Cancer and Nutrition
Writer and Curator: Larry H. Bernstein, MD, FCAP
The following discussions have been a topic of great interest and much controversy. In this discussion I shall not cover the topics related to Alternative and Complementary Medicine that is discussed elsewhere. However, there is significant reason to explore the relationships of vitamin and micronutrient insufficiencies to cancer. The following nutritional subjects will be the focus of these discussions.
- Transthyretin (TTR)
- Vitamin A (retinoids and retinol) and retinol-binding protein (RBP)
- Vitamin C
- Vitamin D
- Magnesium (Mg++)
Cancer, homocysteine, Alzheimer’s Disease, and cardiovascular disease
1 Transthyretin
1.1 Plasma Transthyretin Indicates the Direction of both Nitrogen Balance and Retinoid Status in Health and Disease
Ingenbleek Yves1 and Bienvenu Jacques2,3,*
1Laboratory of Nutrition, Faculty of Pharmacy, University Louis Pasteur Strasbourg 1, France; 2Laboratory of Immunology, Hospices Civils de Lyon and 3INSERM U 851, University Claude Bernard Lyon 1, France
The Open Clinical Chemistry Journal, 2008; 1:1-12
Abstract: Whatever the nutritional status and the disease condition, the actual transthyretin (TTR) plasma level is determined by opposing influences between anabolic and catabolic alterations. Rising TTR values indicate that synthetic processes prevail over tissue breakdown with a nitrogen balance (NB) turning positive as a result of efficient nutritional support and / or anti-inflammatory therapy. Declining TTR values point to the failure of sustaining NB as an effect of maladjusted dietetic management and / or further worsening of the morbid condition. Serial measurement of TTR thus appears as a dynamic index defining the direction of NB in acute and chronic disorders, serving as a guide to alert the physician on the validity of his therapeutic strategy. The level of TTR production by the liver also works as a limiting factor for the cellular bioavailability of retinol and retinoid derivatives which play major roles in the brain ageing process. Optimal protein nutritional status, as assessed by TTR values within the normal range, prevents the occurrence of vascular and cerebral damages while maintaining the retinoid-mediated memory, cognitive and behavioral activities of elderly persons.
INTRODUCTION Measurement of transthyretin (TTR, formerly called prealbumin) was proposed as nutritional marker in The Lancet in 1972 [1]. This proposal was largely disregarded by the scientific community during the decade following its publication. TTR testing is now the most utilized nutritional marker worldwide, having received the strong support of the Prealbumin Consensus Group [2]. A minority of workers, however, remain doubtful [3] or even reluctant [4] to adopt TTR as nutritional index, stressing the point that its synthesis is also influenced by inflammatory conditions [3,4] and by other extra-nutritional factors such as natural or synthetic corticosteroids [5] and androgens [6]. The aim of the present review paper is to clarify the complex relationships linking malnutrition and inflammation, throwing further insight into a nutrition domain of increasing public health.
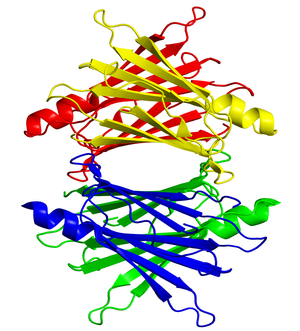
EVOLUTION, STRUCTURE AND FUNCTIONS TTR is a highly conserved protein in vertebrate species already secreted by the choroid plexus of reptiles 300 millions years ago and remaining confined within the cerebrospinal fluid (CSF) [10]. Synthesis and secretion of TTR by the liver evolved much later, about 100 millions years ago, in birds and eutherian mammals [11]. Production of TTR by the liver and by the choroid plexus is regulated separately [12]. The human TTR gene has been localized on the long arm of the chromosome 18q23 [13]. The nucleotide sequences of the entire TTR gene, including the 5′ (transcription initiating site) and the 3′ (untranslated site) flanking regions have been described [14,15]. The gene spans 6.9 kilobases (kb) and consists of 4 exons and 3 introns [14,15]. The hepatic TTR mRNA measures 0.7 kb encoding a pro-TTR-monomer undergoing a cleaving process to release the native TTR monomer [16]. Four identical subunits each 127 amino acids (AAs) length coalesce noncovalently to generate the fully mature nonglycosylated molecule whose molecular mass (MM) reaches 55 kDa [17]. Two binding sites for thyroid hormones are buried inside the central channel of the TTR heterodimer [18]. The secondary, tertiary and quarternary conformation structures of the TTR protein have been reported using 1.8 Å Fourier analysis [18]. One TTR monomer binds to a small companion protein (21 kDa MM) to which a single retinol is bound (all-trans-retinol), hence its RBP denomination [19]. X-ray crystallographic studies have shown that RBP possesses an eight-stranded -barrel core that completely encapsulates the retinol molecule [20]. Under usual circumstances, RBP is almost entirely saturated with retinol, explaining that the 3 components of the retinol circulating complex (RCC) of 76 kDa MM has a close 1:1:1 stoichiometry [21]. Aggregation of TTR to holo-RBP occurs within the endoplasmic reticulum prior to extracellular RCC secretion [22]. The TTR protein was first discovered in human CSF in 1942 [23] and soon after in human serum. Human TTR transports about 20% of the intravascular pool of both thyroid hormones (Thyroxine [T4], triiodothyronine [T3]) and at least 90-95% of the retinol circulating pool. The term transthyretin was recommended by the International Nomenclature Committee [26] stressing the dual conveying role played by TTR in all eutherians.
The biological half-life of TTR is approximately 2 days [27] whereas that of holo-RBP (RBP + bound retinol) is half a day [28]. By contrast, apo-RBP devoid of its retinol ligand displays a significantly reduced half-life of 3.5 hr [28] and undergoes rapid glomerular leakage with subsequent tubular disintegration and recycling of its AA residues. It is therefore assumed that TTR plays an important role in the safeguard of the retinol pool. The catabolic site of TTR is mainly the liver, followed by muscle mass, skin and kidneys [29]. The TTR molecule displays microheterogeneity [30] and tissue deposits occur throughout the normal ageing processes [31]. In contrast, TTR is characterized by a very large genetic polymorphism affecting about 100 different point mutations [32], leading to misfolded forms of the protein and occurrence of amyloid disorders in several organs. The tetrameric TTR protein is recognized as a component of the normal pancreatic cell structure, preserving its integrity against the risk of apoptosis [40]. Finally, normal TTR production is required for the maturation of brain neural stem cells [41] and for the control of spatial reference memory performances [42].
SIGNIFICANCE OF TTR THROUGHOUT THE HUMAN LIFESPAN Significant alterations in the levels of protein intakes by humans affect protein synthesis, turnover and breakdown and determine the outcome of total body N (TBN). Anabolism occurs when the rate of AA incorporation into protein exceeds that of oxidative losses, yielding a positive NB. Catabolism is the result of protein breakdown prevailing over protein synthesis [43]. Increasing gestational age is accompanied by a slow and predictable rise in TTR values correlated with birth weight and proved useful in distinguishing between small, appropriate and large for gestational age infants [47,48]. Starting from birth until 100 years of age, our reference TTR values [54] are those collected in the monograph ” Serum Proteins in Clinical Medicine ” edited by the Foundation for Blood Research. The plasma TTR concentrations in healthy neonates are approximately two thirds those measured in healthy mothers and thereafter increase slowly until the onset of puberty without displaying sexual differences. The rate of protein synthesis similarly increases linearly during the prepubertal period [55], consistent with superimposable N accretion rates [56]. Human puberty is characterized by major hormonal and metabolic alterations leading to increased height velocity and weight gain [60]. The onset of puberty requires close interrelationships between the effects triggered by growth hormone and insulin-like growth factors, by thyroid and steroid hormones, by insulin and sex hormones [60]. Whereas androgens strongly promote the development of muscle mass in males and lipolytic effects on visceral and subcutaneous fat, estrogens have minimal effect on the female musculature while stimulating the accrual of subcutaneous fat depots [60]. Body composition studies indicate prepubertal redistribution of FM and FFM with a significantly higher S-shaped elevation of FFM in male adolescents compared with the blunted curve recorded in teenaged girls [61,62]. TTR values manifest closely paralleled sex- and age-peculiarities in process of time that are best explained by the deeper androgenic impregnation of male subjects [6,43]. The musculature is by weight the main component of FFM, representing 37% of body mass [61]. In healthy adults, the sex-related difference in plasma TTR-RBP concentrations is maintained at plateau levels after sexual maturity [54,63]. Normal TTR plasma values are stabilized around 290-320 mg/L in males and around 250-280 mg/L in females [54,63]. Starting from the sixties, TTR concentrations progressively decline over time, disclosing a steeper slope in elderly men that reflects a relatively more rapid deterioration of their muscle mass [43]. As a result, the earlier TTR sexual difference disappears by about the age of 70 years [43]. This correlates with the age-dependent curvilinear drop of TBN, characterized by an accelerated decrease after 65 years [64]. Taken together, the plasma TTR evolutionary patterns reveal a parallelism with FFM so that TTR serves as an indicator of muscle mass. The data show that age and gender are significant co-variates of TTR which require separate blood reference values [54].
TTR AS INDEX OF PROTEIN DEPLETION / REPLETION STATES There exists a long-lasting debate aimed at identifying the most effective protein sources, level of energy-yielding substrates and the proportion among these for the support of protein metabolism. Under usual conditions, glucose functions as the major energy substrate for protein synthesis. If the carbohydrate energy is lacking, glucose must be synthesized by gluconeogenesis, mainly from the conversion of endogenous or dietary protein [65]. This corresponds to a form of nutritional wastage which augments the cost of protein synthesis, as documented by an increased urinary excretion of urea. The above metabolic pattern stands in broad conformity with the concept that ” protein synthesis occurs in the flame of sugars ” [66].
FAO/WHO/UNU recommends for healthy adults the safe level of 0.75 g k-1 day-1 protein intake [67]. Although this amount of protein sustains normal growth and keeps unmodified the concentration of most biological parameters, such intake appears to be marginally inadequate to maintain the metabolic reserve capacities that are required to mount optimal responses to stress [68]. Studies have disclosed that TTR plasma level and pool size remain unaltered because its synthetic and catabolic rates are both downregulated concomitantly [69]. Changes occurring during prolonged starvation causes the N balance to turn negative despite efforts to minimize protein catabolism [70]. There is a direct correlation between the rate of liver protein synthesis and intrahepatic concentrations of individual free AAs [71]. It is likely that the dietary limitation of some AAs such as tryptophan [72] or leucine [73] could specifically exert inhibitory effects on the transcriptional [74] or translational [75] regulation of protein synthesis. Consequently, protein depletion causes a decrease in TTR mRNA [72,74,76].
Transcription of the TTR gene in the liver is directed by CCAAT/enhancer binding protein (C/EBP) bound to nuclear factor 1 (NF1) [74]. Multiple hepatocyte nuclear factors (HNFs) function in the regulation of TTR gene expression [77]. It has been recently shown that one of them (HNF-4) plays prominent roles before and after injury [78]. The drop of liver TTR mRNA levels to about half as an effect of protein deprivation [74] is accompanied by a corresponding diminished secretion of mature TTR molecules in the bloodstream.
The rapidly turning over TTR protein is exquisitely sensitive to any change in protein and/or energy supply, being clearly situated on the cutting edge of the equipoise. This is documented in preterm infants in whom AA supply is responsible for maintaining normal protein synthesis which may be somewhat modulated by fluctuations in energy intake [79]. In the declared stage of protein malnutrition, the serial measurement of TTR may serve to grade the severity of the disease spectrum, from mild [90] to severe [1] forms. Both metabolic and structural N compartments undergo exhausting processes as documented by the fall of nitrogenous compounds in the urine of protein-depleted subjects [91]. The relative dominance of urea over ammonia catabolites [92] reflects the more intense turnover rate of tissues belonging to the readily mobilizable N pool. Decreased TTR plasma values are indeed correlated with the involution of the gut mucosa [93] and with the extent of liver dysfunction, more pronounced in the kwashiorkor disease with massive hepatic steatosis than in marasmus with limited fatty liver infiltration [1]. The structural N compartment nevertheless participates in the loss of body protein reserves, consistent with the reduced urinary output of creatinine [91], 3-methylhistidine [94] and soluble hydroxyproline [95]. The resulting sarcopenia [96,97] and the concomitant depression of immune mechanisms [98,99] render an account of the higher morbidity / mortality rates affecting TBN-depleted patients identified by the lowest TTR and RBP plasma concentrations [100]. The mortality risk of malnourished children in Central Africa becomes likely when SA and TTR reach the threshold of 16 g /L and 65 mg /L, respectively [101].
During nutritional rehabilitation from protein malnutrition, the restoration of visceral proteins occurs at different rates depending on the type of protein and the size of its plasma pool. TTR and RBP recovery appears as the main result of increased production rates by the liver [102]. Most studies contend the view that the trajectory outlined for TTR correlates with the fluctuations of body N mass, especially during the anabolic phase of growth and clinical recovery from protein malnutrition. Using impedance parameters for assessing the N compartment still remaining in place in the stressed body of adults undergoing renal dialysis, nephrologists were able to demonstrate close relationships between TTR and phase angle, reactance and resistance values [105]. In elderly noninfected persons, FFM index measured by dual X-ray absorptiometry exhibits the highest correlation with TTR (r = 0.64) compared to RBP (r = 0.52) [106]
TTR AS NITROGEN INDEX IN INFLAMMATORY DISORDERS Inflammatory disorders of any cause are initiated by activated leukocytes releasing a shower of cytokines working as autocrine, paracrine and endocrine molecules [107]. Cytokines regulate the overproduction of acute-phase proteins (APPs), notably that of CRP, 1-acid glycoprotein (AGP), fibrinogen, haptoglobin, 1-antitrypsin and antichymotrypsin [107]. APPs contribute in several ways to defense and repair mechanisms, being characterized by proper kinetic and functional properties [107]. Interleukin-6 (IL-6) is regarded as a key mediator governing both the acute and chronic inflammatory processes, as documented by data recorded on burn [108], sepsis [109] and AIDS [110] patients. IL-6-NF possesses a high degree of homology with C/EBP-NF1 and competes for the same DNA response element of the IL-6 gene [111]. IL-6-NF is not expressed under normal circumstances, explaining why APP concentrations are kept at baseline levels. In stressful conditions, IL-6-NF causes a dramatic surge in APP values [107,112] with a concomitant suppressed synthesis of TTR as demonstrated in animal [113] and clinical [114] experiments. Under acute stressful conditions, protein turnover is strongly stimulated by augmented tissue breakdown (mainly in the muscle mass) and enhanced specific tissues synthesis (mainly in the liver and at the site of injury). Proteolysis releases AA residues which are preferentially incorporated into the hepatic precursor pool involved in the production of APPs [115,116]. The rate at which proteins are degraded generally exceeds the rate of AA mobilization for protein synthesis [117,118] yielding a net negative NB associated with an increased urinary output of urea and ammonia [119]. Creatininuria and 3-methylhistidinuria are significantly elevated and remain highly correlated (r = 0.97) attesting to the substantial participation of the skeletal musculature to the stress responses [117]. The gap between degradative and synthetic processes widens in proportion to the severity of injury, resulting in correspondingly increased urinary N catabolites [43]. Serious injury affecting otherwise healthy adults may trigger urinary N losses reaching 40 g/day or 250 g/week, which corresponds to about 15% of TBN [43]. In long-lasting debilitating disorders, the persisting negative NB may deplete the baseline body cell mass by about 45%, carrying ominous prognostic significance [120].
