Pyrroloquinoline quinone (PQQ) – an unproved supplement
Larry H. Bernstein, MD, FCAP, Curator
LPBI
Pyrroloquinoline quinone (PQQ)
Pyrroloquinoline quinone (henceforth PQQ) is a small quinone molecule which has the ability to be a REDOX agent, capable of reducing oxidants (an antioxidant effect) and then being recycled by glutathione back into an active form. It appears to be quite stable as it can undergo several thousand cycles before being used up, and it is novel since it associates with protein structures inside the cell (some antioxidants, mostly notably carotenoids like β-carotene and Astaxanthin, are located at specific areas of a cell where they exert proportionally more antioxidant effects due to proximity; PQQ seems to do this near proteins like carotenoids do so at the cell membrane).
The aforementioned REDOX functions can alter protein function and signaling pathways, and while there is a lot of promising in vitro (outside of a living model) research on what it could do there are only a few promising results of PQQ supplementation, mostly related to either altering some signaling pathways or via its benefits to mitochondria (producing more of them and increasing their efficiency).
It is a coenzyme in bacteria (so, to bacteria, this would be something like a B-vitamin) but this role does not appear to extend to humans. Since this does not extend to humans, the designation of PQQ as a vitamin compound has fallen through and it is only considered ‘vitamin-like’ at best.
PQQ seems to modify oxidation in a cell after binding to some proteins, and this modulatory role it plays can alter the signalling processes that go on in a cell. Due to PQQ being a REDOX agent (capable of both reducing and oxidizing) it is not a pure antioxidant, but it is involved in a cyclical antioxidative cycle with an antioxidant enzyme known as glutathione
For human evidence, the limited evidence we have right now suggests a possible neuroprotective role in the aged (no research in clinical situations of neurodegeneration nor in youth) and it may have an antiinflammatory role. This limited evidence also suggests that the main claim of PQQ, an enhancement of mitochondrial function, occurs in otherwise healthy humans given PQQ supplementation.
The animal evidence that might apply to humans (using oral supplementation at doses similar to what humans use) include a radioprotective effect, possible benefits to insulin resistance, and being a growth factor when PQQ is added to the diet over a long period of time. Higher than normal oral doses in rodents seem to also enhance peripheral neurogenesis (nerve growth outside of the brain) but not necessarily in the brain.
A large amount of the evidence for a direct antioxidant role or the neurological actions related to NMDA signalling of PQQ seem to use very high concentrations in cells, due to possible transportation issues to the brain and low concentrations of PQQ found in the blood following oral ingestion.
It holds a potential to modify signalling in humans, and although the oxidation in the blood (easiest thing to measure) in mostly unaffected it also retains the potential to act as an intracellular antioxidant. The enhancement of mitochondrial function may also occur, but beyond some alterations in signalling and the mitochondrial biogenesis most other properties of PQQ are unlikely to extend to humans.
- Sources and Structure
1.1. Sources
Pyrroloquinoline quinone (PQQ) is a quinone molecule that was first identified as an enzymatic cofactor in bacteria, acting as a prosthetic group similar to how B-vitamins work in humans.[1] It is doubtful that PQQ is an enzymatic cofactor in humans, although it still appears to have affinity to proteins in the human body and can bind to them to confer biolgical effects. The proteins that seem to bind to PQQ are called quinoproteins,[2] and via modifying their actions in the body PQQ can exert biological activity.
PQQ was once thought to be a novel vitamin compound, although this view has since had doubts cast upon it and is no longer seen as accurate. Despite the lack of a vitamin role in mammals, it does appear to have growth promoting properties in rodents and may be active in humans following supplementation
PQQ naturally occurs in most foods (in miniscule amounts) although the highest levels can be found in:
- Fermented Soybeans products such as Nattō (highest estimate of 61+/-31 ng/g wet weight,[3] lower estimates in the range of 1.42 +/- 0.32ng/g[4])
- Green Soybeans (9.26+/-3.82ng/g wet weight)[3]
- Spinach (7.02 +/- 2.17ng/g fresh weight)[4]
- Rape blossoms (blossoms of the brassica napus plant at 5.44 +/- 0.8ng/g fresh weight)[4]
- Field Mustard (5.54 +/-1.50ng/g fresh weight)[4]
- Tofu (24.4+/-12.5ng/g wet weight)[3]
- Teas from Camellia Sinensis, aka Green Tea (around 30ng/g dry weight of leaves)[3] with the lower range of estimates at 0.16 +/- 0.05[4]
- Green peppers, Parsely, and Kiwi fruits (around 30ng/g wet weight or so)[3] although some estimates are lower (2.12 +/- 0.40ng/g for green peppers)[4]
- Human Breast milk at 140-180ng/mL (total PQQ and IPQ)[5]
Overall content of PQQ in foods seems to range from 0.19-7.02ng/g fresh weight in one study[4] up to 3.7-61ng/g in another,[3] low numbers may not adequately reflect total content in foods due to excluding IPQ in the measurements whereas higher levels tend to include both PQQ and IPQ.[5]
PQQ is present in a wide variety of foods, but currently the estimates of its contents are quite variable. This may be due to confusion as to whether solely PQQ should be counted or PQQ conjugates (it is not known if these confer dietary benefit). In general, the PQQ content of food products listed above is substantially lower than the content of supplemented PQQ (10-20mg) and food ingestion is unlikely to replicate the effects of supplementation due to the magnitude of difference
It should be noted that due to an affinity of PQQ to bind to amino acids and form imidazolopyrroloquinoline derivatives that the PQQ content of foods may not be the same as the total bioactive amounts of PQQ,[6] probably due to rapid association with proteins forming amino acid conjugates (Imidazolopyrroloquinoline, or IPQ).[7] Human milk, for example, contained 15% PQQ and 85% IPQ derivatives. That being said, no direct studies have been undertaken to see whether PQQ and IPQ have similar or different properties in vivo.
