Personalized Cardiovascular Genetic Medicine at Partners HealthCare and Harvard Medical School
Curator: Aviva Lev-Ari, PhD, RN
UPDATED on 5/4/2015
Goes to Clinic @MGH: Clinically validated versions of Exome Sequencing and Analysis using Broad-developed methods like Hybrid Capture, the Genome Analysis Toolkit (GATK), and MuTect
Center for Personalized Genetic Medicine, Partners HealthCare and Harvard Medical School
We are organized into four principal service areas:
- sequence analysis,
- genotyping,
- expression analysis, and
- bioprocessing/sample management
Our platforms include next generation sequencing with Illumina HiSeq2000 and GA ii analyzers as well as Sanger sequencing using ABI 3730 XL sequence analyzers. Targeted custom genotyping is offered using Sequenom and Illumina GoldenGate panels as well as GWAS scale projects using Illumina Infinium and DNA methylation analysis using Illumina bead arrays. Expression analysis is available with capabilities for processing total RNA on either Affymetrix or Illumina arrays.
Through services from our BioSample Services Facility (BSF) and Partners Biorepository for Medical Discovery (PBMD) teams we provide a research platform for handling samples in a standardized manner to provide consistency from sample to sample. The BSF is able to assist investigators to configure projects utilizing your own samples or coupled with the PCPGM-PBMD we are able to support the integration of cohorts of samples selected from the PBMD into analysis on our genomics platforms.
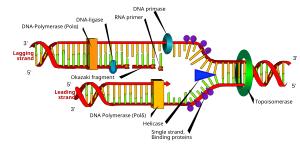
DNA Sequencing
The DNA Sequencing Group at the Partners HealthCare Center for Personalized Genetic Medicine has a strong history of producing high quality, dependable, and informative results for collaborators and clients. The DNA Sequencing Group participated in the Human Genome Project, building the STS-Based BAC map for Human Chromosome 12, and providing Chromosome 12 tiling path clones to the Baylor Human Genome Sequencing Center for sequencing.
The group sequenced 113 BACs for the Mouse Genome Project, contributing 24 megabases of finished mouse sequences to the published Mouse Genome, as well as providing draft sequences for unique strains of several bacterial genomes, including Pseudomonas aeruginosa, and Vibrio cholerae. More recently, the group participated in identifying mutations linked to numerous diseases, either in collaborations or by providing client laboratories with full service resequencing and analysis.
Services by Project Goals
Mutation Identification via Resequencing
This facility provides full-service resequencing of regions of interest in one or more genomic DNAs, including the following:
- Discussion of the scope of the project and a cost quote
- Identification of genes in the region of interest as needed, with the Investigator
- Primer design using our automated system, to amplify desired regions
- Primer ordering
- QC of the primers on DNA standards, if required
- PCR amplification of DNA provided by Investigator
- PCR clean-up
- Sequencing reactions
- Sequencing reaction clean-up
- Sequence application to the ABI 3730 XL Analyzer
- Chromatograms are made available to Investigator over web (GIGPAD)
- Data assembly and analysis using Phred Phrap and PolyPhred
- One round of repeats and redesign if necessary
- Report of variations found throughout sequence
- Trouble shooting for 100% coverage if desired
Research Services
- Fee-for-service sequencing
- Fragment analysis / genotyping (Microsatellite Instability)
Technology Development
- New technology testing and development
- Collaborative Protocol development
- Beta-test site for instrumentation and software
Clinical Diagnosis
- Diagnostic test development
- Sequencing for clinical diagnostics group
Genomic Sequencing Projects
- Human
- Rodent
- Bacteria
Advancing Translational Genomics through Personalized Medicine Projects
The mission of the Partners HealthCare Center for Personalized Genetic Medicine (PCPGM) is to utilize genetics and genomics to promote and implement personalized medicine in caring for patients throughout the Partners HealthCare system and in health care nationally and globally.
The Personalized Medicine Project program was developed to support the clinical research efforts of junior Partners HealthCare investigators for translational genetics and genomic projects to advance personalized medicine. The goal of this program is to identify biological markers that can be used as potential predictive tests. This will be accomplished by:
- Leveraging the Partners HealthCare Research Patient Data Registry (RPDR) and the Partners Biorepository for Medical Discovery (PBMD), centralized locations where Partners HealthCare patient data and/or samples are stored.
- Identifying novel biological markers or new uses for existing markers.