Inadequate nutritional management [122], multiple injuries, occurrence of severe sepsis and metabolic complications result in persistent proteolysis [124] and subnormal TTR concentrations [66]. The evolutionary patterns of urinary N output and of TTR thus appear as mirror images of each other, which supports the view that TTR might well reflect the depletion of TBN in both acute and chronic disease processes. Even in the most complex stressful conditions, the synthesis of visceral proteins is submitted to opposing anabolic or catabolic influences yielding ultimately TTR as an end-product reflecting the prevailing tendency. Whatever the nutritional and/or inflammatory causal factors, the actual TTR plasma level and its course in process of time indicates the exhaustion or restoration of the body N resources, hence its likely (in)ability to assume defense and repair mechanisms. The serial measurement of TTR appears as a dynamic tool pointing to the direction and magnitude of NB, predicting therefore the disease outcome. Hundreds of studies are reporting the clinical usefulness of TTR measurement. TTR is recommended for the assessment and nutritional follow-up of a large panel of hospitalized patients in internal medicine settings [130,131], in general surgery [132,133] and intensive care units [134,135]. Low TTR values thus appear to nonspecifically reflect the extent of liver damage rather than its etiology. Liver N tissue only represents by weight a minor proportion of TBN but its intense turnover rate (10 to 20-fold more rapid than that of muscle tissue) [43] and its critical involvement in the orchestration of most major metabolic and immune pathways [145] explains why liver failure of any cause is usually associated with varying degrees of clinical malnutrition [142].
The nutritional management of kidney patients has met noticeable improvement along the past decades. Until the mid 1980s TTR was regarded as unreliable and discarded, leaving the way for the general use of SA in kidney studies. The turning point came in 1987 when a careful statistical analysis stated that TTR was the most representative marker within a large battery of currently measured parameters [149]. The most recent studies clearly incline towards the common use of TTR superseding that of SA [8, 151-155]. It has been confirmed, mainly in intensive care renal units, that the serial measurement of TTR works as a strong independent predictor of long-term survival, allowing identification of the patients in need of nutritional intervention [151,155] or at risk of reduced life expectancy [154, 155]. Using proportional hazards regression models, the relative risk of death was inversely related to TTR concentrations in 8,157 hemodialyzed patients [155]. TTR is currently measured as nutritional marker in tropical areas where bacterial, viral and parasitic diseases are still highly prevalent, usually in connection with defective immune and vitamin A status, including malaria[156], trypanosomiasis [157], schistosomiasis [158], measles[159], shigellosis [160], and AIDS patients exhibit declining TTR values as the morbid condition worsens [161].
In westernized societies, elderly persons constitute a growing population group. A substantial proportion of them may develop a syndrome of frailty characterized by weight loss, clumsy gait, impaired memory and sensorial aptitudes, poor physical, mental and social activities, depressive trends. Hallmarks of frailty combine progressive depletion of both structural and metabolic N compartments [162]. Sarcopenia and limitation of muscle strength are naturally involutive events of normal ageing which may nevertheless be accelerated by cytokine-induced underlying inflammatory disorders [163,164]. Depletion of visceral resources is substantiated by the shrinking of FFM and its partial replacement by FM, mainly in abdominal organs, and by the down-regulation of indices of growth and protein status [162]. Due to reduced tissue reserves and diminished efficiency of immune and repair mechanisms, any stressful condition affecting old age may trigger more severe clinical impact whereas healing processes require longer duration with erratical setbacks. As a result, protein malnutrition is a common finding in most elderly patients [165] with significantly increased morbidity and mortality rates [166,167].
Measurement of visceral protein status is proved useful throughout the entire ageing lifespan. A wide range of co-morbidities associated with defective protein nutritional status is described in aging persons who become more prone to develop pressure sores [163], osteoporosis [170], oral candidiasis [171] and nuclear cataract [172]. The isolation and purification of rat TTR [173] has allowed to set up animal models. In normal rats, TTR manifests highly significant correlations with nutrient intakes and with visceral and carcass N stores [174]. In tumor-bearing rats, the progressive exhaustion of body protein mass towards cachexia states is correlated with declining TTR values [175]. TTR is currently utilized as indicator of protein nutritional status in cancer patients [176,177]. TTR is held as the most powerful test overall for evaluating visceral protein status of children with solid tumors [178] and leukemias [179] both at the time of diagnosis and throughout chemotherapy. In bone marrow transplantation for malignancies, TTR accurately reflects at any point changes in the patient’s clinical status [180]. TTR has proved to be a useful marker of nutritional alterations with prognostic implications in large bowel cancer [181], bronchopulmonary carcinoid tumor [182], ovarian carcinoma [183] and bladder epithelioma [184]. Many oncologists have observed a rapid TTR fall 2 or 3 months prior to the patient’s death [181]. In cancer patients submitted to surgical intervention, most postoperative complications occurred in subjects with preoperative TTR 180 mg/L [185]. Two independent studies came to the same conclusion that a TTR threshold of 100 mg/L is indicative of extremely weak survival likelihood and that these terminally ill patients better deserve palliative care rather than aggressive therapeutic strategies [185,186].
The AGP/TTR couple is recommended in chronic inflammatory disorders, notably in several cancer types [192,193]. Working along the same lines is the prognostic inflammatory and nutritional index (PINI) [194] which is successfully applied on large cohorts of patients. TTR also participates in the development of screening formulas recently generated by innovative analytical tools such as surface-enhanced laser desorption/ionization (SELDI) or matrix-assisted laser desorption/ionization (MALDI) coupled with time of flight mass spectrometry (TOF-MS). The advent of these sophisticated and costly proteomic fingerprinting studies of serum or other biological fluids are nevertheless promising in that they tentatively strive to identify the early stages of several disease conditions such as hepatitis B [195], tuberculosis [196], Alzheimer’s disease [197] or neoplastic disorders [198]. These proteomic detecting systems usually combine classical APP reactants with some minor biological compounds scarcely measured in routine laboratory practice such as cathepsin D, hemopexin, neopterin or vitronectin. The fact that most, if not all, of these fingerprinting formulas embody TTR measurement indicates that there exists among workers a large consensus considering this carrier-protein as the most reliable indicator of protein depletion in morbid circumstances.
PROGRESS IN TTR RESEARCH : THE BRAIN AGEING PROCESS Dementia, defined as significant memory impairment and loss of intellectual functions, is a common and devastating public health problem, affecting an estimated 2-4% individuals over the age of 65 years. Two distinct clinicopathological conditions are usually taken into consideration as causative factors: Alzheimer’s disease (AD), a chronic and continuously progressing illness for which the only widely accepted risk conditions are age and family history of the disease; and cerebral infarction, a brain deteriorating process evolving along episodic and repetitive bouts so as to generate a syndrome of multi-infarct dementia (MID) [199]. The rates of both AD and MID increase dramatically with age, leading to coexisting pathologies with intermingled symptomatology [200]. In support to this mixed cases concept are the report of equally increased blood-brain barrier permeability in both AD and MID patients [201] and the accumulation of amyloid -protein in the brain of MID subjects mimicking AD pathology [202]. There exists considerable overlap between AD and MID clinical symptoms, giving rise to a continuum of patients in whom pure AD and pure MID represent the two extreme poles [200]. The elevated homocysteine (Hcy) values found in AD patients [208,209] are reportedly associated with dementia [208,210].
The choroid plexus is the sole site of mammalian brain involved in TTR production [214]. Its synthesis rate by the choroid epithelium is estimated 25 to 100 times higher than that of the liver on a weight basis [215]. As a result, TTR is a major component of CSF, constituting 10 to 25 % of total ventricular proteins [216] conveying up to 80% of intrathecal thyroxine [217]. TTR thus constitutes an hormonal carrierprotein fulfilling important ontogenic and functional properties in mammalian nervous structures, a concept further corroborated by the observation of its increased CSF concentration during the neonatal period [218]. The data imply that choroidal TTR facilitates the uptake of thyroxine from the bloodstream, governing its transport and delivery to brain tissues following a kinetic model developed by Australian workers [219]. In comparison, CSF contains 10 to 100 times lower RBP and retinol concentrations than plasma whilst retinyl esters from dietary origin are virtually absent [220]. Although it has been reported that minute amounts of RBP could be produced within the neuraxis [221], the sizeable proportion of retinol molecules required for brain maturation utilizes the RCC transport system to reach the choroid plexus. The very high receptor binding affinity expressed by neural tissues for RBP molecules [222] is confined within the endothelial cells of the brain microvasculature and within the choroidal epithelial cells, the two primary sites of the mammalian blood-brain barrier [223]. The contrast between high RBP binding affinities and low intrathecal concentrations makes it likely that holo-RBP does not experience significant transchoroidal diffusion, strongly suggesting that its retinol ligand is released in free form and readily taken up by membrane or intracellular receptors of neural cells. The dual TTR production, plasma-derived and choroid-secreted, allows complementary stimulation of brain activities. Thyroid hormones and retinoids indeed function in concert through the mediation of common heterodimeric motifs bound to DNA response elements [224,225]. The data also imply that the provision of thyroid molecules within the CSF works as a relatively stable secretory process, poorly sensitive to extracerebral influences [12] as opposed to the delivery of retinoid molecules whose plasma concentrations are highly dependent on nutritional and/or inflammatory alterations [66]. This last statement is documented by mice experiments [226] and clinical investigations [227] showing that the level of TTR production by the liver operates as a limiting factor for retinol transport. Defective TTR synthesis determines the occurrence of secondary hyporetinolemia which nevertheless results from entirely different kinetic mechanisms in the two quoted studies [226,227].
In the TTR knock-out mice model, holo-RBP molecules are normally synthetized and secreted by the liver but undergo rapid kidney leakage in the absence of stabilizing TTR molecules [228]. Despite very low levels of plasma retinol (about 5 % of wild type), these targeted mutated animals remain healthy and fertile, implying that efficient compensatory mechanisms take place. No such increased urinary output of RBP molecules occurs in malnourished patients who develop in proportion to their declining protein status electroretinographic abnormalities and ocular lesions which are pathognomonic symptoms of vitamin A deficiency [229]. During nutritional rehabilitation of malnourished subjects, the 3 RCC components gradually return to normal ranges even without retinol or carotene supplementation, indicating that the retinyl esters normally sequestered in liver stellate cells mandatorily need diet-induced synthesis of new TTR molecules before undergoing retinol conversion and binding as holo-RBP ligand [227]. The prominent place occupied by TTR in defining distal retinoid bioavailability has been too long unrecognized despite the warning expressing that ” overlooking the crucial role of TTR in vitamin A-metabolism results in unachieved or even misleading conclusions ” [66].
Retinol is a precursor substrate that must undergo a two step oxidation procedure to release firstly retinal and thereafter the two active all-trans- and 13-cis-retinoic acids (RAs) [225,230]. The latter converting steps are regulated by retinaldehyde dehydrogenase (RALDH) enzymes whose major sites of expression are the olfactory bulb, the striatum and the hippocampus [231,232]. The intracellular activities exerted by retinoid compounds are mediated by a large variety of specific receptors among which are cellular-RBP (CRBP), cellular-RA-BP (CRABP), RA-nuclear receptors (RARs) and retinoid X receptors (RXRs), each composed of 3 subtypes [225,232]. Retinol is the rate-limiting determinant of the concentration of both RA derivatives [233], implying that any fluctuation in protein status might entail corresponding alterations in the cellular bioavailability of retinoid compounds, with all the more rapid effects as all-trans-RA has a short biological half-life of less than 1 hr [234]. Because protein malnutrition is a common finding in as much as 50 % of elderly AD and MID patients [235], many of them could well suffer permanent hyporetinolemia still accelerating the declining concentration of retinoid molecules observed over the course of normal ageing [231]. Dietary vitamin A is required to modulate early development of brain structure and differentiation [236] together with neuronal plasticity, memory functioning and neurotransmitter signaling during adulthood [237].
The normal decrease of brain retinoid molecules throughout the ageing process principally affects the above-described major sites of RA synthesis [238], a regressive alteration even more pronounced in AD patients [231]. In murine models, early depletion of retinoids causes deposition of amyloid -peptides [239], initiating the formation of Alzheimer plaques. In aged animals, cognitive and memory deficits are associated with down-regulation of the expression of retinoid receptors which may recover their full activities under RA supplementation [240]. Administration of RA similarly restores expression of proteins involved in the control of amyloidogenic pathways [241]. Along the same preventive line is the demonstration that retinol disaggregates preformed amyloid fibrils, more effectively than does RA [242]. Alternatively, TTR participates in the maintenance of memory and normal cognitive processes during ageing by acting on the retinoid signalling pathway as recently reported on TTR-null knock-out mice model [42,243]. Moreover, TTR may bind amyloid -peptide in vitro, preventing its transformation into amyloid neurofibrils [244].