PQQ may form conjugates with dietary protein similar to how it is known to react with proteins in the body, but it is not known if this potential interaction with dietary protein is beneficial or negatively influences bioavailability
1.2. Structure and Properties
Pyrroloquinoline quinone is heat-stable and water soluble,[1] and appears to be stable at ambient temperatures in the form of PQQ disodium salt either as trihydrate (12.7% water[8]) or pentahydrate (22.9% water[9]). It is thought to be a relatively stable REDOX factor in vivo, and is able to carry generally around 20,000 REDOX reactions before degradation,[10][11] and when it carries out REDOX reactions by itself it gets converted into its reduced form known as pyrroloquinoline dihydroquinone (PQQH2)[12] and is replenished (back to the PQQ form) by glutathione.[12]
PQQ binds to proteins via forming a schiff base, which is a spontaneous (no enzyme required) reaction to amino acids found in the protein structures such as lysine.[13] The binding of PQQ to proteins uses the carbonyl groups (C=O),[14]including the three carboxylic groups opposite of the two ketones used in REDOX reactions.
Pyrroloquinline quinone (PQQ) is a quinone structure with three carboxylic acid groups which are used to bind to proteins, and two ketone groups which are involved in the REDOX capacities of the molecule
In some in vitro studies, combining PQQ with reducing agents (SIN-1, sodium borohydride) can form a green precipitate[15] and the reddish coloration of PQQ turns increasing brown when water content is removed.[8]
PQQ (as a powder) appears to be able to change color depending on its hydration status and oxidation status
1.3. Biological Significance
PQQ was initially thought to be synthesized via the α-amino adipic acid-Δ-semialdehyde (AASDH; also known as U26) enzyme[16] although this seems to be incorrect[17][18] since despite this protein having many PQQ binding sites[19] its mRNA levels are not negatively regulated by PQQ levels[20] which would most likely occur if the enzyme synthesized PQQ. It is known to be synthesized (in bacteria) from the amino acids L-Tyrosine and glutamate[21][22] in a process requiring a series of enzymes labelled PqqA-F where PqqA formed the peptide precursor and the other enzymes structurally modify it into active PQQ.[23]
Although mammalian synthesis is not certain, PQQ does occur normally in the mammalian body[24] and approxiamtely 100-400 nanograms of PQQ are thought to be made in humans each day;[3][25] leading some authors to claim an estimated tissue concentration of approximately 0.8−5.9ng/g in humans.[3]
Since complete deprivation from the diet of animals has been shown to hinder growth and reproductive performance,[26][7] it was initially thought that this (paired with the initial guess of endogenous synthesis via AASDH) indicated a vitamin deficiency. However, due to the definition of vitamins being one that requires a disease state to occur during deficiency[16] and no apparent dysfunction aside from impaired growth seen with PQQ deficeincy it was not classified as an essential vitamin;[17] this claim of no vitamin-like property being supported by the idea that AASDH is not actually used for PQQ sythesis in humans.[17][18]
Pyrroloquinoline quinone (PQQ) is known to occur in both the diet and in mammalian tissue, and appears to have biological activity in the body. It was initially thought to be a new vitamin, but this conclusion seems unlikely and it is more likely a bioactive non-vitamin compound.
PQQ has been investigated for being a growth factor in youth (since deprivation in rats impairs growth[26][7]), secondary to its effects at improving mitochondrial biogenesis (making more mitochondria) at seemingly effective doses of 0.2-0.3mg/kg foodstuff (in mice),[27] which is surprisingly close to the levels found in human breast milk.[5] Preliminary evidence for mitochondrial efficacy has also been noted in adult humans given 0.075-0.3mg/kg daily,[28] with the latter dose being close to the recommended 20mg serving for a 150lb adult.
PQQ is thought to be a non-vitamin growth factor, in part due to its naturally high levels in breast milk and reduced growth in rats without dietary PQQ. It may do so via beneficially influencing mitochondrial function
It is seen as a novel REDOX catalyzing agent due to its stability, which prevents most self-oxidation (seen in catechins) and polymerization (tannins).[10] A case has been made that PQQs effects are constant between species and bacteria, which aims to validate extrapolation from one species to humans.[10] The potency of PQQ and its quinoproteins in REDOX cycling appears to be approximately 100-fold greater than Vitamin C or other polyphenolic compounds, when in alkaline conditions.[25][29][30]
PQQ, after associating with proteins (not in the role of a cofactor) appears to be capable of REDOX cycling suggesting that it can have conditional prooxidant and antioxidant roles. The association with proteins suggests that it can modify their structures either directly or via modifying the levels of oxidation at the level of the protein (similar to how carotenoids such as Astaxanthin are located at the cellular membrane which localizes their effects)
- Molecular Targets
2.1. Enzymatic Cofactor
Pyrroloquinoline quinone (PQQ) was discovered in 1979 as an enymatic cofactor in bacteria;[31] preliminary evidence in pig kidneys and adrenal glands suggested a similar role in mammals.[32][33][34][35] Doubts were later cast upon the role of PQQ as a mammalian enzymatic cofactor,[36][37][38] and currently the consensus is that PQQ is unlikely to be an enzymatic cofactor in humans as it is in bacteria and plants.