- Focusing on tests that could:
- improve diagnostic sensitivity or specificity;
- further stratify patient groups with a given diagnosis;
- predict improved clinical outcomes; or
- assist with selection of therapies or methods to manage disease.
Harvard Medical School Genetics Training Program
The Harvard Medical School (HMS) Genetics Training Program is one of the oldest and largest programs in the country. It was founded by Drs. John Littlefield at the Massachusetts General Hospital and Park Gerald at Children’s Hospital Boston in the early 1970’s. The program has trained scientists and clinicians who have become leaders in academic genetics, and has supported investigators who have made major contributions to the clinical practice of genetics and genetics research.
The HMS Genetics Training Program is accredited by the ABMG in all areas of training – Clinical Genetics, Biochemical Genetics, Cytogenetics, and Molecular Genetics. This provides an opportunity for our trainees to become active candidates for board certification in a discipline(s) of medical genetics in addition to receiving laboratory training. The training laboratories and clinics of the program are centered at HMS and its affiliated institutions including Brigham and Women’s Hospital (BWH), the HMS Department of Genetics, Beth Israel Deaconess Medical Center (BIDMC), Children’s Hospital Boston (CHB), Dana Farber Cancer Institute (DFCI), and Massachusetts General Hospital (MGH). The HMS Genetics Training Program provides trainees the opportunity to take advantage of the extraordinarily rich academic environment offered at HMS and its affiliated institutions as well as the greater Boston scientific community.
Cardiovascular Research Center @MGH
Cardiovascular Research Center investigators have made many groundbreaking discoveries. Among these include:
• first identification of progenitor cells in the heart
• cloning of the first vertebrate cell death genes
• knocking out the genes that produce nitric oxide (NO), showing the importance of this molecule to atherosclerosis and stroke
• clinical use of NO to treat patients with pulmonary hypertension
• development of gene and cell transfer approaches to treat heart failure
• performance of the first large-scale genetic screen in a vertebrate (the zebrafish)
• identification of genes critical to cardiac pacemaking, rhythm, contractile function, and normal heart patterning
• discovery of a new methylase gene responsible for altering DNA structure during an individual’s lifetime
The Cardiovascular Research Center has taken great pride in the training of scientists with MDs and/or PhDs, as well as graduate students from a variety of Boston area institutions.
The Cardiovascular Research Center has two locations, one in the Charlestown Navy Yard and the other on the main campus’s Charles River Plaza complex in the Richard Simches Research Center.
Both the Simches and Navy Yard sites offer state of the art facilities, including tissue culture rooms, warm and cold rooms, histology rooms, autoclave facilities, hot labs, scope rooms and conference rooms. The Navy Yard lab has a topnotch zebrafish facility that is utilized by many scientists both inside and outside the Center, and a transgenic mouse core for both knock-ins and knock-outs. The Navy Yard facilities also contain echocardiogram equipment, specialized microscopes equipped with video capability for making movies, as well as a confocal microscope available to the Center researchers. The Simches lab houses the CVRC Stem Cell Biology + Therapy program, including a dedicated facility for human ES cell based technology, run by Dr. Chad Cowan, and future plans for high throughput screening facility to allow chemical screening in ESX cell based systems. Other cores available to researchers include a Cell Sorting and Flow Cytometry lab and a DNA sequencing core.
The Cardiology Laboratory for Integrative Physiology & Imaging lab is dedicated to large animal studies. An in house interventional cardiologist specializing in large animals performs the surgeries. In addition there are technicians that assist in the daily operations of the lab and can assist in experiment design and project implementation. This lab specializes in large animal imaging, CAT scans and catheter base manipulations. There is also an MRI imaging facility housed in the lab.
http://www2.massgeneral.org/cvrc/about.html
Genomics and Cardiovascular Medicine @MGH
|
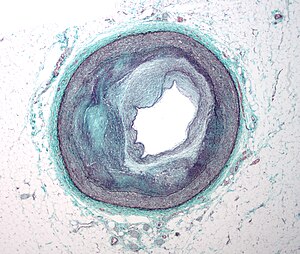
Genetic Basis of Cardiomyopathy
Original gene identification for Arrhythmogenic Right Ventricular Dysplasia/Cardiomyopathy, Autosomal Dominant
McNally E, MacLeod H, Dellefave L. Arrhythmogenic Right Ventricular Dysplasia/Cardiomyopathy, Autosomal Dominant. 2005 Apr 18 [Updated 2009 Oct 13]. In: Pagon RA, Bird TD, Dolan CR, et al., editors. GeneReviews™ [Internet]. Seattle (WA): University of Washington, Seattle; 1993-.
