Human embryonic pluripotent stem cells and healing post-myocardial infarction
Curator: Larry H. Bernstein, MD, FCAP
I present a followup based on several recent posts related to the promise of using induced human pluripotent stem cells for repair of ischemia damaged myocardium postinfarct and related effect of heart failure (HF). There has been a change in the concept of cardiovascular risk related to the emergent knowledge of the biology underlying oxidative stress. The more recent discovery of the relationship between ongoing inflammation and clinical outcomes has led to a variety of blood-based assays which may impart additional knowledge about an individual’s propensity for future cardiovascular events (1). Vascular injury and repair are significantly mediated by circulating endothelial progenitor cells (1). Circulating progenitor endothelial cells are defined by co-expression of the markers CD34, CD309 (VEGFR-2/KDR) which are measured by pre-enrichment flow cytometry with specific identification of cell markers (CD34, CD133) and endothelial cell antigens (KDR/VEGFR-2, CD31) (2), used in the assessment of various diseases and physiological states. Improvements in flow cytometry include the Attune® cytometer, which enables the collection of more than 4,000,000 live white blood cell (WBC) events in just 35 minutes (3). Using these methods of analyses, it became evident that circulating endothelial progenitor cells have angiogenic potential.
Activators and inhibitors have been tested for their ability to modulate angiogenesis in early phase clinical trials, and in the case of anti-Flk1 antibodies clinical utility has been demonstrated for anti-tumor strategies (4). Extending this concept further, we pose that just as the progenitor role invoked for angiogenesis, transcriptional networks and interactions are involved in the morphogenesis of the developing vertebrate heart. The identities of crucial regulators involved in defined events in cardio-genesis are being uncovered at a rapid rate. Tissue development and regeneration involve tightly coordinated and integrated processes: selective proliferation of resident stem and precursor cells, differentiation into target somatic cell type, and spatial morphological organization. (4, 5, 6). However, our ability to cross the divide between knowledge and change has not been easy, as reported by Aviva Lev-Ari (7). In a two-day-old mouse, a heart attack causes active stem cells to grow new heart cells; a few months later, the heart is mostly repaired. But in an adult mouse, recovery from such an attack leads to classic after-effects: scar tissue, permanent loss of function and life-threatening arrhythmias (7, 8).
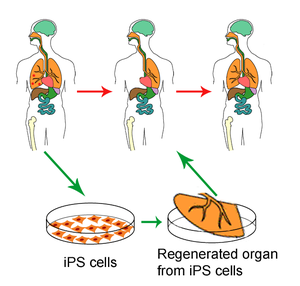
Myocardial cell replacement therapies are hampered by a paucity of sources for human cardiomyocytes and by the expected immune rejection of allogeneic cell grafts. The success using dermal fibroblasts from HF patients reprogrammed by retroviral delivery of Oct4, Sox2, and Klf4 or by using an excisable polycistronic lentiviral vector resulted in HF-hiPSCs induced to differentiate into cardiomyocytes (HF-hiPSC-CMs)(9). Multi-electrode array recordings revealed adequate responses to stimulation. Further study with in vivo transplantation in the rat heart revealed the ability of the HF-hiPSC-CMs to engraft, survive, and structurally integrate with host cardiomyocytes and within 48 hours the tissues were beating together. Human-induced pluripotent stem cells thus can be established from patients with advanced heart failure and coaxed to differentiate into cardiomyocytes, which can integrate with host cardiac tissue (10). The success of the approach rests on modifying the myocardial electro-physiological substrate using cell grafts genetically engineered to express specific ionic channels (11). The expressed potassium channels alter the local myocardial electrophysiological properties by reducing cardiac automaticity and prolonging refractoriness. The key feature involves reprogramming a patient’s own skin cells by delivering three genes followed by a small molecule called valproic acid to the cell nucleus (12).
An alternative approach avoiding the caveats of limited graft survival, is to stimulate a resident source, restricted homing to the site of injury and host immune rejection (13). Thymosin β4 restores vascular potential to adult epicardial-derived progenitor cells with injury. Specifically, it activates adult progenitors to re-express a key embryonic epicardial gene, Wilm’s tumour 1 (Wt1). It was inferred that embryonic reprogramming would mobilize this cell population and differentiation would give rise to de novo cardiomyocytes. Delivery of Tβ4, in conjunction with GMT (an acronym for three genes that normally guide embryonic heart development), into the damaged region resulted in reduction of scar area and improvement in cardiac function compared to GMT or Tβ4 alone. Thymosin-beta4 facilitates cardiac repair after infarction by promoting cell migration and myocyte survival. Additionally, the tetra peptide Ac-SDKP was reported to reduce left ventricular fibrosis in hypertensive rats, reverse fibrosis and inflammation in rats with MI, and stimulate both in vitro and in vivo angiogenesis. Effects of Ac-SDKP, such as the enhancement of angiogenesis and the decrease in inflammation and collagenase activity, are similar to those described for thymosin-beta4. However, there are conflicting reports (14-18).