Pyrroloquinoline quinone (PQQ) was first discovered as a bacterial enzymatic cofactor (being required by bacterial enzymes to function properly) and preliminary evidence suggested it could play the same role in mammals, which would make PQQ a vitamin. But further study found no quality evidence supporting this role in mammals; it is currently believed that PQQ does not act as an enzymatic cofactor in humans
2.2. REDOX Signalling
REDOX (REDuction OXidation) signalling refers to stimulation or inhibition of cellular signalling systems by molecules that can switch from an oxidized state to a reduced state, such as the well-known REDOX-acting supplements Vitamin C andAlpha-Lipoic Acid.[39] Pyrroloquinoline quinone (PQQ) may have this property as well, although its primary mode of action seems to be acting on known REDOX proteins in the cell; this is in line with its high binding affinity for some proteins, despite not acting as their coenzyme.[40][21] For example, PQQ may function as a mammalian growth factor via signal transduction modification by both oxidation and redox cycling[41], and has been shown to improve insulin signalling in mice by redox cycling.[42]
PQQ may have an indirect influence on REDOX signalling in a cell by modifying the actions of proteins, which may underlie some antioxidative (and prooxidative) changes in a cell similar to any other REDOX agent
2.3. Thioredoxin Reductase 1
PQQ has been noted to partially inhibit thioredoxin reductase 1 (TrxR1), which is an enzyme in the cytosol that reducesthioredoxin.[43] PQQ has low potency yet high affinity in binding to TrxR1 and seems to outcompete thioredoxin binding.[44] When PQQ binds to TrxR1, the enzyme’s activity is modified so it acts more on an alternate substrate known as juglone.[45] Overall, NADPH oxidase activity of TrxR1 (a measure of the activity of this enzyme) is increased in the presence of 10-50µM PQQ due to increased activity of the TrxR1-Juglone interaction.[45]
Pyrroloquinoline quinone (PQQ) binds to an antioxidant enzyme (TrxR1) and alters its function, reducing its affinity towards its normal substrate and increasing its affinity towards an alternate substrate. Overall activity of this enzyme appears to be enhanced at high concentrations of PQQ, but the effect of more physiologically realistic (nanomolar) concentrations are not known
Inhibition of TrxR1 activity is known to cause an increase in the activity of the Nrf2 protein, which acts on the nucleus (via the antioxidant response element or ARE) to increase antioxidant gene expression.[46][47][48] Since oral supplementation of PQQ appears to influence a large amount of genes under control of TrxR1-related transcripts[49] it is thought that TrxR1 inhibition by PQQ occurs in vivo.[49]
It is thought that PQQ inhibits thioredoxin reductase (TrxR1) when ingested orally, since genes that would normally be activated when TrxR1 is inhibited do seem to be activated with PQQ in rats
2.4. Glutathione Reductase
PQQ has also been shown to inhibit glutathione reductase, but despite a decreased KM towards juglone (which would increase NAPDH oxidation and enzyme activity) the Kcat was also reduced and enzyme activity remains similar with or without PQQ.[45] However, GSSG reduction with 5µM PQQ was reduced approximately 2-fold relative to control.[45]
An inhibitory effect has been noted in regards to glutathione reductase as well, although the practical significance of this particular enzyme interaction is not known
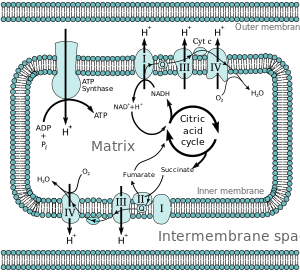
2.5. Mitochondrial Biogenesis
In rats, PQQ depletion is known to influence genetic expression (238 out of 10,000 tested genes) and dietary repletion is known to influence 847 transcripts;[49] of these, the major pathways affected include Thioredoxin and MAPK signalling but also PGC-1α, a positive regulator of mitochondrial biogenesis[50]).[49] PQQ activates PGC-1α via CREB phosphorylation[51]and appears to positively regulate mitochondrial biogenesis in vivo. It also has other possible roles in blood pressure regulation, cellular cholesterol homeostasis, energy production, and protection of mitochondrial activity, all of which are beneficially associated with increased PGC-1α activity[10][50]).
When studies are undertaken in rats comparing a PQQ deficient diet, in which the rats must rely solely on de novobiogenesis of PQQ) against PQQ sufficient diets, the PQQ supplemented diets tend to promote up to 20-30% more mitochondria in the liver (on a mass basis, as assessed by mtDNA) over the rats’ lifetime.[27][26][10][52][7][49][51] Decreased permeability of the mitochondrial membrane has also been noted without alterations in functional capacity or mitochondrial size,[26] along with the mitochondrial count per cell increasing 60% from 56.8+/-7.8 to 91+/-6.6 with 2mg/kg PQQ fed by gavage starting from 2 weeks of age in rats on a PQQ deficient diet.[26]
Pyrroloquinoline quinone (PQQ) appears to be capable to increasing the activity of PGC-1α, which then promotes mitochondrial proliferation and membrane stabilization. This occurs in rats using oral doses similar to those in humans, and occurs secondary to CREB phosphoylation; this may suggest bioenergetic benefits of supplementation, but human evidence does not yet exist
When humans supplement PQQ (0.075-0.3mg/kg for one week at a time for each dose), urinary lactate decreased by 15% along with a reduction in urinary pyruvic acid.[28] A minor reduction of fumarate was noted, but other Kreb’s cycle intermediates (Isoaconitate, Citric acid, 2-oxoglutarate, and succinate) were not altered in the urine.[28] It was hypothesized, on the assumption that urinary metabolites reflect cellular energy status, that this indicated an increase in mitochondrial efficiency.[53][54]
A nonsignificant decreasing trend in urinary 4-hydroxyphenylacetate was noted with PQQ;[28] decreases in this and other urinary metabolites tend to suggest increased β-oxidation rates.[55]
The currently lone human study using doses of PQQ commonly found in supplements suggest that supplementation may increase mitochondrial efficiency
2.6. PTP1B
Pyrroloquinoline quinone (PQQ) is known to enhance signalling of some MAPK proteins, most notably ERK1/2, to significant extents, rivalling its effects on thioredoxin and PGC-1α.[49][56] This may be secondary to oxidative changes on the PTP1B protein; the changes occur when PQQ facilitates the production of hydrogen peroxide by associating with other proteins[57]) within a cell via direct REDOX cycling.[41] Hydrogen peroxide then modifies PTP1B on Cys-215.[58] The change of Cys-215 from a sulfenic acid moiety (-SOH) into a more oxidized sulfinic acid (–SO2H) or sulfonic acid (–SO3H) causes reversible inhibition of PTP1B.[59][60]
PTP1B is a negative regulator of the insulin receptor,[61] and is also a negative regulator of the epidermal growth factor receptor (EGFR).[58] By alleviating a negative inhibition, PQQ (via H2O2) can enhance signalling through the EGFR resulting in more ERK1/2 activation.