Summary
Disease characteristics. Autosomal dominant arrhythmogenic right ventricular dysplasia/cardiomyopathy (ARVD/C) is characterized by progressive fibrofatty replacement of the myocardium that predisposes to ventricular tachycardia and sudden death in young individuals and athletes. It primarily affects the right ventricle; with time, it may also involve the left ventricle. The presentation of disease is highly variable even within families, and affected individuals may not meet established clinical criteria. The mean age at diagnosis is 31 years (±13; range: 4-64 years).
http://www.ncbi.nlm.nih.gov/books/NBK1131/
Pan Cardiomyopathy Panel
@the Center for Personalized Genetic Medicine of Partners HealthCare and Harvard Medical School
The Pan Cardiomyopathy (PCM) Panel contains 51 cardiomyopathy genes including Titin (TTN), which encodes the largest human protein. This panel covers genes associated with HCM, DCM, RCM, LVNC, ARVC and CPVT and uses a combination of Next Generation Sequencing technology and conventional Sanger sequencing.
For illustrative reference, click to see one of our images or diagrams. Genes on Pan Cardiomyopathy Panels, Disease-Gene Associations, Gene Cellular Location.
Please select on the disease to read more: HCM,DCM, ARVC/CPVT, or LVNC.
Current Tests:
Pan Cardiomyopathy Panel – 51 genes
- HCM Panel – 18 genes§
- DCM Panel – 27 genes§
- ARVC/CPVT Panel – 8 genes§
- LVNC Panel – 10 genes§
§Optional reflex to remaining genes
Storage Cardiomyopathy – please select a disease to learn more
- Unexplained Hypertrophy Panel (LAMP2 & PRKAG2)
- Fabry Disease – GLA Gene Sequencing
- Transthyretin Amyloidosis – TTR Gene Sequencing
For any other single gene tests, please call the LMM at 617-768-8499 or lmm@partners.org.
For Variant Classification Rules – Lab for Molecular Medicine (LMM)
http://pcpgm.partners.org/sites/default/files/LMM/Resources/LMM_VariantClassification_05.26.11.pdf
For LMM Reference Sequences
http://pcpgm.partners.org/sites/default/files/LMM/Resources/LMMRefSeq-2.20.13.pdf
When to order which panel?
The Pan Cardiomyopathy panel may shorten the “testing odyssey” when a clear diagnosis has not been established. However, because many genes have not yet been associated with more than one cardiomyopathy, interpretation of novel variants may be more difficult when they are found in a gene that is not (yet) known to cause the patient’s cardiomyopathy. Please note: We are expecting an increase in “variants of unknown significance” and recommend careful consideration of the following factors when deciding whether to order the full panel or the disease specific sub-panels. The Pan Cardiomyopathy Panel may be best suited for patients who have already exhausted current testing options or whose clinical diagnosis is not yet clear. It may also be a good first line test for patients who have a family history where the number of living affected relatives would allow segregation analysis to establish or rule out pathogenicity for “variants of unknown significance (VUSs)”. Finally, the patient’s personal preferences should be considered as VUSs can cause anxiety.
Disease Backgrounds
Hypertrophic cardiomyopathy (HCM) is characterized by unexplained left ventricular hypertrophy (LVH) in a non-dilated ventricle. With a prevalence estimated to be ~1/500 in the general population, HCM is the most common monogenic cardiac disorder. To date, over 1000 variants have been identified in genes causative of HCM, most of which affect the sarcomere, the contractile unit of the cardiac muscle. In addition, defects in genes involved in storage diseases, such as LAMP2, PRKAG2 and GLA, typically cause systemic disease but may also result in predominant cardiac manifestations, which can mimic hypertrophic cardiomyopathy (HCM). For additional information about HCM, please visit GeneReviews.