Protein malnutrition, as assessed by diminished TTR plasma values, causes the elevation of Hcy concentrations [245]. There exists an inverse correlation between both TTR and Hcy parameters, explaining why malnourished elderly persons incur increasing risk of Hcy-depended thrombovascular complications [213]. The defective mechanism is situated at the level of cystathionine–synthase (CS), an enzyme governing the crossroad of remethylation and transsulfuration pathways [246]. Japanese workers have recently provided experimental validation of the metabolic anomaly, showing that rats given methionine (Met)-deprived nutriture manifest depressed CS activity with subsequent elevation of Hcy plasma levels [247]. Among all essential AAs consumed in human nutrition, Met is regarded as the most critically available because its withdrawal from the customary diet causes the deepest negative NB, being almost as great as when a protein-free regimen is ingested [248]. Met is implicated in a large spectrum of metabolic and enzyme activities and participates in the conformation of a large number of molecules of survival importance [213]. Due to the fact that plant products are relatively Met-deficient, vegan subjects are more exposed than omnivorous to develop hyperhomocysteinemia – related disorders [249]. Dietary protein restriction may promote supranormal Hcy concentrations which appears as the dark side of adaptive attempts developed by the malnourished and/or stressed body to preserve Met homeostasis. Summing up, we assume that the low TTR concentrations reported in the blood [235] and CSF [250] of AD or MID patients result in impairment of their normal scavenging capacity [244] and in the excessive accumulation of Hcy in body fluids [245], hence causing direct harmful damage to the brain and cardiac vasculature. In addition, depressed TTR concentrations indirectly inhibit the multitude of retinoid-dependent cerebral functioning pathways [231,243] allowing the development of amyloidogenic processes [239]. The practical consequences of these findings imply that the correct assessment of nutritional status is recommended in all elderly patients. The mental and cognitive dysfunctions of old age that are not genetically programmed but result from varying energy, protein and vitamin-deficiencies may be substantially prevented and sometimes improved provided that appropriate nutritional measures are undertaken.
CONCLUDING REMARKS In spite of classical criticisms [3,4], TTR is regarded as a robust and reliable indicator of protein nutritional. Taking into account the gender- and age-specificities, TTR appears as the sole plasma protein reflecting the fluctuations of TBN pools. The relationship linking alterations of TTR plasma levels with body N reserves are documented both in animal models [175] and in human subjects [105,106]. Uncomplicated malnutrition primarily affects the metabolic N pool, reducing protein syntheses and NB to levels compatible with survival, an adaptive response well identified by declining TTR values. In inflammatory disorders, both metabolic and structural N pools participate in varying proportions in the cytokine-induced responses of the stressed body, resulting in TBN shrinking and concomitant depression of TTR concentrations. Abatement of the stressful condition and/or efficient nutritional rehabilitation allows restoration to normal levels of both TBN pools and TTR values following parallel slopes. TTR thus appears as a dynamic index predicting the outcome of the disease. We attached more importance to the trend outlined by its serial appraisal than to any single measurement. Whatever the causal factor, depletion of TBN reserves attenuates the body’s capacity to mount appropriate immune and repair mechanisms. A number of clinical investigations have advocated the level of plasma TTR as predictor of the length of hospital stay (LOS) and of mortality rate [252, 255]. Not surprisingly, unrecognized malnutrition entails longer LOS, increased number of complications and higher care costs whereas early detection and treatment of high risk patients significantly alleviate the financial burden of hospitalization while improving the prognostic outcome of the patients [252-256]. The last statement is documented by the first prospective and randomized survey showing that reduced morbidity and mortality rates are depending on protein N intake and correlated with rising TTR concentrations [257]. Providing elderly persons with optimal protein nutritional status in order to insure their protection against the risk of neurodeterioration is the last message released by the fascinating TTR plasma protein.
Points to consider:
- Protein energy malnutrition has an unlikely causal relationship to carcinogenesis. Perhaps the opposite is true. However, cancer has a relationship to protein energy malnutrition without any doubt. PEM is the consequence of cachexia, whether caused by dietary insufficiency, inflammatory or cancer.
- Protein energy malnutrition leads to hyperhomocysteinemia, and by that means, the relationship of dietary insufficiency of methionine has a relationship to heart disease. This is the significant link between veganism and cardiovascular disease, whether voluntary or by unavailability of adequate source.
1.2 Downsizing of Lean Body Mass is a Key Determinant of Alzheimer’s Disease
Yves Ingenbleek, and Larry H. Bernstein
Journal of Alzheimer’s Disease 44 (2015) 745–754
http://dx.doi.org:/10.3233/JAD-141950
Lean body mass (LBM) encompasses all metabolically active organs distributed into visceral and structural tissue compartments and collecting the bulk of N and K stores of the human body. Transthyretin (TTR) is a plasma protein mainly secreted by the liver within a trimolecular TTR-RBP-retinol complex revealing from birth to old age strikingly similar evolutionary patterns with LBM in health and disease. TTR is also synthesized by the choroid plexus along distinct regulatory pathways. Chronic dietary methionine (Met) deprivation or cytokine-induced inflammatory disorders generates LBM downsizing following differentiated physiopathological processes. Met-restricted regimens downregulate the transsulfuration cascade causing upstream elevation of homocysteine (Hcy) safeguarding Met homeostasis and downstream drop of hydrogen sulfide (H2S) impairing anti-oxidative capacities. Elderly persons constitute a vulnerable population group exposed to increasing Hcy burden and declining H2S protection, notably in plant-eating communities or in the course of inflammatory illnesses. Appropriate correction of defective protein status and eradication of inflammatory processes may restore an appropriate LBM size allowing the hepatic production of the retinol circulating complex to resume, in contrast with the refractory choroidal TTR secretory process. As a result of improved health status, augmented concentrations of plasma-derived TTR and retinol may reach the cerebrospinal fluid and dismantle senile amyloid plaques, contributing to the prevention or the delay of the onset of neurodegenerative events in elderly subjects at risk of Alzheimer’s disease.
Transthyretin and Lean Body Mass in Stable and Stressed State
A Second Look at the Transthyretin Nutrition Inflammatory Conundrum
Stabilizers that prevent transthyretin-mediated cardiomyocyte amyloidotic toxicity
Thyroid Function and Disorders
http://pharmaceuticalintelligence.com/2015/02/05/thyroid-function-and-disorders/
Proteomics, Metabolomics, Signaling Pathways, and Cell Regulation: a Compilation of Articles in the Journal http://pharmaceuticalintelligence.com
Malnutrition in India, high newborn death rate and stunting of children age under five years
Vegan Diet is Sulfur Deficient and Heart Unhealthy
http://pharmaceuticalintelligence.com/2013/11/17/vegan-diet-is-sulfur-deficient-and-heart-unhealthy/
How Methionine Imbalance with Sulfur-Insufficiency Leads to Hyperhomocysteinemia
http://pharmaceuticalintelligence.com/2013/04/04/sulfur-deficiency-leads_to_hyperhomocysteinemia/
Amyloidosis with Cardiomyopathy
http://pharmaceuticalintelligence.com/2013/03/31/amyloidosis-with-cardiomyopathy/
Advances in Separations Technology for the “OMICs” and Clarification of Therapeutic Targets
Sepsis, Multi-organ Dysfunction Syndrome, and Septic Shock: A Conundrum of Signaling Pathways Cascading Out of Control
Automated Inferential Diagnosis of SIRS, sepsis, septic shock
1.3 Transthyretin Blocks Retinol Uptake and Cell Signaling by the Holo-Retinol-Binding Protein Receptor STRA6
Daniel C. Berry, Colleen M. Croniger, Norbert B. Ghyselinck, Noa Noya
Vitamin A is secreted from cellular stores and circulates in blood bound to retinol-binding protein (RBP). In turn, holo-RBP associates in plasma with transthyretin (TTR) to form a ternary RBP-retinol-TTR complex. It is believed that binding to TTR prevents the loss of RBP by filtration in the kidney. At target cells, holo-RBP is recognized by STRA6, a plasma membrane protein that serves a dual role: it mediates uptake of retinol from extracellular RBP into cells, and it functions as a cytokine receptor that, upon binding holo-RBP, triggers a JAK/STAT signaling cascade. We previously showed that STRA6-mediated signaling underlies the ability of RBP to induce insulin resistance. TTR blocks the ability of holo-RBP to associate with STRA6 and thereby effectively suppresses both STRA6-mediated retinol uptake and STRA6-initiated cell signaling. Consequently, TTR protects mice from RBP-induced insulin resistance, reflected by reduced phosphorylation of insulin receptor and glucose tolerance tests. The data indicate that STRA6 functions only under circumstances where the plasma RBP level exceeds that of TTR and demonstrate that, in addition to preventing the loss of RBP, TTR plays a central role in regulating holo-RBP/STRA6 signaling.
1.4 Transthyretin Amyloidosis
1.4.1 (Adapted from a Review in Amyloid: Int J Exp Clin Invest 3:44-56, 1996)
While it was expected that variations in clinical presentation (FAP-I, II, III, IV) were the result of heterogeneity in etiology or pathogenesis of the hereditary amyloidosis, it was not until the discovery by Costa, et al., in 1978 showing transthyretin as a constituent of the fibril deposits, that the biochemical basis of these syndromes could be pursued (Costa, et al., 1978). This resulted in the discovery of the first variant form of transthyretin mutation reported in 1983. In 1989 there were approximately 12 known mutations and in 2002 there are at least 90. Over 80 of these mutations are associated with amyloidosis. In addition, there is evidence that normal transthyretin may for amyloid especially in the heart and be the basis for senile cardiac amyloidosis (Westermark, 1990).
The transthyretin amyloidoses by definition are all associated with tissue deposits of fibrils having transthyretin as a major protein constituent. While there are a number of other constituents of the amyloid deposits, including proteoglycan, amyloid P component, and various lipoproteins, it is transthyretin that is the essential ingredient in this type of amyloid.
It would appear that the signals for down regulating production of transthyretin (cytokines such as IL1 and IL6) are the same as those which cause the positive acute phase response of serum amyloid A and C reactive protein (Costa, et al., 1986). The negative acute phase phenomenon of transthyretin is used by clinicians to monitor nutritional status of their patients.
Transthyretin is firmly entrenched in the phylogenetic evolution of vertebrate species being present in both birds and reptiles and its primary structure has been stable throughout evolution (Richardson, 1994).
While plasma transthyretin is predominantly synthesized by the adult liver, it is also synthesized by the choroids plexus of the brain and mRNA is also present in the retinal pigment epithelium, pituitary and pancreas19, 20 . Choroid plexus synthesis would appear to be necessary for the thyroid hormone across the basement membrane into the cerebral spinal space.
The binding of RBP to transthyretin saves this small protein (21,000 daltons) from plasma clearance via filtration in the kidney. However, when the complex gives up retinal, RBP dissociates from transthyretin and goes to meet its fate. Transthyretin evidently can recirculate to bind more RBP-vitamin A. Plasma residence time of transthyretin is approximately 20-24 hours, representing a plasma half-life of no more than 15 hours (Benson, et al., 1996). This is really very rapid turnover for a plasma protein, compared to plasma residence time of apolipoprotein AI which is 5 days, and that of albumin which is approximately 27 days (t ½ =19 days).
Most variants of transthyretin are not associated with amyloidosis. Most variants of transthyretin are not associated with any postulated “hot spots” in the coding region. The Ser6 variant is the only known polymorphism, prevalence of approximately 12% in the Caucasian population. All the other mutations are present in less than 2% of the population, except in the restricted areas of Northern Sweden where greater than 2% of inhabitants have the Met30 gene and in African Americans, when considered as a group, where approximately 3% have a Val122Ile mutation. One possible explanation of the large number of pathogenic mutations in transthyretin is that the amyloidosis is a delayed onset disease and, therefore, there is a lessened degree of selection against perpetuation of a pathogenic mutation.
Variations on the theme include the involvement of the vitreous of the eye in a number of the kindreds. Approximately a third of transthyretin mutations are associated with vitreous deposits of amyloid; however, this finding is not uniform within families. In different kindreds, a single mutation may have different presentations. Most notably, Swedish patients with Met30 transthyretin have a high incidence of vitreous opacities with presentation at a fairly advanced age (58 years); whereas Portuguese patients have a lower incidence of vitreous opacities, but have presentation of neuropathy at an early age (mean 32 or 33 years). Some transthyretin variants present as pure cardiomyopathy (e.g. Met111) (Frederikson, et al., 1962). The Indiana/Swiss kindred (Ser84) has 100% incidence of cardiomyopathy (Benson and Dwulet, 1983) and this also appears to be true for the Appalachian kindred (Ala60) (Benson, et al., 1987).
Significant renal amyloidosis is less common than cardiac amyloidosis in most of the kindreds. Recently attention has been directed toward kindreds having transthyretin amyloidosis with extensive leptomeningeal amyloid. This is the hallmark of the Ohio kindred with oculoleptomeningeal amyloidosis (Gly30) (Goren, et al., 1980; Peterson, et al., 1997) and a recently reported kindred from Hungary (Gly18) in which the first clinical manifestation is dementia (Vidal, et al.,1996). The His69 mutation has been associated with vitreous opacities alone (Zeldenrust, et al., 1994), but in another family causes oculoleptomeningeal amyloidosis. Features of the disease in particular kindreds make familiarity with the different clinical expressions of the various transthyretin variants essential.
1.4.2 An insight to the conserved water mediated dynamics of catalytic His88 and its recognition to thyroxin and RBP binding residues in human transthyretin
Avik Banerjeea & Bishnu P. Mukhopadhyaya
http://dx.doi.org:/10.1080/07391102.2014.984632
Human transthyretin (hTTR) is a multifunctional protein involved in several amyloidogenic diseases. Besides transportation of thyroxin and vitamin-A, its role towards the catalysis of apolipoprotein-A1 and Aβ-peptide are also drawing interest. The role of water molecules in the catalytic mechanism is still unknown. Extensive analyses of 14 high-resolution X-ray structures of human transthyretin and MD simulation studies have revealed the presence of eight conserved hydrophilic centres near its catalytic zone which may be indispensable for the function, dynamics and stability of the protein. Three water molecules (W1, W2 and W3) form a cluster and play an important role in the recognition of the catalytic and RBP-binding residues. They also induce the reorganisation of the His88 for coupling with other catalytic residues (His90, Glu92). Another water molecule (W5) participate in inter-monomer recognition between the catalytic and thyroxin binding sites. The rest four water molecules (W6, W*, W# and W†) form a distorted tetrahedral cluster and impart stability to the catalytic core of hTTR. The conserved water mediated recognition dynamics of the different functional sites may provide some rational clues towards the understanding of the activity and mechanism of hTTR.
1.4.3 Amyloid Formation by Human Carboxypeptidase D Transthyretin-like Domain under Physiological Conditions*
Javier Garcia-Pardo, Ricardo Graña-Montes, Marc Fernandez-Mendez, et al.
Proteins can form amyloid aggregates from initially folded states. The transthyretin-like domain of human carboxypeptidase D forms amyloid aggregates without extensive unfolding. The monomeric transthyretin fold has an inherent propensity to aggregate due to the presence of preformed amyloidogenic structural elements. Generic aggregation from initially folded states would have a huge impact on cell proteostasis.