By acting as a direct REDOX couple, PQQ can inhibit PTP1B activity via hydrogen peroxide production within a cell. This inhibition of PTP1B enhances growth factor signalling (via EGFR signalling) and can enhance insulin sensitivity in a cell (by enhancing insulin receptor signalling)
- Pharmacology
3.1. Absorption
PQQ is absorbed well in the intestines, but its absorption is highly variable; 62% of PQQ is absorbed on average in rats in a fed state, with a range from 19-89%.[62]
3.2. Serum
A single dose (0.2mg/kg) of PQQ ingested by humans in a fruit-flavored drink has a tmax of about two hours and a Cmax of approximately 9nM.[28] Doubling PQQ dose from 0.075mg/kg PQQ daily for one week to 0.15mg/kg and then 0.3mg/kg in healthy subjects increased plasma PQQ levels in a linear manner. Fasting blood levels of PQQ ranged from 2 to 14nM when measurements were taken on day four of supplementation.[28] These levels may be similar to the steady state values as they were measured after the fourth day of dosing on the morning after PQQ was ingested.[28]
Daily supplementation of pyrroloquinoline quinone (PQQ) appears to increase plasma PQQ concentrations to a steady state level of around 10nM in humans
3.3. Distribution
PQQ appears to be eliminated from mice 24 hours after ingestion except in the skin and kidneys, which retain detectable levels of PQQ following oral ingestion.[62] In the skin, it was noted that 0.3% of the ingested dose was detectable six hours following a dose and 1.3% of the oral dose was detected after 24 hours. Greater than 95% of the PQQ in the blood seems to be associated with the blood cell fraction, with less than 5% remaining in the plasma fraction.[62]
3.4. Elimination
86% of an ingested dose of PQQ in mice appears to be eliminated via the kidneys within 24 hours of oral ingestion[62] and is excreted in a manner directly correlated with serum levels in humans;[28] in humans, less than 0.1% of the ingested dose is detected as unmodified PQQ, suggesting that PQQ is highly metabolized prior to elimination.[28]
3.5. Mineral Bioaccumulation
Pyrroloquinoline quinone has been noted to bind directly to metals such as uranium. This explains the toxicity of uranium to bacteria, which depend on PQQ as a cofactor for enzymes;[63] uranium displaces a calcium ion which is required to bind PQQ to certain enzymes in bacteria.[64][65]
Pyrroloquinoline quinone has a known affinity for some minerals, but the role of PQQ in the human body in regards to minerals is not known. It is unlikely to play a role in heavy mineral elimination due to the very low serum concentrations of PQQ
- Interactions with Neurology
4.1. Glutaminergic Neurotransmission
The NMDA receptor possesses a sulfhydryl REDOX modulatory site that is susceptible to oxidation[66] where oxidation suppresses NMDA signalling and reduction enhances NMDA signalling.[67][68] PQQ (50µM) does not affect basal currents through the receptor, but it can block reducing agents from enhancing signalling[69][70] in the 5-200µM range. The reduction of signalling is thought to be due to acting on the REDOX site, since PQQ can reduce excitotoxicity but fails to protect from H2O2 (which causes toxicity independent of the NMDA receptor).[71]
This mechanism is thought to underlie protective benefits of PQQ supplementation[70] seen at low concentrations of 5µM (other mechanisms require PQQ concentrations of up to 50µM in order to become appreciable).[71]
PQQ appears to have a regulatory effect on the glutamate receptor known as NMDA, by causing some oxidation of the REDOX site and preventing excess reduction from occurring it can suppress abnormal spikes in NMDA signalling; since an excess of NMDA signalling can be toxic, the result is a neuroprotective effect. This is thought to be applicable to oral supplementation due to a low concentration being required
4.2. Neuroprotection
100µM PQQ has been noted to protect cells from glutamate-induced cytotoxicity[72][73] associated with an increase in antioxidant enzyme activity, as assessed by Nrf2 and HO-1.[72] This is thought to be downstream of Akt/PI3K and GSK-3β activation,[74] of which the former is known to occur with PQQ in the 50-100µM range in vitro.[74]
PQQ also appears to prevent an increase in JNK signalling seen with NMDA-mediated toxicity, but it is not related to the protective effects on cellular survival[74] and PI3K activation cannot fully predict the protective effects of PQQ.[72]
PQQ appears to be related to an activation of PI3K/Akt signalling, which is known to cause an induction in antioxidant enzymes via Nrf2. This is thought to underlie some of the protective benefits of PQQ on cellular structure seen in vitro, but its significance to oral supplementation is not known
Protective effects against glutamate have been noted when PQQ is directly injected into the brain in a manner that is associated with the aforementioned antioxidant effects (PI3K activation and Nrf2/HO-1 induction).[73]
Injections of PQQ into the brain are known to be neuroprotective, but it is not known if this applies to oral ingestion as well
4.3. Neurogenesis
In fibroblastic cells (L-M), incubation of PQQ disodium salt (approximately 100µg/mL) for 24 hours has resulted in a peak 40-fold increase in Nerve Growth Factor (NGF) synthesis, with minor (around 5 to 10-fold) increases at 10-20µg/mL[75][76]in a manner dependent on COX2 induction[77] and PI3K/Akt.[78] Prostaglandins D2 and E2 (from Arachidonic acid) have been reported in vitro,[77] and while they were not tested as a mandatory intermediate the former (and its metabolite prostaglandin J2) are known to promote NGF synthesis in the 6.3-25µg/mL range[77] via CHRT2[79] extending to a variety of cell lines.[80][81][82]
This increase in NGF synthesis has also been noted in isolated mouse astrocytes exceeding Alpha-Lipoic Acid (ALA) in potency, but less than ALA in c/3T3 (embyotic fibroblast) cells.[83]
When tested in vitro, PQQ appears to concentration-dependently increase NGF synthesis up to a peak efficacy at 100µg/mL. The increase noted in isolated cells appears to be quite large. Eicosanoid signalling appears to be involved in this phenomena, suggesting that PQQ works via manipulating the actions of eicosanoids
When fat-soluble derivatives were tested (PQQ trimethyl esters) at injections of 0.1-1mg/kg every other day, it was noted that peripheral sciatic nerves had enhanced regeneration;[75] injections into the periphery failed to cause an increase in NGF in the neocortex, thought to be due to poor diffusion of PQQ across the blood brain barrier due to complexation with proteins in serum.[75] A pharmaceutical modification of the PQQ enzyme (oxapyrroloquinoline; OPQ) was able to enhance brain NGF concentrations,[75] and since OPQ is known to be metabolized into PQQ in bacteria (hypothesized to occur in rodents) and is fat soluble it was thought to act as a prodrug.