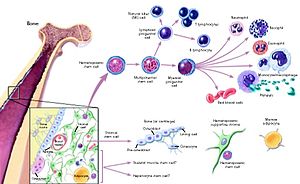
There are other studies that show promise. There has been the first infusion of stem cells into the coronary artery (19). This result was at least as effective as intramyocardial injection in limiting LV remodeling and improving both regional and global LV function. The intracoronary route appears to be superior in terms of uniformity of cell distribution, myocyte regeneration, and amount of viable tissue in the risk region. Another finds that down regulation of leukocyte HIF-1? Expression resulted in decreased recruitment of WBC to the sites of inflammation and improvement in cardiac function following MI (20). Irradiated 6-to 8-week-old C57/BL6J mice received 50 000 GFP HIF-1? or scramble siRNA transfected hematopoietic stem cells. Down regulation of HIF-1? suppressed WBC cytokine receptors CCR1,-2, and-4, which are necessary for WBC mobilization and recruitment to inflammatory cytokines following MI. There also have been cited limitations to success in older patients (21). The findings suggest that coronary artery disease and cardiac remodeling in chronic ischemia has a significant negative correlation between the age of the patient and the number of migrated ckit-positive cells.
Lymphocytes infiltrate and react with ischemia damaged heart tissue, which can impair proper tissue healing. In a study with isoproterenol induced myocardial necrosis TNF-α, IFN-γ and CCL-5, but not FOXP3 + expression, was increased in draining lymph nodes, indicating that the observed lymphocyte population that proliferated in response to cardiac components presented a pro-inflammatory and pro-fibrotic profile. The group was rendered tolerant by myocardial gavage and expressed cardiac FOXP3 + earlier than did the control group, and showed a milder inflammatory infiltrate, lower MMP-9 expression, less collagen deposition, and improved cardiac performance when compared to animals that received only isoproterenol administration (22). Patients with acute myocardial infarction show high circulating levels of neuropeptide Substance P (SP) and NK1-positive cells that co express Progenitor Cell (PC) antigen, such as CD34, KDR, and CXCR4. Moreover, NK1-expressing PC is abundant in infarcted hearts, highlighting the role of SP in reparative neovascularization (23). Do CD4 + T cells become activated and influence wound healing after experimental MI? To study the role of CD4 + T cells in wound healing and remodeling, CD4 + T-cell- deficient mice (CD4 knockout [KO], MHCII) and T-cell receptor-transgenic OT-II. Within the infarcted myocardium, CD4 KO mice displayed higher total numbers of leukocytes and proinflammatory monocytes (18.3±3.0 104/mg WT versus 75.7±17.0 10 4/mg CD4 KO, P<0.05), and MHCII and OT-II mice displayed significantly greater mortality. Collagen matrix formation in the infarct zone was severely disturbed in CD4 KO and MHCII mice, as well as in OT-II mice (24).
Thus, it appears that CD4T cells become activated after MI and facilitate wound healing of the myocardium. Inflammation and immune responses are integral components in he healing process after myocardial infarction. Importantly, dendritic cell (DC) infiltration occurs in the infarcted heart. In concert with the previous two studies, DC-ablated infarcts had enhanced monocyte/ macrophage recruitment. Among these cells, marked infiltration of proinflammatory Ly6C high monocytes and F4/80 + CD206 – M1 macrophages and, conversely, impaired recruitment of anti-inflammatory Ly6C low monocytes and F4/80 + CD206 + M2 macrophages in the infarcted myocardium were identified in the DC-ablated group compared with the control group (25). Thus, the DC is a potent immunoprotective regulator during the post-infarction healing process via its control of monocyte/macrophage homeostasis. Despite the recent successes, there are a number of interlocking and possibly explanatory processes to control in the mix.
What about medical therapies? Here too there is a factor in engaging eNOS or iNOS activity as detailed in the presentation by Aviva Lev-Ari (26). 60–70% of major cardiovascular events cannot be prevented with current approaches focused on LDL, such as statin therapy, and low HDL levels are particularly common in males with early-onset atherosclerosis. She makes the point that there is compelling evidence that HDL is not solely a marker of lower risk of cardiovascular disease but instead is a mediator of vascular health.