1.5 Evolutionary changes to transthyretin: evolution of transthyretin biosynthesis Samantha J. Richardson
FEBS Journal 276 (2009) 5342–53
http://dx.doi.org:/10.1111/j.1742-4658.2009.07244.x
Thyroid hormones are involved in growth and development, particularly of the brain. Thus, it is imperative that these hormones get from their site of synthesis to their sites of action throughout the body and the brain. This role is fulfilled by thyroid hormone distributor proteins. Of particular interest is transthyretin, which in mammals is synthesized in the liver, choroid plexus, meninges, retinal and ciliary pigment epithelia, visceral yolk sac, placenta, pancreas and intestines, whereas the other thyroid hormone distributor proteins are synthesized only in the liver. Transthyretin is synthesized by all classes of vertebrates; however, the tissue specificity of transthyretin gene expression varies widely between classes. This review summarizes what is currently known about the evolution of transthyretin synthesis in vertebrates and presents hypotheses regarding tissue-specific synthesis of transthyretin in each vertebrate class.
1.6 Distinctive binding and structural properties of piscine transthyretin
C Folli, N Pasquato, I Ramazzina, R Battistutta, G Zanotti, R Berni
FEBS Letters 555 (2003) 279-284
http://dx.doi.org:/10.1016/S0014-5793(03)01248-1
The thyroid hormone binding protein transthyretin (TTR) forms a macromolecular complex with the retinol-specific carrier retinol binding protein (RBP) in the blood of higher vertebrates. Piscine TTR is shown here to exhibit high binding affinity for L-thyroxine and negligible affinity for RBP. The 1.56 Ang resolution X-ray structure of sea bream TTR, compared with that of human TTR, reveals a high degree of conservation of the thyroid hormone binding sites. In contrast, some amino acid di¡erences in discrete regions of sea bream TTR appear to be responsible for the lack of protein-protein recognition, providing evidence for the crucial role played by a limited number of residues in the interaction between RBP and TTR. Overall, this study makes it possible to draw conclusions on evolutionary relationships for RBPs and TTRs of phylogenetically distant vertebrates.
1.7 Protein Synthesis at the Blood-Brain Barrier: The Major Proteins Ecreted By Amphibian Choroid Plexus Is A Lipocalin
- Achen, PJ. Harms, T Thomas, SJ. Richardson, REH. Wettenhall, G Schreiber J Biol Chemistry Nov 1992; 267(32): 23167-70Among the proteins secreted by choroid plexus of vertebrates, one protein is much more abundant than all others. In mammals, birds, and reptiles this protein is transthyretin, a tetramer of identical 15-kDa sub- units. In this study choroid plexus from frogs, tadpoles, and toads incubated in vitro were found to synthesize and secrete one predominant protein. However, this consisted of one single 20-kDa polypeptide chain. It was expressed throughout amphibian metamorphosis. Part of its amino acid sequence was determined and used for construction of oligonucleotides for polymerase chain reaction. The amplified DNA was used to screen a toad choroid plexus cDNA library. Full-length cDNA clones were isolated and sequenced. The derived amino acid sequence for the encoded protein was 183 amino acids long, including a 20-amino acid preseg- ment. The calculated molecular weight of the mature protein was 18,500. Sequence comparison with other proteins showed that the protein belonged to the lipocalin superfamily. Its expression was highest in choroid plexus, much lower in other brain areas, and absent from liver. Since no transthyretin was detected in proteins secreted from amphibian choroid plexus, abundant synthesis and secretion of transthyretin in choroid plexus must have evolved only after the stage of the amphibians.
2 Vitamin A
2.1 Retinoic acid pathways and cancer
2.1.1 Vitamin A, Cancer Treatment and Prevention: The New Role of Cellular Retinol Binding Proteins
Elena Doldo,Gaetana Costanza,Sara Agostinelli,Chiara Tarquini, et al.
BioMed Research International 2015; Article ID 624627, 14 pages
http://dx.doi.org/10.1155/2015/624627
Retinol and vitamin A derivatives influence cell differentiation, proliferation, and apoptosis and play an important physiologic role in a wide range of biological processes. Retinol is obtained from foods of animal origin. Retinol derivatives are fundamental for vision, while retinoic acid is essential for skin and bone growth. Intracellular retinoid bioavailability is regulated by the presence of specific cytoplasmic retinol and retinoic acid binding proteins (CRBPs and CRABPs). CRBP-1, the most diffuse CRBP isoform, is a small 15KD acytosolic protein widely expressed and evolutionarily conserved in many tissues. CRBP-1 acts as chaperone and regulates the uptake, subsequent esterification, and bioavailability of retinol. CRBP-1 plays a major role in wound healing and arterial tissue remodeling processes. In the last years, the role of CRBP-1-related retinoid signaling during cancer progression became object of several studies. CRBP-1 downregulation associates with a more malignant phenotype in breast, ovarian, and nasopharyngeal cancers.Reexpression of CRBP-1 increased retinol sensitivity and reduced viability of ovarian cancer cells in vitro. Further studies are needed to explore new therapeutic strategies aimed at restoring CRBP-1-mediated intracellular retinol trafficking and the meaning of CRBP-1 expression in cancer patients’ screening for a more personalized and efficacy retinoid therapy.
Metabolism of Retinol and Its Derivatives. Vitamin A can be acquired from the diet either as preformed vitamin A (primarily as retinyl ester, retinol, and in much smaller amount as retinoic acid) or provitamin A carotenoids (Figure1). Dietary retinyl esters are converted to retinol within the lumen of the small intestine or the intestinal mucosa and then reesterified to form retinyl ester (RE) within the enterocyte [1]. Provitamin A carotenoids, absorbed by the mucosal cells, are converted first to retinaldehyde and then to retinol [1]. After secretion of the nascent chylomicrons into the lymphatic system, the bulk of dietary vitamin A is taken up by hepatocytes and hydrolyzed again.The free retinol binds the epididymal retinoic acid binding protein (ERABP) and the retinol binding protein (RBP) [2] and into plasma transthyretin. Free retinol can be transferred to hepatic stellate cells for storage. Hepatocytes and hepatic stellate cells are very rich in retinyl ester hydrolases and in cellular retinol binding protein type 1 (CRBP-1). CRBP-1 is necessary to solubilize retinol in the aqueous environment of the cell [1].
Intracellular Trafficking of Retinoids. A cell-surface receptor named stimulated by retinoic acid 6 (STRA6) mediates vitamin A uptake from RBP [3]. Intracellular retinoid bioavailability is regulated by the presence of specific cytoplasmic retinol and retinoic acid binding proteins, CRBPs and CRABPs (Figure2). In the cytoplasm vitamin A and derivatives are bound to cytoplasmic proteins: cellular retinol binding proteins (CRBPs) which comprised four isoforms, CRBP-1 and CRBP-2 and CRBP-3 and CRBP-4. CRBP-1, are the most represented isoform in many tissues. Cellular retinoic acid binding proteins (CRABPs) comprised two isoforms, CRABP-1 and CRABP-2. CRBPs specifically bind retinol, while CRABPs and well-characterized members of the fatty acid binding proteins (FABPs) bind retinoic acid (RA). These proteins control the availability of ligands and determine the physiological response of cells and tissues to vitamin A [4]. Cellular retinoic acid binding proteins may regulate the interactions between retinoic acids and their nuclear receptors by regulating the concentrationof present retinoic acids [5]. Retinoids can activate gene expression by specific nuclear retinoid acid receptors. Two distinct classes of nuclear proteins, the retinoic acid receptors (RARs), and the retinoid X receptors (RXRs) have been identified. Each class consists of 𝛼, 𝛽,and 𝛾 subtypes. RARs and RXRs form either homodimers or heterodimers and function as transacting nuclear transcriptional factors [6]. RAR can be activated by both all-trans and 9-cis RA, whereas RXR is only activated by 9-cis-RA.
2.1.2 Retinoids, retinoic acid receptors, and cancer.
Tang XH1, Gudas LJ.
Annu Rev Pathol. 2011; 6:345-64
http://dx.doi.org:/10.1146/annurev-pathol-011110-130303
Retinoids (i.e., vitamin A, all-trans retinoic acid, and related signaling molecules) induce the differentiation of various types of stem cells. Nuclear retinoic acid receptors mediate most but not all of the effects of retinoids. Retinoid signaling is often compromised early in carcinogenesis, which suggests that a reduction in retinoid signaling may be required for tumor development. Retinoids interact with other signaling pathways, including estrogen signaling in breast cancer. Retinoids are used to treat cancer, in part because of their ability to induce differentiation and arrest proliferation. Delivery of retinoids to patients is challenging because of the rapid metabolism of some retinoids and because epigenetic changes can render cells retinoid resistant. Successful cancer therapy with retinoids is likely to require combination therapy with drugs that regulate the epigenome, such as DNA methyltransferase and histone deacetylase inhibitors, as well as classical chemotherapeutic agents. Thus, retinoid research benefits both cancer prevention and cancer treatment.
2.1.3 Molecular pathways: current role and future directions of the retinoic acid pathway in cancer prevention and treatment.
Connolly RM1, Nguyen NK, Sukumar S.
Clin Cancer Res. 2013 Apr 1; 19(7):1651-9
http://dx.doi.org:/10.1158/1078-0432.CCR-12-3175
Retinoids and their naturally metabolized and synthetic products (e.g., all-trans retinoic acid, 13-cis retinoic acid, bexarotene) induce differentiation in various cell types. Retinoids exert their actions mainly through binding to the nuclear retinoic acid receptors (α, β, γ), which are transcriptional and homeostatic regulators with functions that are often compromised early in neoplastic transformation. The retinoids have been investigated extensively for their use in cancer prevention and treatment. Success has been achieved with their use in the treatment of subtypes of leukemia harboring chromosomal translocations. Promising results have been observed in the breast cancer prevention setting, where fenretinide prevention trials have provided a strong rationale for further investigation in young women at high risk for breast cancer. Ongoing phase III randomized trials investigating retinoids in combination with chemotherapy in non-small cell lung cancer aim to definitively characterize the role of retinoids in this tumor type. The limited treatment success observed to date in the prevention and treatment of solid tumors may relate to the frequent epigenetic silencing of RARβ. Robust evaluation of RARβ and downstream genes may permit optimized use of retinoids in the solid tumor arena.
Vitamin A is derived from animal and plant food sources and has critical functions in many aspects of human biology. Its natural derivatives and metabolized products (retinoids) such as β-carotene, retinol, retinal, isotetrinoin, all-trans retinoic acid (ATRA), 9-cis retinoic acid, and 13-cis retinoic acid have important roles in cell differentiation, growth, and apoptosis (1). Synthetic retinoids are also available and include bexarotene and fenretinide. In clinical practice, retinoids have a wide range of dermatologic indications including for psoriasis, acneiform, and keratinization disorders (2). Systemic retinoids are approved by the U.S. Food and Drug Administration (FDA) for the treatment of cutaneous T-cell lymphoma (3) and acute promyelocytic leukemia (APL; refs. 4, 5). However, the chemopreventive and therapeutic effects of retinoids in solid tumors remain controversial. Therefore, an overview of the research to date and future directions in this area is the focus of this review.
Retinoic acid and the retinoic acid receptor pathway
Retinoic acids (RA) exert their functions through their specific receptors. The 2 distinct classes of receptors are retinoic acid receptors (RAR) and retinoic X receptors (RXR). Each class contains 3 different subtypes—α, β, and γ (6). ATRA and fenretinide can bind specifically to RARS, 13-cis RA and bexarotene only to RXRS, and 9-cis RA to RARS or RXRS (refs. 1, 5; Table 1). The expression of these receptors is regulated by the receptors themselves, other nuclear receptors such as ERα, or by other subtypes in the same family (5, 7). Upon the binding of ligands, RARs and RXRs form heterodimers and function as ligand-dependent transcription factors to activate their downstream effectors by binding to the retinoic acid response elements (RARE) located in the 5′-region of RA downstream genes (5). The above model of RAR or RXR function via binding to RARE is considered the RA classical or genomic pathway. Activation of the classical pathway will trigger cell differentiation, cell arrest, and eventual apoptosis (8).
Table 1. Select clinical trials evaluating retinoids in solid tumors
| Retinoid | Other names | Target | Clinical trial setting |
| ATRA | Tretinoin | RAR | Advanced NSCLC Phase II randomized (n = 107) |
| 13-cis RA | Isotretinoin Roaccutane Accutane | RXR | Primary prevention: H+N cancer |
| Advanced solid tumorsPhase I (n = 13) | |||
| Metastatic breast cancer Phase II randomized (n = 99) | |||
| 9-cis RA | Alitretinoin | RAR RXR | Metastatic breast cancerPhase I (n = 12) |
| Fenretinide | 4-OH Phenylretinamide | RAR | Primary prevention: women at high risk of breast cancer Randomized double-blind 2 × 2 design (n = 235) |
| Secondary prevention: early breast cancerPhase III randomized (n = 2,867) | |||
| Bexarotene | RXR | Chemotherapy-naïve advanced NSCLC Phase III randomized (n = 623) | |
| Metastatic breast cancer Phase II single arm (n = 148) |
The function of RA and its receptors involves not only the classical pathway but also multiple other important pathways. RAs have been shown to regulate NF-κB (9), IFN-γ (10), TGF-β (11), VEGF (12), mitogen-activated protein kinase (MAPK; ref. 13), and chromatin remodeling (14). Furthermore, RARs and RXRs can form heterodimers with other types of receptors, including the estrogen receptor-α (ERα; refs. 7, 15), AP-1 receptor (16), peroxisome proliferator-activated receptor (PPAR; ref. 17), liver X receptors (LXR; refs. 18, 19), and vitamin D receptor (VDR; ref. 20; Fig. 1). When RARs/RXRs heterodimerize with these receptors, they are involved in regulating their partner receptor’s pathways, referred to as nonclassical or nongenomic pathways (5). Interestingly, these pathways often regulate processes that have functions opposite to the classical pathway. For example, a study has shown that RA activation of the PPARβ/δ pathway resulted in upregulation of prosurvival genes (17), contrary to the known differentiation function of RARs and RXRs in response to RA. The function of RAs, which involves nongenomic pathways, may provide opportunities for cancer cells to develop resistance to RA treatment, discussed later in this review. Another important function of RARA is the regulation of stem cell differentiation (11). RAs target stem cells via both genomic and nongenomic pathways such as the Notch pathway and inflammation (10, 11). In summary, RAs and their receptors play important roles as regulators of critical processes in cells.