When tested later, PQQ added to silicon tubes confirmed an increase in the rate of physical recovery in a mouse model of physical nerve injury with benefits seen after four weeks extending to twelve weeks.[84] This improvement was associated with an increase in well-myelinated neurons.[84]
In a spinal cord injury model, 5mg/kg PQQ injected into the spine daily for a week after injury was able to suppress the expression of iNOS after one day (a biomarker for inflammation[85][86]) and improved both locomotor performance and neuronal health (axonal density) in the area relative to control.[87] Benefits to peripheral nerve function (in a rat model of sciatic nerve injury) have been noted orally; a low dose (20mg/kg) prevented hyperalgesia from the nerve injury while only the higher dose (40mg/kg) prevented muscular atrophy and lipid peroxidation.[88]
The enhancement of neurogenesis has been noted in the periphery (tissue excluding the brain) with injections of low doses of PQQ, but an increase in neurogenesis in the brain has failed to be noted which is thought to be due to transportation issues to the brain. While there are no oral studies in rodents yet, PQQ has been noted to enhance peripheral neurogenesis following nerve injuries
4.4. Neurooxidation
As mentioned in the glutaminergic section, the oxidative effects of PQQ on the NMDA modulatory site[69][70] can ultimately cause a reduction in NMDA-induced superoxide formation in the neuron[71] at concentrations (5uM) that do not affect oxidation per se (no effect against hydrogen peroxide which circumvents the receptor).[71]
The anti-glutaminergic effects that occur at lower concentrations may also ultimately cause anti-oxidative effects by suppressing NMDA signalling, despite this mechanism being reliant on the pro-oxidant effects of PQQ
PQQ does not appear to influence the toxicity of peroxynitrate (a combination of nitric oxide and the superoxide radical), despite inhibiting its formation.[89] When using SIN-1 as a way to produce peroxynitrate and induce cell death in vitro, PQQ at 100uM abolished cell death prior to peroxynitrate formation with an EC50 of 15+/-8.4uM, yet actually potentiated pre-existing peroxynitrate toxicity (also seen with superoxide dismutase, an anti-oxidant enzyme, when catalase was not present).[15] The mechanism appears to be through sequestering the superoxide radical without significantly influencing nitric oxide, as PQQ does not appear to modify many parameters of nitric oxide or peroxynitrate per se yet potentiated a SIN-1 induction of cGMP and production of nitrate, theoretically caused by a backlog of nitric oxide that could not convert to peroxynitrate due to less free superoxide radicals.[15] Interactions with PQQ and superoxide radicals has been noted previously.[90][91]
Can prevent superoxide radical induced cell death, but does not significantly influence nitric oxide cell death per se
4.5. Epilepsy and Convulsions
NMDA receptors are involved in the pathology of seizures (as seizures are involved with excessive NMDA signalling[92][93]) and the REDOX modulatory site that PQQ is known to interact with (suppressing high levels of activity) is further implicated[94] since seizures are associated with a high level of reducing agents in the brain[95][96] which can act upon that site to promote increased NDMA signalling;[94] it is thought that PQQ could have a therapeutic role (seen with pharmaceutical NMDA antagonist[97][98]) since by its oxidative role it hinders this particular site on NMDA receptors[69][70]and PQQ is thought to not associated with side-effects from excess suppression due to only suppressing high levels of NMDA signalling but not basal levels.
When seizures occur, they are potentiated by excessive signalling through the NMDA receptors and due to this NMDA receptor antagonists (or anything that can suppress excess signalling) are thought to be therapeutic. Since PQQ has been implicated in suppressing excess NMDA signalling, it is being investigated for anti-epileptic effects
Application of 200µM PQQ to isolated neurons undergoing epileptic activity can fully abolish such activity if induced by reducing agents (no effect on epileptic activity induced by other means),[94] supporting the role PQQ plays in epilepsy via NMDA antagonism which may occurs to limited levels at concentrations as low as 5µM.[71]
In vitro evidence support a role for PQQ, but due to quite high concentrations being used (relative to what is seen in the blood) and a hypothesized low transportation to the brain it is not sure if this will occur in a living organism following oral ingestion
4.6. Hypoxia and Stroke
Pyrroloquinoline quinone (PQQ) appears to have protective effects against ischemia (assessed by infart size) when 10mg/kg is injected either 30 minutes prior to ischemia (reducing the infarct size from a 95+/-3.6% increase to 68.8+/-10.4%)[99] and is slightly less effective when injected immediately after rather than preloaded (37.6% reduction seen previously reduced to 18.5%).[99] This has been replicated elsewhere with 3-10mg/kg (70-81% protection) but not 1mg/kg was given an hour after MCAO injury.[100]
Injections of PQQ have been noted to have protective effects in rats subject to stroke, but due to high injection doses being used and the low dose being ineffective preliminary evidence does not appear to look promising for oral supplementation of PQQ in this role; oral testing, however, has not yet been conducted
4.7. Brain Injury
Injections (intraperitoneal) of PQQ in the range of 5-10mg/kg to rats for three days prior to tramautic brain injury was able to dose-dependently protect the brain from injury with the highest dose appearing to confer absolute protection (assessed by histology and cognitive behaviour post-injury).[101]
4.8. Memory and Learning
When injected into rats at 10mg/kg bodyweight, PQQ does not appear to cause overt behavioural changes in regards to sedation, activty, or heart rate[99] with no alterations in EEG readings being observed.[99]
Several morphological changes are associated with PQQ that may confer pro-cognitive effects, such as proliferation of Schwann cells secondary to PI3K/Akt activation,[78] PQQ is also able to induce production of Nerve Growth Factor (NGF)[76] secondary to COX induction;[77] increases in NGF have been observed in vivo when using trimethylesters (for permeability into the brain) with a maximal increase of 1.7-fold over baseline associated with a PQQ metabolite named oxazopyrroloquinoline.[75]
PQQ supplementation has also been associated with preventing stress-associated (oxidative stress mediated) declines in memory[102] reducing damage done by methylmercury toxicity,[103][104] and reducing memory impairment induced by a lack of oxygen;[105] at 20mg/kg bodyweight PQQ has a potency nonsignificantly different than 200mg/kg Vitamin E (as R-R-R-Alpha tocopherol) in reversing age-related memory decline in rats.[105] which, together with its neuroprotective status, assure it a position as a rehabilitative Nootropic.