Dilated cardiomyopathy (DCM) is characterized by ventricular chamber enlargement and systolic dysfunction with normal left ventricular wall thickness. The estimated prevalence of DCM is 1/2,500 and about 20-35% of cases have a family history showing a predominantly autosomal mode of inheritance. To date, over 40 genes have been demonstrated to cause DCM, encoding proteins involved in the sarcomere, Z-disk, nuclear lamina, intermediate filaments and the dystrophin-associated glycoprotein complex. Variants in some genes cause additional abnormalities: LMNA variants are frequently found in DCM that occurs with progressive conduction system disease. Variants in the TAZ gene cause Barth syndrome, an X-linked cardioskeletal myopathy in infants. In addition, variants in several genes (including LMNA, DES, SGCD, TCAP and EMD) can cause DCM in conjunction with skeletal myopathy. For additional information about DCM, please visit GeneReviews.
Arrhythmogenic Right Ventricular Cardiomyopathy (ARVC) is estimated to affect approximately 1/5,000 individuals in the general population, about half of which have a family history. The disease is characterized by replacement of myocytes by fatty or fibrofatty tissue, mainly in the right ventricle. The resulting manifestations are broad and include ventricular tachyarrhythmias and sudden death in young individuals and athletes. ARVC is typically inherited in an autosomal dominant fashion with incomplete penetrance and variable expressivity and to date, 5 ARVC genes (DSP, DSC2, DSG2, PKP2, TMEM43) have been identified, all but one (TMEM43) encode components of the desmosome. For more information about ARVC, please visit GeneReviews.
Catecholaminergic polymorphic ventricular tachycardia (CPVT) is typically characterized by exercise induced syncope due to ventricular tachycardia in individuals without structural heart disease. Two CPVT genes are known to date (RYR2 – autosomal dominant; CASQ2 – autosomal recessive). For more information about CPVT, please visit GeneReviews.
Left ventricular noncompaction (LVNC) has recently been established as a specific type of cardiomyopathy and is characterized by a spongy appearance of the left ventricular myocardium, resulting from an arrest in normal cardiac development. LVNC can be found in isolation or in association with other cardiomyopathies (HCM, DCM) as well as congenital cardiac abnormalities. The population prevalence is not known but LVNC is reported in ~0.014% of echocardiograms. LVNC is often familial and the genetic spectrum is beginning to emerge although it is not yet well defined. LVNC genes reported to date include ACTC, DTNA, LDB3, MYBPC3, MYH7, TAZ, and TNNT2 (Montserrat 2007, Klaassen 2008; Kaneda 2007, Zaragoza 2007; reviewed in: Maron 2006, Finsterer 2009). For more information about LVNC, please visit OMIM.org.
For any additional information, please contact us at 617-768-8500 or lmm@partners.org.
| Genes: 51 genesMethodology: A combination of next generation sequencing technology and Sanger sequencingAnalytical Sensitivity:Substitutions: 100% (95%CI=98.5-100)Small InDels: 95% (95%CI=83-99)Clinical Sensitivity: See below.Additional Links: |
Cardiomyopathy
| Price | TAT | CPT Codes | ||||
|---|---|---|---|---|---|---|
| Pan Cardiomyopathy Panel (51 Genes) – lmPCM-pnlAv2_L |
||||||
| $3,950 | 8-12 wks | 81479 | ||||
| HCM Panel (18 Genes) – lmPCM-pnlB_L | ||||||
| $3,200 | 8-12 wks | 81479 | ||||
| DCM Panel (27 Genes) – lmPCM-pnlCv2_L | ||||||
| $3,850 | 8-12 wks | 81479 | ||||