The RARs and their action. In a series of enzymatic steps, vitamin A (retinol) is metabolized through the oxidizing action of retinaldehyde (RDH) to retinal, and by retinaldehyde dehydrogenase (RALDH), to RA. RA has 3 different isomers: all-trans, 9-cis, and 13-cis RA. RA is transported to the nucleus by the protein cellular RA–binding protein (CRABP) and delivered to the RARα. RARα heterodimerizes with and binds to RARE present most often in gene promoters. In the classical pathway of RA action, RA binds to dimers of RARα and RXRs (α, β, or γ) to induce expression of its downstream target genes, including RARβ. Upon activation, RARβ can regulate its own expression and that of its downstream genes, the function of which is mainly to inhibit cell growth. Alternatively, RA can be bound and transported to the nucleus by other factors such as FABP5. This delivers RA to other nonclassical receptors such as PPARβ/δ and ERα which activate nongenomic pathways such as PDK-1/Akt or the ERα pathway. Contrary to the differentiation functions attributed to the classical pathway, the nongenomic pathways exert strong antiapoptotic and proliferative effects on cancer cells. It is believed that the classical and nongenomic pathways are controlled by the relative abundance of their own ligands. RA has a stronger affinity for RARs than for the other receptors, and the classical pathway plays a dominant role over the nongenomic pathways. Thus, if RA is present with other ligands such as estrogen, signaling through the classical pathway is preferred to result in cell differentiation and growth inhibition.
http://clincancerres.aacrjournals.org/content/19/7/1651/F1.small.gif
Retinoids and cancer
The retinoids have been investigated extensively for the prevention and treatment of cancer, predominantly because of their ability to induce cellular differentiation and arrest proliferation. RA-regulated tumor suppressor genes, when expressed, can inhibit tumor growth (21). Among the 3 RARs, RARβ has been well known for its tumor-suppressive effects in epithelial cells (5, 8, 22). Exogenous expression of the RARβ gene can cause RA-dependent and -independent apoptosis and growth arrest (23). RARβ-induced growth arrest and apoptosis is mediated through RARα (24). As RA ligand-bound RARα binds to the RARE on the RARβ promoter, multiple activator proteins assemble at the site and result in the upregulation of the RARβ gene (5). The expression of RARβ results in the transactivation and expression of a number of its target genes that mediate cell differentiation and death (5, 6, 8). The ability of ATRA to initiate differentiation of promyelocytic leukemic cells to granulocytes is the basis of the dramatic success of retinoic acid therapy for acute promyelocytic leukemia harboring the RAR/PML translocation (4) and confirms the important role of RARβ in tumor growth inhibition. It is also becoming increasingly clear that RARβ expression is lost early in carcinogenesis or is epigenetically silenced (25) in many solid tumors, providing an opportunity for novel treatment strategies to be investigated using retinoids together with epigenetic modifiers that promote reexpression of silenced genes, described further below.
The retinoids have an established role in the treatment of certain hematologic malignancies, with FDA approval for use in cutaneous T-cell lymphoma and APL. Bexarotene (an RXR-selective retinoid or rexinoid) is associated with an overall response rate of approximately 50% in patients with refractory advanced-stage mycosis fungoides, a cutaneous T-cell lymphoma (3). ATRA, a synthetic retinoid, exhibited improvements in disease-free and overall survival when compared with chemotherapy alone in APL, with long-term remissions occurring in almost 70% of cases (4). The success of retinoids in treating this disease relates to the underlying chromosomal translocation and production of the PML/RARα fusion protein and the ability of retinoids to induce differentiation and inhibition of cell growth in this setting (26, 27). Clinical trials investigating the role of retinoids in the prevention and treatment of solid tumors will now be outlined with a focus on cancers of the upper aerodigestive tract (oropharyngeal and lung) and breast (Table 1).
2.1.4 Retinoid Pathway and Cancer Therapeutics
Nathan Bushue and Yu-Jui Yvonne Wan
Adv Drug Deliv Rev. 2010 Oct 30; 62(13): 1285–1298.
http://dx.doi.org/10.1016%2Fj.addr.2010.07.003
The retinoids are a class of compounds that are structurally related to vitamin A. Retinoic acid, which is the active metabolite of retinol, regulates a wide range of biological processes including development, differentiation, proliferation, and apoptosis. Retinoids exert their effects through a variety of binding proteins including cellular retinol binding protein (CRBP), retinol-binding proteins (RBP), cellular retinoic acid-binding protein (CRABP), and nuclear receptors i.e. retinoic acid receptor (RAR) and retinoid × receptor (RXR). Because of the pleiotropic effects of retinoids, understanding the function of these binding proteins and nuclear receptors assists us in developing compounds that have specific effects. This review summarizes our current understanding of how retinoids are processed and act with the emphasis on the application of retinoids in cancer treatment and prevention.
Vitamin A and its derivatives (retinoids) exert a wide range of effects on embryonic development, cell growth, differentiation, and apoptosis. Vitamin A has been used as a treatment for thousands of years. The Egyptian papyruses Kahun 1 (ca. 1825 B.C.) and Ebers (ca. 1500 B.C.) described how the liver was used to cure eye diseases such as night blindness. Greek scholar Hippocrates (460-327 B.C.) described in the second book of “Prognostics” a method for curing night blindness: “raw beef liver, as large as possible, soaked in honey, to be taken once or twice by mouth.” Chinese medicine used pigs’ liver as a remedy for night blindness, as described by Sun-szu-mo (7th century A.D.) in his “1000 Golden Remedies”. Given that the liver is where the body stores excess vitamin A, the liver represents the best source of vitamin A available for treatment in the pre-pharmaceutical world.
The effect of vitamin A on growth was first described in a mouse experiment done by G. Lunin (1881) [2], in which one group of mice was fed pure casein, fat, sucrose, minerals, and water, and another group was fed whole dried milk. The milk-fed group was healthy and grew normally, while the other group was sick and ultimately died. Thus, something in milk was essential for survival. Elmer McCollum at University of Wisconsin-Madison as well as Lafayette Mendel and Thomas Burr Osborne at Yale University independently discovered vitamin A. McCollum began his study in 1907 by feeding cows hay with wheat, oats, or yellow maize.
Wheat-fed cows did not thrive, became blind and gave birth to dead calves prematurely. Oat-fed cows fared somewhat better, but the yellow maize-fed cows were in excellent condition, produced vigorous calves, and had no miscarriages. McCollum postulated that performing the same nutritional study using small animals, such as rodents, which require less food, provide faster reproduction and experimental outcome. Using rats, he found a diet of pure protein, pure milk sugar, minerals, and lard (or olive oil) inhibited growth, while addition of butterfat or an ether extract of egg yolk to the diet restored health. Thinking that he had found a fat-soluble factor that promoted growth in rats, he saponified butterfat, extracted the unsaponifiable mixture into ether, and added the extract to oliveoil and that extract could support growth. This essential component to support growth and development was named “fat-soluble factor A,” and later renamed vitamin A [1].
There are over 4,000 natural and synthetic molecules structurally and/or functionally related to vitamin A. Vitamin A cannot be synthesized by any animal species and is only obtained through diet in the form of retinol, retinyl ester, or β-carotene (Figure 1). Ingested vitamin A is stored as retinyl esters in hepatic stellate cells. Retinol is reversibly oxidized by retinol dehydrogenases to yield retinal. Subsequently, retinal may be irreversibly oxidized to all-trans retinoic acid (all-trans RA) by retinal dehydrogenases and further oxidized by cytochrome P450 enzymes (mainly CYP26) in hepatic tissue. Retinol has six biologically active isoforms that include all-trans, 11-cis, 13-cis, 9, 13-di-cis, 9-cis, and 11, 13-di-cis, with all-trans being the predominant physiological form. Endogenous retinoids with biological activity include all-trans RA, 9-cis RA, 11-cis retinaldehyde, 3,4-didehydro RA, and perhaps 14-hydroxy-4, 14-retro retinol, 4-oxo RA, and 4-oxo retinol [3–5]. All-trans RA isomerizes under experimental and physiological conditions. Different isomers activate different receptors and thus lead to different biological effects. RAs designed to be receptor specific can improve efficacy and avoid unwanted side effects. Retinoids that specifically bind to RXR are called rexinoids and have been effective in cancer treatment. Retinoids are comprised of three units: a bulky hydrophobic region, a linker unit, and a polar terminus, which is usually a carboxylic acid. Modification of each unit has generated many more compounds. Please refer to recent reviews [6–8].
2.1.4.1 Retinoid Pathway
http://www.ncbi.nlm.nih.gov/pmc/articles/PMC2991380/bin/nihms229611f1.jpg
Retinoids absorbed from food are converted to retinol and bound to CRBP in the intestine. Then, retinol is converted to retinyl esters and enters into blood circulation. The liver up takes retinyl esters, which are converted to retinol-RBP complex in the hepatocyte. In the serum, the retinol-RBP complex is bound to transthyretin (TTR) in a 1:1 ratio to prevent elimination by the kidney and to ensure retinol is delivered to the target cell. The uptake of retinol by the target cell is mediated by a trans-membrane protein named “stimulated by retinoic acid 6” (STRA6), which is a RBP receptor. In the target cell, retinol either binds to CRBP or is oxidized to retinaldehyde by retinol dehydrogenase (RDH) in a reversible reaction. Then, retinaldehyde can be oxidized by retinaldehyde dehydrogenase (RALDH) to RA. In the target cell, RA either binds to CRABP or enters the nucleus and binds to nuclear receptors to regulate gene transcription. Alternatively, RA can mediate via nongenomic mechanism and regulate cellular function. Hepatocytes not only process retinoids, but also are the target cells. In addition, hepatocytes located next to the storage site (stellate cell). Thus, retinoid-mediated signaling must have a profound effect in regulating hepatocyte function and phenotype [36, 190, 191].
2.1.4.2 Retinoid Binding Proteins
There are various types of retinoid-binding proteins, which locate in intracellular and extracellular compartments and associate with isomeric forms of retinoids. Hence, retinoids are either associated with cellular membranes or bound to a specific retinoid binding protein. These binding proteins along with nuclear receptors mediate the action of retinoids. Their interactions are summarized in figure 1. Retinoid-binding proteins solubilize and stabilize retinoids in aqueous spaces. In addition to this general role, specific retinoid-binding proteins have distinct functions in regulating transport and metabolism of specific retinoids. For example, the parent vitamin A molecule, all-trans retinol, circulates in blood bound to serum retinol binding protein (RBP). Inside the cells, all-trans retinol and its oxidation product, all-trans retinal, are associated with different isoforms of cellular retinol-binding proteins (CRBP), while all-trans RA intracellularly binds to cellular retinoic acid-binding protein isoforms (CRABP).
2.1.4.3 RBP
Retinol is secreted from its storage pools and circulates in blood by binding to RBP. The main storage site for vitamin A and the main site of synthesis of RBP is the liver, although other tissues (including adipose tissue, kidney, lung, heart, skeletal muscle, spleen, eye and testis) also express this protein. Secretion of RBP from the liver is regulated by the availability of retinol [9]. Vitamin A deficiency inhibits RBP secretion, leading to protein accumulation in the endoplasmic reticulum of hepatic parenchymal cells. In the presence of retinol, RBP associates with retinol, moves to the Golgi apparatus and is secreted into blood. The mechanism by which retinol initiates RBP secretion from cells is not known. In blood, RBP is bound to the small protein transthyretin, which in addition to associating with RBP functions as a carrier protein for thyroid hormones. Binding of RBP to transthyretin prevents the loss of this smaller protein by filtration in the renal glomeruli. The transthyretin-RBP-retinol complex transports retinol in the circulation and delivers it to target tissues [10].
Important insights into the biological role of RBP have been obtained by studies of mice and humans in which the RBP gene is disrupted. RBP-deficient mice display both reduced blood retinol levels and impaired visual function during the first months of life. When maintained on a vitamin A-sufficient diet, they acquire normal vision by 5 months of age, even though their blood retinol level remains low. A striking phenotype of the RBP-null mice is that they possess larger than normal hepatic vitamin A storage, but are dependent on a continuous dietary intake of vitamin A [11], further proving the importance of RBP as a transporting protein. A study of two human siblings that harbored point mutations in their RBP gene and exhibited undetectable plasma RBP levels revealed that these sisters suffered from night blindness and mild retinal dystrophy but did not exhibit other clinical symptoms of vitamin A deficiency [12]. Taken together, RBP is critical for the mobilization of retinol from hepatic storage pools; however, RBP is not essential for the delivery of retinol to target tissues. Supply of vitamin A to target tissues in the absence of RBP is likely to be accomplished via newly absorbed retinyl esters or β-carotene present in circulating chylomicrons. Increased RBP has been shown to contribute to insulin resistance and type 2 diabetes [11]. All-trans RA has recently been shown to increase insulin sensitivity in diabetic mice while lowering RBP [13]. The effect on binding proteins must be considered when retinoids are used for disease treatment.
2.1.4.4 SRA6
The stimulated by retinoic acid gene 6 (STRA6) encodes the cell surface RBP receptor, which binds specifically to RBP and mediates retinol uptake from holo-RBP [14]. STRA6 is a widely expressed transmembrane protein. In mouse mammary epithelial cells, STRA6 expression can be up regulated by Wnt1 and retinoids. In addition, STRA6 mRNA levels are up regulated in mouse mammary gland tumors and human colorectal tumors [15]. Importantly, while the RBP-null mice and humans give rise to relative mild phenotypes, STRA6-null mice develop anophthalmia, congenital heart defects, diaphragmatic hernias, alveolar capillary dysplasia, lung hypoplasia, and mental retardation. These findings suggest that STRA6 may have additional functions that are not related to RBP transport [16].
2.1.4.5 CRBP
CRBPs belong to the family of fatty acid binding proteins in which expression of CRBP family members are tissue specific. For example, CRBP-II is expressed only in the enterocytes of the intestine, while CRBP-I and -III are expressed throughout embryonic and adult tissues [17]. Knockout studies for CRBP isoforms have identified differences in function due to altered tissue localization. CRBP-I knockout mice are healthy. However, they have low levels of hepatic retinyl esters [18], and their hepatic lipid droplets appear to be smaller and less abundant than in wild type littermates. CRBP-II-null mice have impaired retinol uptake, but they develop and reproduce normally under vitamin A-enriched diet, albeit with reduced retinol storage [19]. Reduction of vitamin A in the maternal diet of CRBP-II-null mice during gestation results in neonatal mortality immediately following birth [19]. CRBP-III null mice have impaired vitamin A incorporation into milk, but they are otherwise healthy [20]. CRBP-I and CRBP-III compensate for each other to maintain normal retinoid homeostasis, but the compensation is incomplete during lactation [20]. The binding affinity of CRBP-I towards retinol is about 100-fold higher than that of CRBP-II. They display a similar binding affinity towards retinal and CRBP-II associates with retinol and retinal with similar affinities. CRBPs, and especially CRBP-I with its high affinity for retinol, may sequester retinol from its ability to disrupt cell membranes. Epigenetic silencing of CRBP is a common event in human cancers [21]. Silencing CRBP reduces the availability of retinyl esters in the bloodstream and decreases the body’s ability to metabolize retinol [22].
