γ inhibitor that inhibited growth of B-cell lymphoma cell lines with a concomitant reduction in the downstream biomarker, pAKT. Additionally, the compound showed cytotoxicity in a panel of lymphoma primary cells. Findings provide a rationale for future clinical trials in B-cell malignancies.
Disclosures:Vakkalanka:Rhizen Pharmaceuticals, S.A.: Employment, Equity Ownership; Incozen Therapeutics Pvt. Ltd.: Employment, Equity Ownership.Viswanadha:Incozen Therapeutics Pvt. Ltd.: Employment. Bertoni:Rhizen Pharmaceuticals SA: Research Funding.
PI3K Dual Inhibitor (RP-6530)
| Therapeutic Area | Respiratory , Oncology – Liquid Tumors , Rheumatology | Molecule Type | Small Molecule |
|---|---|---|---|
| Indication | Peripheral T-cell lymphoma (PTCL) , Non-Hodgkins Lymphoma , Asthma , Chronic Obstructive Pulmonary Disease (COPD) , Rheumatoid Arthritis | ||
| Development Phase | Phase I | Rt. of Administration | Oral |
Description
Rhizen is developing dual PI3K gamma/delta inhibitors for liquid tumors and inflammatory conditions.
Mechanism of Action
While alpha and beta isoforms are ubiquitous in their distribution, expression of delta and gamma is restricted to circulating hematogenous cells and endothelial cells. Unlike PI3K-alpha or beta, mice lacking expression of gamma or delta do not show any adverse phenotype indicating that targeting of these specific isoforms would not result in overt toxicity. Dual delta/gamma inhibition is strongly implicated as an intervention strategy in allergic and non-allergic inflammation of the airways and other autoimmune diseases. Scientific evidence for PI3K-delta and gamma involvement in various cellular processes underlying asthma and COPD stems from inhibitor studies and gene-targeting approaches. Also, resistance to conventional therapies such as corticosteroids in several COPD patients has been attributed to an up-regulation of the PI3K delta/gamma pathway. Disruption of PI3K-delta/gamma signalling therefore provides a novel strategy aimed at counteracting the immuno-inflammatory response. Due to the pivotal role played by PI3K-delta and gamma in mediating inflammatory cell functionality such as leukocyte migration and activation, and mast cell degranulation, blocking these isoforms may also be an effective strategy for the treatment of rheumatoid arthritis as well.
Given the established criticality of these isoforms in immune surveillance, inhibitors specifically targeting the delta and gamma isoforms would be expected to attenuate the progression of immune response encountered in airway inflammation and rheumatoid arthritis.

http://www.rhizen.com/images/backgrounds/pi3k%20delta%20gamma%20ii.pngtps:/
Clinical Trials
Rhizen has identified an orally active Lead Molecule, RP-6530, that has an excellent pre-clinical profile. RP-6530 is currently in non-GLP Tox studies and is expected to enter Clinical Development in H2 2013.
In December 2013, Rhizen announced the start of a Phase I clinical trial. The study entitled A Phase-I, Dose Escalation Study to Evaluate Safety and Efficacy of RP6530, a dual PI3K delta /gamma inhibitor, in patients with Relapsed or Refractory Hematologic Malignancies is designed primarily to establish the safety and tolerability of RP6530. Secondary objectives include clinical efficacy assessment and biomarker response to allow dose determination and potential patient stratification in subsequent expansion studies.
Partners by Region
Rhizen’s pipeline consists of internally discovered (with 100% IP ownership) novel small molecule programs aimed at high value markets of Oncology, Immuno-inflammtion and Metabolic Disorders. Rhizen has been successful in securing critical IP space in these areas and efforts are on for further expansion in to several indications. Rhizen seeks partnerships to unlock the potential of these valuable assets for further development from global pharmaceutical partners. At present global rights on all programs are available and Rhizen is flexible to consider suitable business models for licensing/collaboration.
In 2012, Rhizen announced a joint venture collaboration with TG Therapeutics for global development and commercialization of Rhizen’s Novel Selective PI3K Kinase Inhibitors. The selected lead RP5264 (hereafter, to be developed as TGR-1202) is an orally available, small molecule, PI3K specific inhibitor currently being positioned for the treatment of hematological malignancies.
https://patentscope.wipo.int/search/en/detail.jsf?docId=WO2014195888
….Intermediates
- 01 Sep 2015 Phase-I clinical trials in Hematological malignancies (Second-line therapy or greater) in USA (PO) (NCT02567656)
- 18 Nov 2014 Preclinical trials in Multiple myeloma in Switzerland (PO) prior to November 2014
- 18 Nov 2014 Early research in Multiple myeloma in Switzerland (PO) prior to November 2014
| WO2011055215A2 | Nov 3, 2010 | May 12, 2011 | Incozen Therapeutics Pvt. Ltd. | Novel kinase modulators |
| WO2012151525A1 | May 4, 2012 | Nov 8, 2012 | Rhizen Pharmaceuticals Sa | Novel compounds as modulators of protein kinases |
| WO2013164801A1 | May 3, 2013 | Nov 7, 2013 | Rhizen Pharmaceuticals Sa | Process for preparation of optically pure and optionally substituted 2- (1 -hydroxy- alkyl) – chromen – 4 – one derivatives and their use in preparing pharmaceuticals |
| US20110118257 | May 19, 2011 | Rhizen Pharmaceuticals Sa | Novel kinase modulators | |
| US20120289496 | May 4, 2012 | Nov 15, 2012 | Rhizen Pharmaceuticals Sa | Novel compounds as modulators of protein kinases |
| WO 2011055215 |