| ARVC/CPVT Panel (8 Genes) – lmPCM-pnlD_L | ||||||
| $3,000 | 8-12 wks | 81479 | ||||
| LVNC Panel (10 Genes) – lmPCM-pnlE_L | ||||||
| $3,200 | 8-12 wks | 81479 | ||||
| Remaining Pan Cardiomyopathy Genes (HCM Reflex) – lmPCM-pnlFv2_L | ||||||
| $2,000 | 8-12 wks | 81479 | ||||
| Remaining Pan Cardiomyopathy Genes (DCM Reflex) – lmPCM-pnlGv2_L | ||||||
| $2,000 | 8-12 wks | 81479 | ||||
| Remaining Pan Cardiomyopathy Genes (ARVC/CPVT Reflex) – lmPCM-pnlHv2_L | ||||||
| $2,000 | 8-12 wks | 81479 | ||||
| Remaining Pan Cardiomyopathy Genes (LVNC Reflex) – lmPCM-pnlIv2_L | ||||||
| $2,000 | 8-12 wks | 81479 | ||||
| Remaining Pan Cardiomyopathy Genes (Version 1 Reflex) – lmPCM-pnlL_L | ||||||
| $750 | 8-12 wks | 81479 | ||||
| Unexplained Cardiac Hypertrophy Panel (2 genes) – lmUCH-pnlA_L | ||||||
| $1,500 | 3 wks | 81479 | ||||
| ABCC9 Gene Sequencing – lmABCC9-a_L | ||||||
| $1,800 | 3 wks | 81479 | ||||
| ACTC Gene Sequencing – lmACTC-a_L | ||||||
| $700 | 3 wks | 81405 | ||||
| ACTN2 Gene Sequencing – lmACTN2-a_L | ||||||
| $1,500 | 3 wks | 81479 | ||||
| CSRP3 Gene Sequencing – lmCSRP3-a_L | ||||||
| $900 | 3 wks | 81479 | ||||
| CTF1 Gene Sequencing – lmCTF1-a_L | ||||||
| $800 | 3 wks | 81479 | ||||
| DES Gene Sequencing – lmDES-a_L | ||||||
| $750 | 3 wks | 81479 | ||||
| DSC2 Gene Sequencing – lmDSC2-a_L | ||||||
| $1,150 | 3 wks | 81479 | ||||
| DSG2 Gene Sequencing – lmDSG2-a_L | ||||||
| $1,075 | 3 wks | 81479 | ||||
| DSP Gene Sequencing – lmDSP-a_L | ||||||
| $1,700 | 3 wks | 81479 | ||||
| DTNA Gene Sequencing – lmDTNA-a_L | ||||||
| $1,500 | 5-6 wks | 81479 | ||||
| EMD Gene Sequencing – lmEMD-a_L | ||||||
| $450 | 3 wks | 81479 | ||||
| GLA Gene Sequencing – lmGLA-a_L | ||||||
| $700 | 3 wks | 81405 | ||||
| LAMP2 Gene Sequencing – lmLAMP2-a_L | ||||||
| $700 | 3 wks | 81405 | ||||
| LDB3 Gene Sequencing – lmLDB3-a_L | ||||||
| $950 | 3 wks | 81406 | ||||
| LMNA Gene Sequencing – lmLMNA-a_L | ||||||
| $700 | 3 wks | 81406 | ||||
| MYBPC3 Gene Sequencing – lmMYBPC3-a_L | ||||||
| $1,500 | 3 wks | 81407 | ||||
| MYH7 Gene Sequencing – lmMYH7-a_L | ||||||
| $1,700 | 3 wks | 81407 | ||||
| MYL2 Gene Sequencing – lmMYL2-a_L | ||||||
| $700 | 3 wks | 81405 | ||||
| MYL3 Gene Sequencing – lmMYL3-a_L | ||||||
| $700 | 3 wks | 81405 | ||||
| PKP2 Gene Sequencing – lmPKP2-a_L | ||||||
| $1,500 | 3 wks | 81479 | ||||
| PLN Gene Sequencing – lmPLN-a_L | ||||||
| $400 | 3 wks | 81479 | ||||
| PRKAG2 Gene Sequencing – lmPRKAG2-a_L | ||||||
| $1,000 | 3 wks | 81406 | ||||
| SCN5A Gene Sequencing – lmSCN5A-a_L | ||||||
| $1,700 | 5-6 wks | 81407 | ||||
| SGCD Gene Sequencing – lmSGCD-a_L | ||||||
| $1,100 | 3 wks | 81405 | ||||
| TAZ Gene Sequencing – lmTAZ-a_L | ||||||
| $700 | 3 wks | 81406 | ||||
| TCAP Gene Sequencing – lmTCAP-a_L | ||||||
| $700 | 3 wks | 81479 | ||||
| TMEM43 Gene Sequencing – lmTMEM43-a_L | ||||||
| $700 | 3 wks | 81479 | ||||
| TNNI3 Gene Sequencing – lmTNNI3-a_L | ||||||
| $700 | 3 wks | 81405 | ||||
| TNNT2 Gene Sequencing – lmTNNT2-a_L | ||||||
| $1,000 | 3 wks | 81406 | ||||
| TPM1 Gene Sequencing – lmTPM1-a_L | ||||||
| $700 | 3 wks | 81405 | ||||
| TTN Gene Sequencing – lmTTN-a_L | ||||||
| $3,000 | 8-12 wks | 81479 | ||||
| TTR Gene Sequencing – lmTTR-a_L | ||||||
| $485 | 3 wks | 81404 | ||||
| VCL Gene Sequencing – lmVCL-a_L | ||||||
| $1,500 | 3 wks | 81479 | ||||
Congenital Heart Disease/Defects
| Price | TAT | CPT Codes | |
|---|---|---|---|
| Congenital Heart Disease Panel A (GATA4, NKX2-5, JAG1) – lmCHD-pnlA_L | |||