2.1.4.6 CRABP
CRABP-I and -II have been identified with a high affinity for all-trans RA. In humans, these isoforms display 74 percent sequence identity and are highly conserved among species; however, these CRABP isoforms display different patterns of expression across cells and developmental stages. In adults, CRABP-I is expressed ubiquitously, while CRABP-II is only expressed in the skin, uterus, ovary, and the choroid plexus. Both CRABPs are widely expressed in the embryo, although they do not usually co-exist in the same cells. The biological functions of CRABPs are not completely understood. In mouse knockout stu dies, disruption of either CRABP-I or -II only display mild defects in limb development [23], which suggests CRABPs may be involved in generation of appropriate RA concentration gradients in the developing limb bud. Both CRABP isoforms are present in cytosol and nucleus and thus may deliver the ligand directly to the nuclear receptor. The differential role of these two binding proteins remains to be studied (reviewed in [24] and [25]). Increased CRABP-I expression may also contribute to RA resistance of cancer cells [26]. The effect of CRABP on cancer therapy deserves more attention.
2.1.4.7 Retinoic Acid Receptors
The major breakthrough in understanding RA’s function occurred upon identifying and cloning the receptors for RA [27, 28]. RA regulates gene expression by binding to its nuclear receptors, which in turn activates transcription of their downstream target genes. Thus, retinoids exert their biological functions primarily by regulating gene expression. This was predicted by Sporn and Roberts in 1983, when they wrote: “Ultimately, it would appear that the problem of the molecular mechanism of action of retinoids in control of differentiation and carcinogenesis is converging on one of the central problems of all biology, the control of gene expression.” [29]
2.1.4.8 RAR and RXR
Two distinct classes of receptors for retinoids have been identified: retinoic acid receptors (RAR) and retinoid × receptors (RXR). Each class of receptor contains three subtypes – α, β, and γ. RARs can be activated by both all-trans and 9-cis-RA, while, RXRs are exclusively activated by 9-cis RA. However, due to the conversion of all-trans to 9-cis RA, high concentrations (10−5 M) of all-trans RA can also activate gene transcription in cells transfected with RXRs [30].
RXRs can form homo- and heterodimers with other receptors. In fact, RXRs are promiscuous receptors forming heterodimers with many different kinds of receptors, which include receptors for fatty acids [peroxisomal proliferator activated receptors (PPAR)], bile acids [farnesoid × receptor (FXR)], oxysterols [liver × receptor (LXR)], xenobiotics [pregnane × receptor (PXR) and constitutive androstane receptor (CAR)], vitamin D [vitamin D receptor (VDR)], and RA (RAR). RXRs can also form homodimers. Hypervitaminosis A leads to bone fracture suggesting that vitamin A and D compete for the same receptor [31]. Within these heterodimers, RXRs can exist as both active and silent partners. When it serves as an active partner, 9-cis RA and the ligand for the heterodimeric partner can activate the heterodimer, and addition both ligands give synergistic induction in gene transcription. For example, RXR is an active partner for PPAR. Similarly, heterodimeric complexes of RXR with LXR or FXR also retain 9-cis RA responsiveness. Thus, RAs can regulate PPAR- and FXR-mediated pathways [32]. Recently, we demonstrated that RAs could also activate PXR-, VDR, and CAR-mediated signaling and thus regulated xenobiotic metabolism and potentially its own oxidation [33–35]. When RXR serves as a silent partner, the heterodimer of RXR and its partner does not respond to RA. Regardless of their active or silent role, RXRs must be present in order to exert biological actions of various nuclear receptors. Using hepatocyte RXRα-deficient mice [36, 37], we have demonstrated that RXRα does play vital roles in xenobiotic (alcohol, acetaminophen) and endobiotic (fatty acid, cholesterol, amino acid, and carbohydrate) metabolism [33–40]. Thus, RXR functions as an auxiliary factor and determines the effects of other hormones, making RXR a master regulator. The structure of nuclear receptors is summarized in recent review articles [7, 38].
Existing data suggest that the binding protein and receptor work together to exert the specific effect of RAs. For example, RAs can bind to both PPARβ, the receptor for fatty acids, and RAR. Fatty acid-binding protein 5 (FABP5) and CRABP-II are specific binding proteins that channel RAs from the cytosol into the nucleus for binding to either PPARβ or RAR, respectively [39]. The ratio of FABP5/CRABP-II concentrations determines which receptor is activated. By activating PPARβ, RAs induce expression of genes affecting lipid and glucose homeostasis, such as the insulin-signaling gene pyruvate dehydrogenase kinase 1 (PDK1), which enhances insulin action. Hence, RAs stimulate lipolysis and reduce triglyceride content. RA implantation into obese mice causes up regulation of PPARβ as well as an increased expression of PPARβ target genes, including PDK1, which led to weight loss [40].
2.1.4.9 Retinoids and Cancer
Retinoids are widely used to treat visual and dermatological diseases. Their effect on cancer prevention and treatment has received a lot of attention. This review focuses on the action of retinoids on cancer. Retinoids have been used as potential chemotherapeutic or chemopreventive agents because of their differentiation, anti-proliferative, pro-apoptotic, and anti-oxidant effects. Epidemiological studies show that lower vitamin A intake results in a higher risk of developing cancer, which aligns with observations of vitamin A-deficient animals [61]. Altered expression of RA receptors is also associated with malignant transformation of animal tissues or cultured cells. Furthermore, retinoids suppress carcinogenesis in tumorigenic animal models for skin, oral, lung, breast, bladder, ovarian, and prostate [62–68]. In humans, retinoids reverse premalignant human epithelial lesions, induce the differentiation of myeloid cells, and prevent lung, liver, and breast cancer [69–73].
The following is a summary of how major retinoids may work in cancer treatment or prevention.
2.1.4.9.1 All-trans RA (tretinoin)
All-trans RA is the most abundant natural retinoid and has been widely studied for many years. It is currently in clinical trials for the treatment of lymphoma, leukemia, melanoma, lung cancer, cervical cancer, kidney cancer, neuroblastoma, and glioblastoma. The most effective clinical usage of all-trans RA in human disease was demonstrated in treatment of a rare leukemia, acute promyelocytic leukemia (APL). APL is characterized by selected expansion of immature myeloid precursors or malignant myeloid cells blocked at the promyelocytic stage of hemopoietic development. APL cells invariably express aberrant fusion proteins involving the DNA and ligand binding domain of RARα [74, 75]. Other fusion partners include the promyelocytic leukemia zinc finger gene, the nucleophosmin gene, the nuclear mitotic apparatus gene, and the Stat5b gene, while the most common fusion partner is promyelocytic leukemia protein (PML). The PML-RARα chimeric receptor is created by a balanced reciprocal chromosomal translocation, t(15;17)(q22:q11). The expressed PML-RARα chimeric receptor alters normal function of RARs. PML-RARα can form a homodimer through the coiled-coil motif of PML, inhibiting RARα’s ability to bind to RA responsive elements, thereby preventing activation of downstream target genes [76, 77]. In addition, RXR is an essential component of the oncogenic PML/RARα complex suggesting RXR can be a drug target for APL [78, 79]. In 1995, the FDA approved all-trans RA for treating APL. The all-trans RA-induced differentiation of APL cells is due to both its ability to promote the degradation of the mutant PML-RARα and the dissociation of its co-repressors [80]. All-trans RA also causes cell cycle arrest at G1 phase and inhibits cell proliferation [81]. In addition, high concentration of all-trans RA induces post-maturation apoptosis of APL-blasts through the induction of the tumor-selective death ligand tumor necrosis factor-related apoptosis-inducing ligand TRAIL [82].
RA syndrome is a life-threatening complication seen in APL patients treated with all-trans RA. This syndrome is characterized by dyspnea, fever, weight gain, hypotension, and pulmonary infiltrates. It can be effectively treated by giving dexamethasone and holding off all-trans RA treatment in severe cases. An elevated white count is sometimes associated with this syndrome, but is not a prerequisite. The etiology of RA syndrome is not clear; several causes have been speculated including a capillary leak syndrome from cytokine release from the differentiating myeloid cells. Alternatively, all-trans RA may cause the maturing myeloid cells to acquire the ability to infiltrate organs such as the lung [83].
2.1.4.9.2. 9-cis RA (alitretinoin)
9-cis RA differentiates itself from all-trans RA in its ability to activate both RAR and RXR. In addition, 9-cis RA activates PPAR, FXR, PXR, VDR, and CAR via RXR. In preclinical studies, 9-cis RA is effective in the prevention of mammary and prostate cancer [84, 85] and it has also been FDA-approved for the topical treatment of cutaneous lesions of Kaposi’s sarcoma [86]. In addition, 9-cis RA and all-trans RA can individually induce apoptosis of human liver cancer cells [87]. 9-cis RA not only regulates nuclear genes, but also mitochondria gene transcription [88].
2.1.4.9.3. 13-cis RA (isotretinoin)
13-cis RA is unique that it exhibits immunomodulatory and anti-inflammatory responses. It inhibits ornithine decarboxylase, thereby decreasing polyamine synthesis and keratinization [89]. 13-cis RA noticeably reduces the production of sebum and shrinks the sebaceous glands [90]. It stabilizes keratinization and prevents comedones formation [91, 92]. The exact mechanism of action is unknown. This combination of regulating proliferation, differentiation, and inflammation could make 13-cis RA a more effective drug in comparison to other retinoids, which may cause inflammation and irritation [93].
13-cis RA is in clinical trial for different types of cancers, and thyroid cancer received a lot of attention. In follicular thyroid cancer cells, 13-cis RA induces radioiodine avidity of cells formerly unable to accumulate radioiodine [94]. In human thyroid carcinoma cell lines, retinoids induce the expression of type I iodothyronine-5′-deiodinase and sodium/iodide-symporter, which are the thyroid differentiation markers [95]. However, approximately 30% of thyroid tumors dedifferentiate after treatment and thus develop into highly malignant anaplastic thyroid carcinomas [96]. 13-cis RA is also used to treat non-operable thyroid follicular tumors, which fail to uptake radioiodine. 13-cis RA increases the radioiodide uptake in some patients. The beneficial outcome of this treatment was interpreted as partial re-differentiation of thyroid cancer cells. This effect of 13-cis RA requires the existence of functional RXR [96]. The effect of 13-cis RA on thyroid cancer has been reviewed extensively [97]. Besides thyroid cancer, utilizing 13-cis RA for maintenance therapy has significantly improved the outcome of patients with a high-risk form of neuroblastoma [98]. Along the same line of work, Krüppel zinc-finger protein ZNF423 is critical for RA signaling and is likely a prognostic marker for neuroblastoma [99]. 13-cis RA is also effective in preventing head and neck cancer, which is discussed below.
2.1.4.9.4. Synthetic Retinoids
N-(4-hydroxyphenyl) retinamide (Fenretinide or 4HPR) was first synthesized in the late 1960s by R. W. Pharmaceuticals. Since then, the biological properties of fenretinide have been of great interest. Currently, fenretinide is one of the most promising clinically tested retinoids. The modification of the carboxyl end of all-trans RA with an N-4-hydroxyphenyl group resulted in increased efficacy as a chemoprevention agent as well as reduced toxicity when compared with other retinoids [100]. Animal models have demonstrated that treatment with fenretinide prevents chemically induced cancers of the breast, prostate, bladder, and skin [101–104]. Furthermore, the combination of tamoxifen with fenretinide produces efficacy greater than either chemical alone [105].
Natural retinoids like all-trans RA induce differentiation and/or cytostasis in target cells [106–108], while fenretinide has distinct biologic effects including the induction of apoptosis by generating reactive oxygen species (ROS) and lipid second messengers [104]. The apoptotic effect of fenretinide has been documented in a variety of cancer cells including transformed T cells, B cells and breast epithelial cells, as well as bladder, breast, cervical, colon, embryonal, esophageal, head and neck, lung, ovarian, pancreatic, prostate, and skin carcinomas [100]. Furthermore, fenretinide does not induce point mutations or chromosomal aberrations, and is therefore not genotoxic [109]. These qualities suggest that fenretinide could be used for a long-term chemopreventive modality. In animal models, fenretinide has demonstrated chemopreventive efficacy against carcinogenesis of the breast [110], prostate, pancreas, and skin [104, 111, 112]. Moreover, in a clinical setting, fenretinide slowed the progression of prostate cancer in men diagnosed with an early stage of the disease [113]. Fenretinide protected against the development of ovarian cancer and a second breast malignancy in premenopausal women who had been treated to prevent the progression of early-stage breast cancer [114]. It also prevented relapse and the formation of secondary primary lesions in patients following the surgical removal of oral leukoplakia [115]. Recent studies also illustrated the anti-angiogenic [116] and anti-fibrotic [117] effect of fenretinide. Furthermore, long-term fenretinide treatment prevents high-fat diet-induced obesity, insulin resistance, and hepatic steatosis [118].
The mechanisms associated with fenretinide-induced apoptosis have been explored, but are not well-understood [100]. The components that lead to ROS generation and cause cell death are largely unknown. Depending on cell types and models used, the effect of fenretinide has been shown to be RARβ-dependent or -independent [119]. Our data showed that fenretinide-induced apoptosis of human liver cancer cells was RARβ-dependent [120]. Furthermore, induction and cytoplasmic localization of Nur77 dictates the sensitivity of liver cancer cell to fenretinide-induced apoptosis [121]. It seems that fenretinide enriches the cytoplasmic Nur77 to target mitochondria and induce cell death. The relationship between RARβ and Nur77 in mediating fenretinide-induced apoptosis remains to be determined.
A retinoid-related molecule 6-[3-(1-adamantyl)-4-hydroxyphenyl]-2-naphthalenecar-boxylic acid (AHPN) (also called CD437) and it’s analog (E)-4-[3-(1-adamantyl)-4-hydroxyphenyl]-3-chlorocinnamic acid (3-Cl-AHPC) also have Nur77-dependent apoptotic effects [122–124]. AHPN is structurally distinct from fenretinide. AHPN-induced apoptosis activates JNK [125–127], which is required for maximal apoptosis induction and precedes mitochondrial depolarization. Induction of apoptosis of breast and prostate cancer cells by AHPN is also associated with its inhibition of Akt activity [128]. Thus, induction of JNK and inhibition of Akt phosphorylation of Nur77 contribute to Nur77 nuclear export mediated by AHPN [129].