Currently, one study has been conducted in humans using PQQ at 20mg daily or using PQQ at 20mg paired with 300mgCoQ10.[106] This study used the supplements once-daily at breakfast for 12 weeks in persons aged 51.7-52.3yrs with the three tests being a Verbal Memory test (seven words read aloud and then asked to recite), the Stroop Test, and the CogHealth test. The results suggested a tendency towards improvement in the Verbal memory test (nonsignificant) a significant increase in performance in the Stroop test with PQQ+CoQ10 but not PQQ in isolation, and the choice reaction and simple reactions subsets of the CogHealth test showed statistically significant improvements with PQQ and PQQ+CoQ10 but the degree of improvement was not recorded.[106]
General nootropic benefit for those with impaired cognitive function (due to age, neural damage, etc.) but does not have ample evidence to be claimed a cognition promoting nootropic in otherwise healthy. The one study conducted in humans does not claim a 50% or doubling of memory, and was not suited to answer this question
4.9. Sedation
One open-label human study conducted with 20mg PQQ for 8 weeks in 17 persons with fatigue or sleep impairing disorder noted that PQQ was able to significantly improve sleep quality, with improvements in sleep duration and quality appearing at the first testing period 4 weeks after usage while a decrease in sleep latency required 8 weeks to reach significance.[107] This study also noted improved appetite, obsession, and pain ratings that may have been secondary to improved sleep; contentness with life trended toward significance over 8 weeks but did not reach.[107]
- Cardiovascular Health
5.1. Cardiac Tissue
Protective effects have been noted in cardiac myocytes subject to ischemia, secondary to scavenging of peroxynitrate radicals, at injectible doses of 15mg/kg bodyweight 30 minutes prior to ischemia.[108][109] PQQ was studied alongside metprolol as a combiantion anti-oxidant/beta-blocker therapy, and 3mg/kg PQQ and 1mg/kg metprolol were both insignificantly different in reducing mortality (40% of control passed, 8% of PQQ and 14% of metprolol) while no deaths were recorded in combination therapy.[110] Combination was also more effective in reducing infarct size relative to either therapy in isolation, and both groups using PQQ had a reduction of creatine kinase release that was insignificantly different between groups.[110]
The combination therapy study noted increased cardiac mitochondrial respiration with PQQ but neither metprolol nor PQQ+metprolol, and respiration was further increased even in the contrl groups with no ischemia/reperfusion done.[110]
Secondary to the pro-mitochondrial effects and anti-oxidative effects during ischemia/reperfusion, PQQ appears to be cardioprotective under certain contexts
5.2. Atherosclerosis
In otherwise healthy humans supplementing PQQ at 0.075-0.3mg/kg for three weeks (increasing the dose each week), supplementation was associated with a decrease in C-reactive protein concentrations in serum (45%).[28] This study also noted that urinary trimethylamine-N-oxide (TMAO) was reduced[28] and since both C-reactive protein (CRP)[111] and TMAO[112] are thought to be biomarkers for atherosclerosis PQQ is thought to have a role.
5.3. Triglycerides
In rats fed a PQQ deficient diet relative to the same diet fed with 2mg/kg PQQ, plasma diglycerides and triglycerides (DAG and TAG) were elevated 20-50% (higher value related to triglycerides) in the PQQ deficient diet relative to 2mg/kg with no significant difference in free fatty acids,[27] which is similar to levels previously seen with this experimental protocol.[26] The elevation of triglycerides in the deficient mice does not influence the n3/n6 omega fatty acid ratios.[27]
The increase seen in triglycerides may be due to this study being conducted for a long period of time, where previous research has demonstrated that PQQ deficient diets reduce mitochondrial density by 20-30%[26] and levels of mRNA for PPAR, Fatty Acid binding protein, and Acyl CoA oxidase being significantly reduced with PQQ deficiency.[27] Additionally, higher levels of beta-hydroxybutryic acid (indicative of less beta-oxidation) were seen in PQQ deficient rats. Inducing PQQ deficiency from a sufficient state can also elevate triglyceride levels to almost two-fold the previous levels, with the trend being reversed upon acute administration of PQQ in pharmacological amounts (2mg/kg bodyweight).[49]
Appears to reduce triglycerides very potently (to a greater extent than Fish Oil, empirically) in research animals relative to a PQQ deficient diet, and this is thought to be due to increased mitochondrial β-oxidation of fatty acids
The one human study to use supplemental PQQ (0.075-0.3mg/kg for three weeks in escalating doses) failed to find any significant influence on triglyceride concentrations in serum of otherwise healthy adults consuming a standard (but uncontrolled) diet.[28] This study also noted alterations in urinary metabolites (4-Hydroxyphenylacetate and 4-Hydroxyphenylactate) suggestive of an increase in mitochondrial β-oxidation despite no apparent changes in triglycerides.[28]
First study to assess the effects of PQQ on triglycerides has failed to find an influence in otherwise healthy humans
- Interactions with Glucose Metabolism
6.1. Glucose Deposition
PQQ (500nM) has been noted to inhibit protein tyrosine phosphatase 1B (PTP1B) secondary to producing H2O2[41] (H2O2is known to inactivate PTP1B in a reversible manner[58]), and aside from PTP1B being a negative regulator of a growth factor receptor (EGFR[58]) it also negatively influences insulin receptor signalling;[61] inhibition of PTP1B, seen also withBerberine and Ursolic Acid (albeit by different mechanisms), tends to increase the activity of the insulin receptor.