Aviva Lev-Ari examines the phytoestrogen, Genistein, and other drugs. Genistein acutely stimulates Nitric Oxide synthesis in vascular Endothelial cells by a cyclic adenosine 5′-monophosphate-dependent mechanism (Liu et al., 2004). The intracellular signaling pathways for activation of eNOS by genistein were shown independent of PI3K/Akt or ERK/MAPK but depended on the cAMP/PKA cascade. In addition, the genistein action on eNOS was not inhibited by an ER antagonist and was unrelated to tyrosine kinase inhibition. Furthermore, genistein has antiatherogenic effects and inhibits proliferation of vascular endothelial and smooth muscle cells, and in vitro studies suggest a protective role of genistein in the vasculature. In Liu et al., (2004) study, genistein acted directly on BAECs and HUVECs to activate eNOS and NO production through nongenomic mechanisms in whole vascular endothelial cells. In addition, 5-hydroxytryptamine evokes endothelial nitric oxide synthase activation. In this example, eNOS co-localizes with PECAM-1, but not with VE-cadherin and plakoglobin at the intercellular junctions of the endothelium.
Finally, activation of endothelial nitric oxide synthase (eNOS) resulted in the production of nitric oxide (NO) that mediates the vasorelaxing properties of endothelial cells. The responses were effectively blocked by a 5-HT1B receptor antagonist, a 5-HT1B/5-HT2 receptor antagonist, and eNOS selective antagonists, L-Nomega -monomethyl-L-arginine (L-NMMA) and L-N omega-iminoethyl-L-ornithine (L-NIO). This lends credence to a 5-HT1B receptor/eNOS pathway, accounting in part for the activation of eNOS by 5-HT. Finally, a third-generation ß-blocker augments vascular Nitric Oxide release. Nebivolol increases vascular NO productionby causing a rise in endothelial free [Ca2+]i and endothelial NO synthase–dependent NO production. It is a ß1-selective adrenergic receptor antagonist with proposed nitric oxide (NO)–mediated vasodilating properties. Nevertheless, it appears that not nebivolol, but its metabolites augment NO production (Broeders et al., 2000). These findings reveal new insights into interaction with eNOS in vascular therapy: [1] new indications for TDZs as stimulators of eNOS; [2] new indications for beta blockers as NO stimulant. Nebivolol is a vasodilator, thus functions as an antihypertensive.
References:
1. Saha S. Innovations in Bio-instrumentation for Measurement of Circulating Progenitor Endothelial Cells in Human Blood. Pharma Intell. July 8, 2012. http://pharmaceuticalintelligence.com/2012/07/08/innovations-in-bio-instrumentation-for-measurement-of-circulating-progenitor-endothelial-cells-in-human-blood/
(http://www.ncbi.nlm.nih.gov/pubmed/19124422)
2. Ibid (http://www.ncbi.nlm.nih.gov/pubmed/20381496).
3. Ibid (http://zh.invitrogen.com/etc/medialib/files/Cell-Analysis/PDFs.Par.54318.File.tmp/CO24210-Human-CEC_cancer.pdf)
4. Saha S. Endothelial Differentiation and Morphogenesis of Cardiac Precursors. Pharma Intelligence. July 17, 2012.
5. Ibid (http://circres.ahajournals.org/content/90/5/509.full).
6. Ibid (http://www.ncbi.nlm.nih.gov/pubmed/22669846).
7. Aviva-Lev-Ari. Stem cells create new heart cells in baby mice, but not in adults, study shows.Aug 3, 2012. Pharma Intelligence.
8. Krishna Ramanujan http://www.news.cornell.edu/stories/July12/HeartStemCells.html
9. Saha S. Human Embryonic-Derived Cardiac Progenitor Cells for Myocardial Repair. Pharma Intelligence. Aug 1, 2012.
10. Zwi-Dantsis L, Huber I, Habib M, Winterstern A, (..), Gepstein L. Derivation and cardiomyocyte differentiation of induced pluripotent stem cells from heart failure patients. Eur Heart J. 2012 May 22. [Epub ahead of print] (VBL RX, Inc. Tel Aviv, http://www.vblrx.com).
11. Yankelson L, Feld Y, Bressler-Stramer T, Itzhaki I,(..), Gepstein L. Cell therapy for modification of the myocardial electrophysiological substrate. Circulation. 2008 Feb 12; 117(6):720-31. Epub 2008 Jan 22.