While many synthesized RAs are promising for cancer treatment, only a few are FDA-approved or currently undergoing clinical trials for cancer therapy. A number of retinoids, which have been FDA-approved for dermatological purposes, have potential for cancer treatment. Bexarotene (Targretin) is a synthetic retinoid approved by the FDA to treat skin problems caused by cutaneous T-cell lymphoma that are unresponsive to other treatments [130]. Other synthetic retinoids, such as TAC-101 (Taiho Pharmaceutical, Tokyo, Japan) has shown efficacy in inhibiting tumor growth in the liver and markedly increases survival in both the primary HCC and metastatic colon cancer models [131]. TAC-101 is currently in phase II trial for hepatocellular carcinoma and has shown good preliminary success [132]. Another, Tazarotene (AVAGE™) (Allergan, Irvine, CA) is in phase I trials for the treatment of lymphoma [133]. Please see table 1 for a brief characterization of some of retinoids that are in use or in clinical trials.
3 Vitamin C
3.1 American Cancer Society
Vitamin C is an essential vitamin the human body needs to function well. It is a water-soluble vitamin that cannot be made by the body, and must be obtained from foods or other sources. Vitamin C is found in abundance in citrus fruits such as oranges, grapefruit, and lemons, and in green leafy vegetables, tomatoes, potatoes, strawberries, red or green peppers, and cantaloupe.
Vitamin C is found in many vegetables and fruits, especially oranges, grapefruits, and peppers. Many studies have shown a link between eating foods rich in vitamin C, such as fruits and vegetables, and a reduced risk of cancer. On the other hand, the few studies in which vitamin C has been given as a supplement have not shown a reduced cancer risk.
This suggests that the activity of fruits and vegetables in preventing cancer is due to a combination of many things such as vitamins, fiber, and other phytochemicals and not to vitamin C alone (see Phytochemicals). Clinical trials of high doses vitamin C as a treatment for cancer have not shown any benefit. High doses of vitamin C can cause side effects in some people.
3.2 Intravenous vitamin C for cancer
Oct 4, 2013 | By Dr. Ronald Hoffman
For more than 20 years, the Hoffman Center has been using high-dose vitamin C drips in its cancer support protocols. The initial impetus was from Linus Pauling who, together with Ewan Cameron, pioneered the use of high-dose C in cancer in the 1960s.
Now, there’s new interest in this modality for fighting cancer based on new, exciting research under way at the National Institutes of Health.
Cameron and Pauling found that vitamin C helped cancer patients live about four times longer than cancer patients not given vitamin C. They administered high-dose vitamin C in the form of sodium ascorbate given orally and intravenously to treat more than 1,000 cancer patients.
Nonetheless, vitamin C for cancer suffered a setback when Dr. Charles Moertel of the Mayo Clinic, an arch foe of nutritional therapies for cancer, sought to disprove Pauling’s thesis. But he did not follow the Pauling/Cameron instructions or regimen.
Moertel selected a cohort of terminal colon cancer patients who had not responded to all forms of conventional treatment, including surgery, chemo and radiation, and administered 10 grams of vitamin C to them orally. When the patients failed to demonstrate improved survival over patients not receiving vitamin C in the study, Moertel pronounced the vitamin C/cancer hypothesis defunct.
Moertel failed to note that the benefits achieved by Pauling and Cameron’s patients were obtained via both IV and oral C. He ultimately succumbed to cancer himself years later.
Alternative practitioners, meanwhile, sought to resurrect IV vitamin C as a tool in the treatment of cancer, but not until recently has serious academic research resumed.
Dr. Hugh Riordan of Kansas treated hundreds of cancer patients with doses of vitamin C up to 200,000 mg (200 grams) per day in infusions lasting 4-12 hours several times a week. He compiled a series of case histories documenting impressive responses but passed recently, before his work was generally acknowledged.
His protegee, Dr. Jeanne Drisko, Director, KU Integrative Medicine, has undertaken a series of clinical trials to validate the benefits of IV vitamin C in cancer. An FDA approved trial is now underway.
Research at the National Institutes of Health is beginning to suggest that vitamin C deserves another chance to find its niche in the arsenal of anti-cancer therapies. Studies now suggest that even high dose vitamin C given by mouth is poorly absorbed. Blood levels “max out” at doses of 500 mg given several times during the day.
But vitamin C given intravenously is another story. When delivered in a “drip,” much higher concentrations of C can be attained. At these higher concentrations, vitamin C has different characteristics than if given orally. While oral vitamin C boosts immunity and assists tissue repair, it is too weak to do much to kill or inhibit cancer cells. But at high doses delivered directly into the bloodstream, it may act to increase levels of hydrogen peroxide deep in the tissues where cancer cells lurk. Peroxide-mediated killing is one of the white blood cells’ key mechanisms for fighting infection and cancer.
Research currently under way has shown that high concentrations of vitamin C can stop the growth or even kill a wide range of cancer cells. Only intravenous administration of vitamin C can deliver the high doses found to be effective against cancer.
IV vitamin C, when administered by a trained, experienced physician, is safe and well-tolerated, even at doses as high as 100,000 mg (100 grams) per day. Proper blood tests must be done to ensure that it is well-tolerated, and the patient must be monitored. Doses must be gradually adjusted upward. Not all patients are candidates for IV vitamin C. Vitamin C can be safely administered even while patients are undergoing chemo and radiation; in fact, the FDA-approved trial at the University of Kansas Medical Center explicitly permits the co-administration of vitamin C with conventional treatments.
3.3 IV Vitamin C Kills Cancer Cells
by Dr. Julian Whitaker
By now, most people know that vitamin C is a potent antioxidant that has the power to boost immune function, increase resistance to infection, and protect against a wide range of diseases.
But there’s an entirely different and largely unknown role of vitamin C, and that is its ability—when administered in very high doses by intravenous (IV) infusions—to kill cancer cells.
Vitamin C interacts with iron and other metals to create hydrogen peroxide. In high concentrations, hydrogen peroxide damages the DNA and mitochondria of cancer cells, shuts down their energy supply, and kills them outright. Best of all—and unlike virtually all conventional chemotherapy drugs that destroy cancer cells—it is selectively toxic. No matter how high the concentration, vitamin C does not harm healthy cells.
Lab studies reveal that this therapy is effective against many types of cancer, including lung, brain, colon, breast, pancreatic, and ovarian. Animal studies show that when human cancers are grafted into animals, high-dose IV vitamin C decreases tumor size by 41 to 53 percent “in diverse cancer types known for both their aggressive growth and limited treatment options.” Additionally, numerous patient case reports have been written up in medical journals.
Why IV Administration Is Essential
The only way to get blood levels of vitamin C to the concentrations required to kill cancer cells is to administer it intravenously. The body tightly controls levels of this vitamin by limiting intestinal absorption. If you took 10 g (10,000 mg) of vitamin C by mouth at one time, you would only absorb around 500 mg—and you’d get a serious case of diarrhea!
Intravenous administration, however, bypasses this control mechanism, and blood levels rise in a dose-dependent manner. For example, 10 g of IV vitamin C raises blood levels 25 times higher than the same dose taken orally, and this increases up to 70-fold as doses get larger.
4 Expert Q&A: Vitamin D and Cancer Risk
Vitamin D is one of several nutrients that the body needs to stay healthy. It may also play a role in reducing the risk of cancer, and several research studies are exploring this link. Cancer.Net talked with Richard Goldberg, MD, to learn more about current research on vitamin D and what people should know.
Q: What is the role of vitamin D in the body, and what are some sources of this vitamin?
A: One role of vitamin D is to regulate the absorption of calcium by the body. Calcium is the main component of bones and is important in the function of all cells in the body, particularly the heart. People who are vitamin D deficient (don’t get enough) can have weakened bones (a condition called osteoporosis in adults and rickets or osteomalacia in children). Too little calcium (called hypocalcemia) in the body can lead to irregular heartbeat and muscle spasms.
Milk, fish, eggs, and fortified cereals and orange juice are good sources of vitamin D. Milk manufactured in the United States is generally fortified with vitamin D as a way to prevent deficiencies from occurring. Supplemental vitamins are also a source.
Unlike other vitamins that the body cannot produce by itself, vitamin D can either be absorbed directly from the intestine or made from compounds in foods. The body can make vitamin D from nutrients related to cholesterol. These nutrients are then converted to vitamin D as they circulate in the blood when a person’s skin is exposed to sunlight.
Too much vitamin D can also be bad for a person, leading to drowsiness, kidney stones, bone or muscle weakness, and elevated blood calcium, a condition called hypercalcemia that can cause confusion and, in extreme cases, death.
Q: When getting vitamin D from sunlight, how long should a person be exposed to the sun? What are the risks of too much sun exposure?
A: While 90% of the body’s vitamin D comes from exposure to sun (in the absence of vitamin D supplements), the amount of sun exposure needed to produce adequate vitamin D levels is actually quite limited. Sun exposure at the equator is far more intense than in such northern cities as Boston or London, for instance, and is more intense anywhere in summer than in winter. However, it takes only five to ten minutes of exposing the hands and face three times a week to receive adequate sun exposure in the summer in Boston. Exposure of more skin, such as when wearing a bathing suit, requires only a very short time in the sun. Use of sunblock is very important when sun exposure is longer than that to prevent skin cancer, including melanoma, and other sun-induced damage such as wrinkling and pigmentation changes (sunspots). Learn more about protecting your skin from the sun.
Q: How might vitamin D work to help lower the risk of cancer?
A: Laboratory studies have shown that vitamin D deficiency can lead to decreased communication between cells and leads them to stop sticking to one another, a condition that could cause cancer cells to spread. Compared with normal cells, cancer cells remain in an immature state, and vitamin D appears to have a role in making cells mature. Vitamin D also appears to play a role in regulating cellular reproduction, which malfunctions (doesn’t work properly) in cancer. Higher levels of vitamin D lead to cellular adherence, maturation, and communication between cells, all of which may lower cancer risk.
Q: What does research show about vitamin D levels and cancer?
A: Studies in populations have shown that low vitamin D levels are a risk factor for cancer in general, and particularly for prostate, colorectal, and breast cancers.
There are also data that correlate high blood levels of vitamin D with a reduced risk of breast and colorectal cancers. These levels can best be achieved by taking supplemental vitamin D. In colorectal cancer, calcium supplementation may also reduce the risk of polyps (noncancerous growths that may develop on the inner wall of the colon and rectum) and cancer. Numerous studies have tested cancer risk by giving patients supplemental vitamin D, with or without calcium supplementation. While the results are somewhat variable, substantial reduction (on the order of 50%) in the odds of breast and colon cancers with supplementation, have been noted in some studies. People with a personal history of these types of cancer and their relatives may wish to discuss supplementation with their doctors.
5 Magnesium and Cancer Research
5.1 Dr Sircus on Mar 18, 2010
http://drsircus.com/medicine/magnesium/magnesium-and-cancer
Aleksandrowicz et al in Poland conclude that inadequacy of magnesium and antioxidants are important risk factors in predisposing to leukemias.[2] Other researchers found that 46% of the patients admitted to an ICU in a tertiary cancer center presented hypomagnesemia. They concluded that the incidence of hypomagnesemia in critically ill cancer patients is high.[3]In animal studies we find that magnesium deficiency has caused lymphopoietic neoplasms in young rats. A study of rats surviving magnesium deficiency sufficient to cause death in convulsions during early infancy in some, and cardiorenal lesions weeks later in others, disclosed that some of survivors had thymic nodules or lymphosarcoma.[4]
One would not normally think that Magnesium (Mg) deficiency can paradoxically increase the risk of, or protect against cancer yet we will find that just as severe dehydration or asphyxiation can cause death magnesium deficiency can directly lead to cancer. When you consider that over 300 enzymes and ion transport require magnesium and that its role in fatty acid and phospholipids acid metabolism affects permeability and stability of membranes, we can see that magnesium deficiency would lead to physiological decline in cells setting the stage for cancer. Anything that weakens cell physiology will lead to the infections that surround and penetrate tumor tissues. These infections are proving to be an integral part of cancer. Magnesium deficiency poses a direct threat to the health of our cells. Without sufficient amounts our cells calcify and rot. Breeding grounds for yeast and fungi colonies they become, invaders all too ready to strangle our life force and kill us.
Over 300 different enzymes systems rely upon magnesium to facilitate their catalytic action, including ATP metabolism, creatine-kinase activation, adenylate-cyclase, and sodium-potassium-ATPase.[5]
It is known that carcinogenesis induces magnesium distribution disturbances, which cause magnesium mobilization through blood cells and magnesium depletion in non-neoplastic tissues. Magnesium deficiency seems to be carcinogenic, and in case of solid tumors, a high level of supplemented magnesium inhibits carcinogenesis.[6] Both carcinogenesis and magnesium deficiency increase the plasma membrane permeability and fluidity. Scientists have in fact found out that there is much less Mg++ binding to membrane phospholipids of cancer cells, than to normal cell membranes.[7]
Magnesium protects cells from aluminum, mercury, lead, cadmium, beryllium and nickel.
Magnesium in general is essential for the survival of our cells but takes on further importance in the age of toxicity where our bodies are being bombarded on a daily basis with heavy metals.Glutathione requires magnesium for its synthesis.[8] Glutathione synthetase requires ?-glutamyl cysteine, glycine, ATP, and magnesium ions to form glutathione.[9] In magnesium deficiency, the enzyme y-glutamyl transpeptidase is lowered.[10] According to Dr. Russell Blaylock, low magnesium is associated with dramatic increases in free radical generation as well as glutathione depletion and this is vital since glutathione is one of the few antioxidant molecules known to neutralize mercury.[11]Without the cleaning and chelating work of glutathione (magnesium) cells begin to decay as cellular filth and heavy metals accumulates; excellent environments to attract deadly infection/cancer.
There is drastic change in ionic flux from the outer and inner cell membranes both in the impaired membranes of cancer, and in Mg deficiency.
Anghileri et al[12],[13] proposed that modifications of cell membranes are principal triggering factors in cell transformation leading to cancer. Using cells from induced cancers, they found that there is much less magnesium binding to membrane phospholipids of cancer cells, than to normal cell membranes.[14] It has been suggested that Mg deficiency may trigger carcinogenesis by increasing membrane permeability.[15] Magnesium deficient cells membranes seem to have a smoother surface than normal, and decreased membrane viscosity, analogous to changes in human leukemia cells.[16],[17] There is drastic change in ionic flux from the outer and inner cell membranes (higher Ca and Na; lower Mg and K levels), both in the impaired membranes of cancer, and of Mg deficiency. And we find that lead (Pb) salts, are more leukemogenic when given to Mg deficient rats, than when they are given to Mg-adequate rats, suggesting that Mg is protective.[18]
Magnesium has an effect on a variety of cell membranes through a process involving calcium channels and ion transport mechanisms. Magnesium is responsible for the maintenance of the trans-membrane gradients of sodium and potassium.