Sequestering the hydrogen peroxide made from PQQ appears to block its inhibition on PTP1B.[41]
Via prooxidative changes within a cell, PQQ can produce hydrogen peroxide which then impairs PTP1B function. Since PTP1B normally suppresses signalling via the insulin receptor, the result is a compensatory increase in insulin signalling
6.2. Serum Glucose
In young rats (before sexual maturation), PQQ either at 3mg/kg in the diet or having a PQQ deficient diet does not seem to significantly affect blood glucose or insulin levels.[27] An increased glucose AUC was seen when PQQ deficient mice were subject to an oral glucose tolerance test, but no single time point was significnatly different.[27] Injections of PQQ at 4.5mg/kg bodyweight also did not significantly influence blood sugar or insulin levels in healthy rats, but was able to significantly reduce glucose AUC (by 7%) and glucose disposition in diabetic rats fed glucose and injected with PQQ, with no effect of PQQ on fasting glucose levels in rats.[27]
6.3. Insulin resistance
It has potential for alleviating fat-induced insulin resistance (characterized by a dysregulation in beta-oxidation of the TCA cycle) by increasing mitochondrial biogenesis in muscle cells, similar to exercise.[113]
At this moment in time, nothing remarkable about PQQ and glucose metabolism
- Interactions with Obesity
7.1. Metabolic Rate
When comparing a rat diet deemed sufficient in dietary pyrroloquinoline quinone (PQQ; 2mg/kg) to a diet deficient in one, the deficient diet appeared to have a decreased metabolic rate (reaching only 90% of the control rats)[27] with the difference being more prominent during the fed rather than fasted state;[27] it appears that this decreased metabolic rate did not influence the rats of lipolysis nor glycolysis as assessed by the respiratory quotient.[27]
Depleting the rat diet of PQQ appears to reduce their metabolic rates relative to a diet with adequate levels of PQQ, but no studies have investigated whether an increase in metabolic rate occurs with extra supplemental PQQ
- Bone and Joint Health
8.1. Osteoclasts
Pyrroloquinoline quinone (PQQ) has been noted to inhibit RANKL-induced osteclast formation in RAW 264.7 macrophage-like cells at a concentration of 10µM, which occurred at all stages of cell maturation.[114]
RANKL normally signals through the transcription factor NFATc1[115][116] via a particular AP-1 signalling protein that contains c-Fos and c-Jun.[117][118] PQQ inhibited c-Fos induction from RANKL,[114] but other RANKL-induced proteins (NF-kB and MAPKs) were unaffected suggesting that RANKL signalling overall was unaffected.[114]
There is a negative regulatory pathway from RANKL, where RANKL increases IFN-β production which signals via its receptor (IFNAR[119]) to activate STAT1 and JAK1 to suppress the actions of RANKL.[120][121] IFN-β was not affected by PQQ, but the receptor expression (and its targets) appeared to be increased which were thought to underlie the observed inhibitory effects seen with PQQ.[114]
PQQ appears to enhance the negative feedback mechanism controlling osteoclastogenesis (production of osteoclasts, which are negative regulators of bone mass) and via this enhancement overall osteoclast activity is hindered somewhat and this is thought to promote bone mass over time. Due to a higher than normal concentration being used, it is not sure if this occurs following oral supplementation
- Skeletal Muscle and Physical Performance
9.1. Mechanisms
One study using 0.075-0.3mg/kg PQQ supplementation daily for three weeks (increasing with dose each week) in otherwise healthy adults has noted a decrease in overall urinary amino acid levels by approximately 15%,[28] with the decrease in some (serine, asparangine, aspartic acid) being biomarkers for skeletal muscle consumption of nitrogen (via being converted into Glutamine and alanine[28][122]).
Preliminary evidence suggests that oral PQQ supplementation can influence skeletal muscle metabolism in otherwise healthy humans with standard supplemental doses, but the practical significance of this is not yet known
- Immunology and Inflammation
10.1. Mechanisms
PQQ appears to have some interactions with the immune system, as deprivation of PPQ from the diet (relative to a PQQ sufficient diet) appears to cause abnormal immune function in mice, with altered immune response after stressors.[52][7]
A study on parental (intravenous) nutrition found that the addition of 3mcg PQQ to the parental nutrition in mice was able to increase the count of CD8+ cells and lymphocytes in intestinal Peyer’s Patches, although not to the level of oral control.[123]
10.2. Macrophages
Application of PQQ to macrophages in vitro was able to prevent osteoclast differentiation at doses as low as 0.1uM (but more potency at 10uM) secondary to increasing IFN-β secretion; IFNβ is a negative regulator of osteoclast differentiation normally released after inflammation, and PQQ increases its release (and subsequent suppression), which is also demonstrated by increased levels of proteins induced by IFN-β (iNOS, STAT1, JAK1).[114] PQQ was found to phosphorylate NF-kB, p38, and IKKβ in these cells which is a pro-inflammatory response in macrophages.[114]
Practical relevance unknown
- Interactions with Oxidation
11.1. Singlet Oxygen
The reduced form of pyrroloquinoline quinone (PQQ), known as pyrroloquinoline equinol or dihydroquinone pyrroloquinoline (PQQH2) appears to be able to sequester singlet oxygen (1O2) with a potency 6.4-fold less than β-carotene as reference yet higher than that of Vitamin E (2.2-fold) and Vitamin C (6.3-fold).[12]
PQQH2 appears to be produced (via reduction) from PQQ when in a buffer in the presence of glutathione[12] and this process is known to use the semiquinone (PQQH) as an intermediate;[57] exposure to oxygen either by ambient atmosphere or by singlet oxygen readily oxidizes PQQH2 back into PQQ.[12] This suggests that glutathione is capable of recycling PQQ as an antioxidant.