| $1,300 | 4 wks | 81479 | |
| ELN (Elastin) Gene Sequencing – lmELN-a_L | |||
| $1,300 | 4 wks | 81479 | |
| GATA4 Gene Sequencing – lmGATA4-a_L | |||
| $750 | 3 wks | 81479 | |
| JAG1 Gene Sequencing – lmJAG1-a_L | |||
| $1,100 | 3 wks | 81407 | |
| NKX2-5 Gene Sequencing – lmNKX2-5-a_L | |||
| $600 | 3 wks | 81479 | |
Lakdawala NK, Funke BH, Baxter S, Cirino A, Roberts AE, Judge DP, Johnson N, Mendelsohn NJ, Morel C, Care M, Chung WK, Jones C, Psychogios A, Duffy E, Rehm HL, White E, Seidman JG, Seidman CE, Ho CY. Genetic Testing for Dilated Cardiomyopathy in Clinical Practice. J Card Fail 2012, In press.
Neri PM, Pollard SE, Volk LA, Newmark L, Varugheese M, Baxter S, Aronson SJ, Rehm HL, Bates DW. Usability of a Novel Clinician Interface for Genetic Results. J Biomed Informatics. 2012. In press.
Genomics @Brigham and Women’s Hospital and Harvard Medical School
The goal of The Cardiovascular Genome Unit (TCGU) is to foster interdisciplinary interaction between clinical investigators and scientists to comprehensively explore the era of human genomic research. In particular, our aim would be to identify, categorize and characterize the genes and genetic pathways of the vascular and cardiac tissues of the cardiovascular system during oncogenesis, normal function and the pathogenesis of cardiovascular diseases.
The Cardiovascular Genome Unit is responsible for indexing gene expression, profiling gene expression, identifying SNPs and generation of protein profiles from a wide variety of tissues representative of various anatomical regions as well as developmental and pathological stages in the cardiovascular system. This information resource emphasizes on cardiovascular disease and should aid in the discovery of disease causing genes, diagnostic and prognostic markers, drug targets, protein therapeutics and improved therapeutic strategies for cardiovascular disease.
Our laboratory is the curator of a genome-based resource for molecular cardiovascular medicine consisting of over 52,000 ESTs generated from nine heart and artery libraries, representing different developmental stages and disease states (Liew et al 1994, Hwang et al 1997, Dempsey et al 2000).
This comprehensive catalogue of cardiac and hematopoietic genes is an unmined molecular resource for microarray analysis and a genetic gold mine for the discovery of genes that may play a role in cardiovascular disorders. In order to exploit this raw data, we propose to develop cDNA microarrays consisting of known and novel sequence-tagged genes. The arrayed clones provide an excellent substrate for expression profiling of cardiovascular disease, for example heart failure or ischemic heart disease, leading the potential discovery of diagnostic as well as prognostic markers.
In order to accomplish the goals of the center, several cutting edge technologies are being employed.
The human cardiovascular research component of our labs.
One of the most efficient and effective strategies for the identification genes is the Expressed Sequence Tag (EST) approach. In this approach, randomly selected cDNA clones are subjected to automated sequencing (PCR or plasmid templates) to generate a partial sequence from either the 5’- or 3’-end termed an EST. This method allows for large-scale gene tagging and indexing from any tissue- or cell-type of interest. A comprehensive cardiovascular gene index could be developed using a variety of cardiovascular tissues representing different anatomical, developmental and pathological states.