Long ago researchers postulated that magnesium supplementation of those who are Mg deficient, like chronic alcoholics, might decrease emergence of malignancies[19] and now modern researchers have found that all types of alcohol — wine, beer or liquor — add equally to the risk of developing breast cancer in women. The researchers, led by Dr. Arthur Klatsky of the Kaiser Permanente Medical Care Program in Oakland, Calif., revealed their findings at a meeting of the European Cancer Organization in Barcelona in late 2007. It was found that women who had one or two drinks a day increased their risk of developing breast cancer by 10 percent. Women who had more than three drinks a day raised their risk by 30 percent. The more one drinks the more one drives down magnesium levels.
Breast cancer is the second most common cancer killer of women, after lung cancer. It will be diagnosed in 1.2 million people globally this year and will kill 500,000.
According to data published in the British Journal of Cancer in 2002, 4 percent of all breast cancers — about 44,000 cases a year — in the United Kingdom are due to alcohol consumption. It’s an important question though, and one not asked by medical or health officials, is it the alcohol itself or the resultant drop in magnesium levels that is cancer provoking? Though some studies have shown that light- to moderate alcohol use can protect against heart attacks it does us no good to drink if it causes cancer. Perhaps if magnesium was supplemented in women drinkers who were studied there would have been no increase of cancer from drinking.
Alcohol has always been known to deplete magnesium, and is one of the first supplements given to alcoholics when they stop and attempt to detoxify and withdraw.
Researchers from the School of Public Health at the University of Minnesota have just concluded thatdiets rich in magnesium reduced the occurrence of colon cancer.[20] A previous study from Sweden[21] reported that women with the highest magnesium intake had a 40 per cent lower risk of developing the cancer than those with the lowest intake of the mineral.
Magnesium stabilizes ATP[22], allowing DNA and RNA transcriptions and repairs.[23]
The anti-colon cancer effects of calcium are linked to magnesium levels, says a new study. Researchers from Vanderbilt University found that low ratios of the minerals were associated with reduced risk of colorectal cancer, according to findings presented at the Seventh Annual American Association for Cancer Research International Conference on Frontiers in Cancer Prevention Research. Both high magnesium and calcium levels have been linked to reduced risks of colon cancer but studies have also shown that high calcium levels inhibit the absorption of magnesium. According to Qi Dai, MD, PhD, and co-workers, Americans have high calcium intake, but also a high incidence of colorectal cancer. “If calcium levels were involved alone, you’d expect the opposite direction. There may be something about these two factors combined – the ratio of one to the other – that might be at play,” said Dai. The risk of colorectal cancer adenoma recurrence was reduced by 32 per cent among those with baseline calcium to magnesium ratio below the median in comparison to no reduction for those above the median,” said Dai.[24]
Pre-treatment hypomagnesemia has been reported in young leukemic children, 78% of whom have histories of anorexia, and have excessive gut and urinary losses of Mg.[25]
Several studies have shown an increased cancer rate in regions with low magnesium levels in soil and drinking water, and the same for selenium. In Egypt the cancer rate was only about 10% of that in Europe and America. In the rural fellah it was practically non-existent. The main difference was an extremely high magnesium intake of 2.5 to 3g in these cancer-free populations, ten times more than in most western countries.[26]
5.2 Magnesium and cancer: a dangerous liason.
Castiglioni S, Maier JA.
Magnes Res. 2011 Sep; 24(3):S92-100
http://dx.doi.org:/10.1684/mrh.2011.0285
A complex relationship links magnesium and cancer. The aim of this review is to revisit current knowledge concerning the contribution of magnesium to tumorigenesis, from transformed cells to animal models, and ending with data from human studies. Cultured neoplastic cells tend to accumulate magnesium. High intracellular levels of the cation seem to confer a metabolic advantage to the cells, contribute to alterations of the genome, and promote the acquisition of an immortal phenotype. In magnesium-deficient mice, low magnesium both limits and fosters tumorigenesis, since inhibition of tumor growth at its primary site is observed in the face of increased metastatic colonization. Epidemiological studies identify magnesium deficiency as a risk factor for some types of human cancers. In addition, impaired magnesium homeostasis is reported in cancer patients, and frequently complicates therapy with some anti-cancer drugs. More studies should be undertaken in order to disclose whether a simple and inexpensive intervention to optimize magnesium intake might be helpful in the prevention and treatment of cancer.
Even though cancer-associated death rates are falling steadily, the global burden of cancer continues to increase primarily as a result of an aging population, but also because of the adoption of cancer-causing behaviors, including smoking and a western-type diet [1]. In particular, statistical and epidemiological data point to diet as responsible for about 35% of human cancer mortality [2]. There is general agreement about the inverse correlation between the risk of cancer and the regular consumption of fruit, cereals and vegetables, rich sources of many beneficial micronutrients, vitamins and minerals. Magnesium, which is predominantly obtained by eating unprocessed grains and green leafy vegetables, is an essential micronutrient implicated in a wide variety of regulatory, metabolic and structural activities [3]. The occidental diet is relatively deficient in magnesium Presented in part at the European Magnesium Meeting – EUROMAG Bologna 2011, San Giovanni in Monte, Bologna, Italy, June 8-10, 2011. because of the processing of many food items and the preference for calorie-rich, micronutrient-poor foods [4]. Magnesium deficiency complicates chronic gastrointestinal and renal diseases, diabetes mellitus, alcoholism, and therapies with some classes of diuretics and anticancer drugs [4]. A review of the literature reveals the relationship between magnesium and cancer, from the cellular level through to animal models and humans. Although controversy exists about the role of magnesium in tumors, most of the results available point to low magnesium as a factor contributing to tumorigenesis.
5.1.1 Magnesium acts as a secondary messenger, and activates a vast array of enzymes [3, 5]. Since magnesium participates in all major metabolic processes, as well as redox reactions, it is no surprise that it has a direct role in controlling cell survival and growth. In normal diploid cells, the total concentration of magnesium increases throughout the G1 and S phases of the cell cycle. Accordingly, low extracellular magnesium markedly inhibits their proliferation [3]. Conversely, neoplastic cells are refractory to the proliferative inhibition by low extracellular magnesium but, being extremely avid for the cation, it accumulates in these cells even when cultured in low magnesium levels [6]. This avidity is due, at least in part, to an impairment of Na-dependent magnesium extrusion [7], and to the overexpression of one of the magnesium transporters, namely transient receptor potential melastatin (TRPM)7 [8]. High intracellular magnesium seems to provide a selective advantage for the transformed cells since magnesium contributes to regulating enzymes of various metabolic pathways and of the systems involved in DNA repair. Indeed, magnesium forms complexes with ATP, ADP and GTP, necessary for the activity of enzymes implicated in the transfer of phosphate groups such as glucokinase, phosphofructokinase, phosphoglycerate kinase and pyruvate kinase [9], enzymes of glycolysis known to be the pathway used preferentially by neoplastic cells to produce energy [10]. Magnesium also forms complexes with DNA polymerase, ribonucleases, adenylcyclase, phosphodiesterases,guanylate-cyclase, ATPases and GTPases, being therefore implicated in the metabolism of nucleic acids and proteins, and in signal transduction [9]. Since mutation is a driving force in the development of cancer, it is worth noting that magnesium is involved in the inhibition of N-methylpurine DNA-glycosidase, which initiates base excision repair in DNA by removing a wide variety of alkylated, deaminated, and lipid peroxidation-induced purine adducts [11]. In addition, the nuclear Ser/Thr phosphatase PPM1D (also known as WIP1), which is overexpressed in various human primary tumors, requires magnesium for its activity. PPM1D is involved in the regulation of several essential signaling pathways implicated in tumorigenesis [12, 13]. In particular, PPM1D dephosphorylates and, therefore, inactivates the p53 tumor suppressor gene, a canonical suppressor of proliferation. It also complements several oncogenes, such as Ras, Myc, and HER-2/neu, for cellular transformation both in vitro and in vivo [12].
On these bases, it is possible to conclude that high intracellular magnesium has a role in promoting genetic instability. Another peculiarity of tumor cells is their limitless proliferative potential [14, 15]. It is therefore relevant to point out that magnesium is required to activate telomerase [16-18], a specialized DNA polymerase that extends telomeric DNA and counters the progressive telomere erosion associated with cell duplication. The presence of telomerase activity correlates with a resistance to induction of both senescence and apoptosis which are considered to be crucial anticancer defenses [14, 15]. These points are summarized in figure 1, which also underlines the contribution of high intracellular magnesium to some of the hallmarks of cancer, as highlighted by Hanahan and Weinberg [14, 15]. Mentioning only studies performed on neoplastic cells would be simplistic, since tumors are more than just masses of proliferating cancer cells. Rather, they are complex, heterotypic tissues where normal cells in the stroma, far from being passive bystanders, actively collaborate to cancer development and progression [14, 15]. Many of the growth signals driving the proliferation of and invasion by carcinoma cells originate from the stromal cell components of the tumor mass. It is therefore worth noting that low magnesium modulates the functions of a variety of normal cells present in the tumor microenvironment. In particular, endothelial cells cultured in low magnesium release higher amounts of metalloproteases and growth factors [19]. Similar results were obtained in cultured human fibroblasts (unpublished results). In addition, low magnesium promotes endothelial and fibroblast senescence [20], and senescent cells can modify the tissue environment in a way that synergizes with oncogenic mutations to promote the progression of cancers [21]. Only the behavior of microvascular endothelial cells cultured in low magnesium seems not to fit with the picture described above. It is well known that angiogenesis is crucial to nourish the tumor and facilitate its spreading, but low extracellular magnesium impairs acquisition of the angiogenic phenotype by microvascular endothelial cells. Exposure to low magnesium retards endothelial proliferation, migration and differentiation in vitro ([22] and manuscript submitted). Accordingly, magnesium-deficient mice develop tumors which are significantly less vascularized than the controls [23].
Figure 1. Neoplastic cells tend to have high intracellular concentrations of magnesium, which contribute to the regulation of various metabolic pathways and of systems involved in DNA repair, thus providing a selective advantage for the transformed cells. The figure also links the effects of high intracellular concentrations of magnesium on cell functions to some hallmarks of cancer as highlighted by Hanahan and Weinberg [14, 15].
5.1.2 Low magnesium and cancer: a focus on human studies
Several epidemiological studies have provided evidence that a correlation exists between dietary magnesium and various types of cancer. High levels of magnesium in drinking water protect against oesophageal and liver cancer [36, 37]. In addition, magnesium concentration in drinking water is inversely correlated with death from breast, prostate, and ovarian cancers, whereas no correlation existed for other tumors [36, 38, 39]. Epidemiological studies conducted in various countries demonstrate an association between low intake of magnesium and the risk of colon cancer [40-43]. In addition, a large population-based prospective study in Japan shows a significant inverse correlation between dietary intake of magnesium and colon cancer in men but not in women [44]. Intriguingly, the association between low intake of magnesium and colon cancer is linked to the increased formation of N-nitroso compounds, most of which are potent carcinogens [43]. A further link between magnesium and colon neoplasia is highlighted by the association of adenomatous and hyperplastic polyps, which might progress to carcinoma, with a genetic polymorphism of TRPM7 [45], an ubiquitous ion channel with a central role in magnesium uptake and homeostasis [46]. Results concerning the contribution of magnesium to lung cancer are controversial. A first case-control study correlates low dietary magnesium with increased lung cancer risk both in men and women [47]. This link is more evident in the elderly, current smokers, drinkers and in those with a late-stage disease. To explain the protective effect of magnesium against lung cancer, the authors recall that magnesium regulates cell multiplication, protects against the oxidative stress invariably associated with magnesium deficiency [48], and maintains genomic stability. A recent prospective analysis however, does not support the previous report [49]. These contrasting data could result from recall bias, the difficulty in evaluating diet composition and the fact that smoking is a very strong risk factor for lung cancer.
Conclusion Although the evidence is still fragmentary, most of the data available point to magnesium as a chemopreventive agent, so that optimizing magnesium intake might represent an effective and low-cost preventive measure to reduce cancer risk. Doubts remain about supplementing cancer patients with magnesium. The recently revived interest in the relationship between magnesium and tumors, both in experimental and clinical oncology, should encourage more studies that would advance our understanding of the role of magnesium in tumors, and could explore the possibility that optimizing magnesium homeostasis might prevent cancer or help in its treatment.
5.3 A Magnesium Deficiency Increases Cancer Risk Significantly
Wed, May 21, 2008 by: Mark Sircus
http://www.naturalnews.com/023279_magnesium_cancer_calcium.html#ixzz3ZCT65Fiv
Aleksandrowicz et al in Poland conclude that inadequacy of Mg (Magnesium) and antioxidants are important risk factors in predisposing to leukemias. Other researchers found that 46% of the patients admitted to an ICU (Intensive Care Unit) in a tertiary cancer center presented hypomagnesemia.
They concluded that the incidence of hypomagnesemia in critically ill cancer patients is high. In animal studies we find that Mg deficiency has caused lymphopoietic neoplasms in young rats. A study of rats surviving Mg deficiency sufficient to cause death in convulsions during early infancy in some, and cardiorenal lesions weeks later in others, disclosed that some of survivors had thymic nodules or lymphosarcoma.
One would not normally think that Magnesium (Mg) deficiency can paradoxically increase the risk of, or protect against cancer yet we will find that just as severe dehydration or asphyxiation can cause death, magnesium deficiency can directly lead to cancer. When you consider that over 300 enzymes and ion transport require magnesium and that its role in fatty acid and phospholipid acid metabolism affects permeability and stability of membranes, we can see that magnesiumdeficiency would lead to physiological decline in cells setting the stage for cancer. Anything that weakens cell physiology will lead to the infections that surround and penetrate tumor tissues. These infections are proving to be an integral part of cancer. Magnesium deficiency poses a direct threat to the health of our cells. Without sufficient amounts, our cells calcify and rot in. Breeding grounds for yeast and fungi colonies they become, invaders all too ready to strangle our life force and kill us.
Over 300 different enzymes systems rely upon magnesium to facilitate their catalytic action, including ATP metabolism, creatine-kinase activation, adenylate-cyclase, and sodium-potassium-ATPase.
It is known that carcinogenesis induces magnesium distribution disturbances, which cause magnesium mobilization through blood cells and magnesium depletion in non-neoplastic tissues. Magnesium deficiency seems to be carcinogenic, and in case of solid tumors, a high level of supplemented magnesium inhibits carcinogenesis. Both carcinogenesis and magnesium deficiency increase the plasma membrane permeability and fluidity. Scientists have in fact found out that there is much less Mg++ binding to membrane phospholipids of cancer cells, than to normal cell membranes.