12. Huber I, Itzhaki I, Caspi O, Arbel G, (..), Gepstein L. Identification and selection of cardiomyocytes during human embryonic stem cell differentiation. FASEB J. 2007 Aug; 21(10):2551-63. Epub 2007 Apr 13.
13. Aviva Lev-Ari. Resident-cell-based Therapy in Human Ischaemic Heart Disease: Evolution in the PROMISE of Thymosin beta4 for Cardiac Repair. Pharma Intelligence. April 30, 2012.
14. Ibid. Shrivastava S, Srivastava D, Olson EN, DiMaio JM, Bock-Marquette I.
Ann N Y Acad Sci. 2010 Apr; 1194:87-96.
15. Ibid. Smart N, Risebro CA, Clark JE, Ehler E, (..), Riley PR, Thymosin beta4 facilitates epicardial neovascularization of the injured adult heart. Ann N Y Acad Sci. 2010 Apr;1194:97-104
16. Ibid. Smart N, Bollini S, Dubé KN, Vieira JM, (..) Riley PR. Nature. 2011 Jun 8; 474(7353):640-4
17. Ibid. Zhou B, Honor LB, Ma Q, Oh JH, (..), Pu WT. Thymosin beta 4 treatment after myocardial infarction does not reprogram epicardial cells into cardiomyocytes. J Mol Cell Cardiol. 2012 Jan; 52(1):43-7. Epub 2011 Aug 26.
18. Ibid. Scientists Report that Process of Converting Non-Beating Heart Cells into Functional, Beating Heart Cells is Enhanced Using Thymosin Beta 4 in Conjunction with Gene Therapy. Regenerx Biopharmaceuticals, Inc. Nature. Apr. 18, 2012
19. Li, Q., Guo, Y., Ou, Q., Chen, N., (…), Bolli, R. Intracoronary administration of cardiac stem cells in mice: A new, improved technique for cell therapy in murine models. Basic Research in Cardiology 2011; 106 (5), pp. 849-864.
20. Dong, F., Khalil, M.,Kiedrowski, M.,O’Connor, C.,(..) ,Penn, M.S. Critical role for leukocyte hypoxia inducible factor-1α expression in post-myocardial infarction left ventricular remodeling. Circulation Research 2010; 106 (3) , pp. 601-610
21. Aghila Rani, K.G.,Jayakumar, K.,Sarma, P.S., Kartha, C.C. Clinical determinants of ckit-positive cardiac cell yield in coronary disease. Asian Cardiovascular and Thoracic Annals 2009; 17 (2), pp. 139-142.
22. Ramos, G.C., Dalbó, S., Leite, D.P.,Goldfeder, E. ,(..), Assreuy, J. The autoimmune nature of post-infarct myocardial healing: Oral tolerance to cardiac antigens as a novel strategy to improve cardiac healing. Autoimmunity 2012; 45 (3), pp. 233-244.
23. Amadesi, S., Reni, C., Katare, R., Meloni, M., (…),Madeddu, P. Role for substance P-based nociceptive signaling in progenitor cell activation and angiogenesis during ischemia in mice and in human subjects. Circulation 2012; 125 (14) , pp. 1774-1786.
24. Hofmann, U.,Beyersdorf, N.,Weirather, J.,Podolskaya, A.(..), Frantz, S. Activation of CD4 + T lymphocytes improves wound healing and survival after experimental myocardial infarction in mice. Circulation 2012; 125 (13) , pp. 1652-1663.
25. Anzai, A., Anzai, T.,Nagai, S., Maekawa, Y., (…), Fukuda, K. Regulatory role of dendritic cells in postinfarction healing and left ventricular remodeling. Circulation 2012; 125 (10), pp. 1234-1245
26. Lev-Ari A. Cardiovascular Disease (CVD) and the Role of agent alternatives in endothelial Nitric Oxide Synthase (eNOS) Activation and Nitric Oxide Production. Pharma Intelligence. July 19, 2012.
27. Ibid. Li AC, Binder CJ, Gutierrez A, Brown KK, (..), Glass CK. Differential inhibition of macrophage foam-cell formation and atherosclerosis in mice by PPAR-alpha, Beta/delta, and gamma. J Clin Invest 2004; 114:1564-1576.
28. Ibid. Broeders MAW, Doevendans PA, Bekkers BCAM, (…), van der Zee R. Nebivolol: A Third-Generation ß-Blocker That Augments Vascular Nitric Oxide Release, Endothelial ß2-Adrenergic Receptor–Mediated Nitric Oxide Production. Circulation 2000; 102:677.
Read Full Post »