Comparing transcript profiles between different development or disease states is a powerful way to gain insight into the genetic changes underlying these events. This is especially important when looking at complex systems, such as in development or disease (e.g. hypertension or atherosclerosis). There are several unique approaches to this problem, several of which are:
a) EST profile Comparison– After the production of a significant number of ESTs from 2 or more libraries, the frequencies of ESTs can be compared to identify those genes which are differentially expressed. However, normalized or subtracted cDNA libraries cannot be used for this and this method is most effective for finding large differences in expression.
b) cDNA Microarray Hybridization– The recent introduction of the cDNA microarray, a technology capable of analyzing the expression of thousands of genes simultaneously in a single experiment, may provide one of the best ways to delineate gene expression patterns. In the cDNA microarray, cDNA clones are spotted onto a glass slide matrix and hybridized with fluorescently labeled cDNA probes derived from total RNA pools of test and reference cells or tissues. The signal intensity for each probe is quantified and any differences between the two samples becomes readily apparent. Thus, the genetic changes underlying the phenotype of study can be identified at the level of a single gene.
c) Identification of Single Nucleotide Polymorphisms– SNPs are single-base heritable variations in the genome which occur once in approximately 1000 bases in the human genome and occur at a frequency of >1% in the human population. SNPs provide an important genetic resource useful for disease gene discovery. including the identification of disease susceptible genes. SNPs can be identified through comparison of EST sequences, DNA hybridization strategies and direct sequencing of genomic DNA. The generation of a SNP database for genes expressed in the cardiovascular system will provide a valuable resource to aid in disease gene discovery.
d) Quantitative determination of expressed genes– the up- and down- regulated genes are crucial to the phenotypic expression of any given cell. The frequency of gene expressed in development or disease state can be obtained from an EST approach using cDNA libraries as well as its intensity detected using microarrays. Such results can be verified through RT-PCR analysis from the tissue samples. A high through-put analysis of 96 samples can be performed by real-time PCR analyses.
Using our 10,000 element “CardioChip”, we elucidated over 100 differentially expressed genes in end-stage heart failure resulting from dilated cardiomyopathy. The results were published in
Am J Pathol. 2002 June; 160(6): 2035–2043.
Global Gene Expression Profiling of End-Stage Dilated Cardiomyopathy Using a Human Cardiovascular-Based cDNA Microarray
Abstract
To obtain a genomic portrait of heart failure derived from end-stage dilated cardiomyopathy (DCM), we explored expression analysis using the CardioChip, a nonredundant 10,848-element human cardiovascular-based expressed sequence tag glass slide cDNA microarray constructed in-house. RNA was extracted from the left ventricular free wall of seven patients undergoing transplantation, and five nonfailing heart samples. Cy3- and Cy5-labeled (and reverse dye-labeled) cDNA probes were synthesized from individual diseased or nonfailing adult heart RNA, and hybridized to the array. More than 100 transcripts were consistently differentially expressed in DCM >1.5-fold (versus pooled nonfailing heart,P < 0.05). Atrial natriuretic peptide was found to be up-regulated in DCM (19-fold compared to nonfailing, P < 0.05), as well as numerous sarcomeric and cytoskeletal proteins (eg, cardiac troponin, tropomyosin), stress response proteins (eg, HSP 40, HSP 70), and transcription/translation regulators (eg, CCAAT box binding factor, eIF-1AY). Down-regulation was most prominently observed with cell-signaling channels and mediators, particularly those involved in Ca2+ pathways (Ca2+/calmodulin-dependent kinase, inositol 1,4,5-trisphosphate receptor, SERCA). Most intriguing was the co-expression of several novel, cardiac-enriched expressed sequence tags. Quantitative real-time reverse transcriptase-polymerase chain reaction of a selection of these clones verified expression. Our study provides a preliminary molecular profile of DCM using the largest human heart-specific cDNA microarray to date.
Dilated cardiomyopathy (DCM) is characterized clinically by left ventricular dilatation, wall thinning, and homogeneous dysfunction of the myocardium leading to congestive heart failure. Genetically, DCM seems to evolve through primary mutations in the genes of the sarcomeric proteins. 1 However, recent evidence suggests that, despite distinct pathways leading to divergent endpoint phenotypes of each disease, there may exist some overlapping genetic modifiers leading to a conversion of one to the other. 2 How this occurs is under question; to understand this, a better knowledge of the molecular pathways and intermediary regulators is required.
Global analysis of gene expression has proven to be a fruitful means of examining the overall molecular portrait of a particular event as well as seeking out novel candidate transcripts that may play a role in formulating the phenotype or genotype of interest. By using this strategy, multiple genes and pathways in complex disorders can be visualized simultaneously, allowing for a feasible platform from which to investigate new and interesting genes. Using expressed sequence tag technology, our laboratory has generated a compendium of genes expressed in the human cardiovascular system, with the ultimate goal of assembling the intricacies of development and of disease, particularly the pathways leading to heart failure. 3 Through a computer-based in silico strategy, we have been able to identify—in a large scale—both known and previously unsuspected genetic modulators contributing to the growth of the myocardium from fetal through adult, and from normal to a perturbed hypertrophic phenotype. In contrast a gene-by-gene approach in elucidating the genes and mechanisms involved is time-consuming and cumbersome.
Recently, microarray technology has been used as a means of large-scale screening of vast numbers of genes—if not whole genomes—that possess differential expression in two distinct conditions. Although new and exciting developments have arisen in such fields as cancer 4 and yeast, 5 advances in understanding the complexity of cardiovascular disease, 6 specifically DCM, have been limited. One recent study examined gene expression in two failing hearts using oligo-based arrays. 7 Although the GeneChip® (Affymetrix, Santa Clara, CA) offers a carefully controlled systematic method of analysis, its current lack of user flexibility in its design hinders novel gene discovery currently available in tissue-specific arrays. Our laboratory has taken advantage of our vast previously acquired resources and has constructed what we believe to be the first ever custom-made cardiovascular-based cDNA microarray, which we term the “CardioChip.” 8 Its practicality and flexibility has allowed us to conceptualize the molecular events surrounding end-stage heart failure.
This report describes the most informative cDNA microarray-based analysis of end-stage heart failure derived from DCM currently available. Although we believe we have effectively demonstrated reproducibility and reliability of our technology (both for the entire array and for a selection of genes located on it), a larger n from our population would enhance the validity of our conclusions. Certainly, there exists no homogeneous heart failure genotype, especially among only seven DCM patients. Nonetheless, we have demonstrated a common expression pattern among our set of samples, from both microarray and QRT-PCR analysis. We are also limited by the genes (both in number and identity) present on this array. Although we are currently unable to spot every gene and gene cluster on our CardioChip, we have tried to draw from a diverse assortment of genes and gene pathways, both known and unknown. It must be emphasized that this investigation is not exhaustive; by no means does it attempt to fully characterize the molecular basis of heart failure. Its intention is to provide a preliminary portrait of global gene expression in complex cardiovascular disease using cDNA microarray and QRT-PCR technology, and to highlight the effectiveness of our ever-evolving platform for gene discovery. With even more patient samples and a CardioChip toward completeness, we will be in a better position to reap the important benefits from this initial work and expand our body of knowledge.
http://www.pnas.org/content/91/22/10645.full.pdf
Liew CC, Jackowski, G, Ma T, Jung, YC, Sole, MJ. Possible role of nonhistone chromatin proteins associated with heterogeneous nuclear RNA in myocardial differentiation and in the genesis of cardiomyopathy. In: Alpert NR, editor. Perspectives in
Cardiovascular Research. Raven Press; 1983. p. 497-511.
Liew CC, Takihara KY, Jandreski M, Liew J, Sole MJ. Structure and expression of human b-myosin heavy chain gene. In: Carraro U, editor. Sarcomeric and Non-sarcomeric Muscles: Basic and Applied Research Prospects for the 90s. Padova, Italy: Unipress
Padova; 1988. P 11-17.
Liew CC, Takihara KY, Liew J, Sole MJ. Characterization of human cardiac myosin heavy chain genes. In: Wu F, Wu CW, editors. Structure and Function of Nucleic Acids and Proteins, New York, Raven Press; 1990. pp.303-309.
Wang RX, Cukerman E, Chen B, Liew CC. Differential screening and megasequencing of human heart cDNA library: A search for genes associated with heart failure. In: Dhalla NS, Pierce GN, Panagia V, Beamish RE, editors. Boston: Kluwer Academic Press; 1995. P. 67-77.
Dempsey A, Liew CC. Genes involved in normal cardiac development. In: Sheridan DJ, editor. Left Ventricular Hypertrophy. London: Churchill Communications Europe Ltd; 1998: p. 61-70.
Tan K, Dempsey A, Liew CC. Cardiac genes and gene databases for cardiovascular disease genetics. In: Hollenberg NK, editor. Current Hypertension Reports. Philadelphia: Current Science Group; 1999: Vol 1:51-58.
Liew, CC. Expressed Sequence Tags. In: Encyclopedia of Molecular Medicine, Ed: T. Creighton, John Wiley and Son, New York. 2001
Hwang J-J, Dzau V and Liew CC. Genomics and thePathophysiology of Heart Failure. In: Current Cardiology Reports; Current Science Inc; 2001: Vol 3: 198-207.