PQQ and its reduced form PQQH2 appear to form a cyclical relationship where PQQH2 sequesters oxygen radicals, and glutathione reduces it back into PQQ so it may sequester more radicals; the potency of this reaction, on a molecular level, seems intermediate to β-carotene (PQQ is lesser) and Vitamin C/E (greater)
11.2. Reactive Nitrogen Species
One study assessing whether PQQ could directly sequester peroxynitrate (ONOO–) failed to find such a property of PQQ, as despite protecting cells form the toxic effects of SIN-1 (produces nitric oxide and superoxide radicals,[124] of which PQQ scavenged the superoxide radicals[15]) the toxicity of peroxynitrate directly was not protected against (in fact, it appeared to be augmented at 100-300µM PQQ).[15]
Pyrroloquinoline quinone (PQQ), even at impractically high concentrations, does not appear to direct sequester reactive nitrogen species (nitrogen based pro-oxidants) such as peroxynitrate
11.3. Lipid Peroxidation
One human study using supplemental pyrroloquinoline quinone (PQQ) and measuring serum antioxidant capacity via TBARS and TRAP values failed to find any significant influence on TRAP values but noted a decrease in TBARS (indicative of lipid peroxidation) to the degree of 0.2% when measured at peak serum PQQ values (6-12nM) seen with up to 300µg/kg supplementation;[28] this decrease in TBARS was noted to be significantly less than other dietary supplements such as procyanidins from Cocoa Extract which (560mg) can reduce TBARS by 25-35%[125] or sources of anthocyanins such as Aronia melanocarpa or Blueberry.
The decrease in serum biomarkers of lipid peroxidation that is known with PQQ supplementation is probably much too low to be indicative of anything significant
11.4. Radiation
Oral ingestion of 4mg/kg PQQ to mice (more effective than both 2mg/kg and 8mg/kg, as well as the reference drug of 10mg/kg nilestriol[126]) appears to reduce death from gamma irradiation when given an hour before and again seven days after irradiation; damage to select cells tested (white blood cells, reticulocytes, bone marrow cells) was also reduced with 4mg/kg PQQ supplementation to mice.[126]
Oral ingestion of PQQ (estimated human equivalent of 0.32mg/kg) appears to be able to protect mice from gamma irradiation to a respectable degree
- Peripheral Organ Systems
12.1. Liver
An intraperitoneal injection of pyrroloquinoline quinone (PQQ) to rats at 5mg/kg twice before CCl4 liver toxicity appeared to exert protective effects;[127] when tested in vitro, PQQ showed protective effects in isolated liver cells with most potency at 3µM.[127]
12.2. Intestines
Due to the involvement of pyrroloquinoline quinone (PQQ) in bacteria (from where it was discovered in 1979[31]) and the involvement of quinoproteins in the fermentation process [128] (which PQQ associates with) and the above higher count of PQQ recorded in fermented foods; it is hypothesized that fermentation may increase PQQ content. Interestingly, common strains of bacteria in the human intestinal tract do not appear to synthesis much PQQ[129][130] and in antibiotic fed mice (lacking intestinal microflora) it seems that dietary intake is the major determinent of bodily PQQ levels.[130]
Pyrroloquinoline quinone was thought to be synthesized by intestinal bacteria due to its discovery being that of a bacterial cofactor, but preliminary evidence does not support the intestinal microflora as a major producer of PQQ in the body
12.3. Kidney
Pyrroloquinoline quinone (PQQ) was once implicated in being an enzymatic cofacter for diamine oxidase (pig kidney)[32][33]and DOPA decarboxylase (pig kidney)[34] (as well as dopamine β-hydroxylase, albeit in the renal medulla[35]), although it is generally accepted to not be a significant component of eukaryotic enzymes in vivo (in the role of a cofactor) like it is in bacterial and plant enzymes.[36][37][38] Still, it is detectable in the kidney after oral ingestion in the rat[62] and elimination of PQQ is primarily via the urine[62] suggesting it may still play a role independent of being an enzymatic cofactor.
PQQ is not thought to play a role as a cofactor of enzymes in the kidneys like initially thought, but due to being eliminated by the kidneys and accumulating in them following oral ingestion in the rat it is still thought to play a role (perhaps as a REDOX couplet, like other mechanisms)
- Interactions with Cancer
13.1. Leukemia
PQQ has been shown to be cytotoxic to U937 leukemia cells, but not NIH3T3 nor L929 cells, in a dose-dependent manner.[131] Catalase treatment neutralized these effects, as they appear to be secondary to hydrogen peroxide production in cells which PQQ has been repeatedly shown to induce.[132] Superoxide dismutase had no effect on PQQ cytotoxicity, while glutathione or N-AcetylCysteine increased cytotoxicity 2-5fold without affecting the cells on their own (and thus working via PQQ by increasing H2O2 production form PQQ 1.5-2fold).[131] PQQ by itself decreased intracellular glutathione levels, and when glutathione was depleted (via BSO, an inhibitor of γ-glutamylcysteine synthetase) the apoptosis of cells morphed into necrosis, and this necrosis was still mediated by H2O2 due to being inhibited by catalase.[131]
Induces cell death via H2O2, and uses glutathoine to produce even more H2O2 to augment its efficacy. A depletion of glutathione induces necrosis
13.2. Melanoma
PQQ has been implicated in reducing melanogenic (melanin producing) protein expression in cultured B16 cells, where it can inhibit tyrosinase expression and reduce gene activity[133] and can prevent stimulation of tryosinase mRNA by alpha-melanocyte stimulating hormone.[134]
- Interactions with Medical Conditions
14.1. Parkinson’s Disease
Parkinson’s disease is known to be associated with what are known as Lewy Bodies (irregular cytoplasmic inclusions[135][136]) which are comprised of a molecule known as α-synuclein[137] which is known to damange dopaminergic neurons and is involved in the pathology of Parkinson’s disease when it aggregates.[138][139] It is involved in normal physiological function (as a chaperone) when unaggregated,[140] so the process of α-synuclein aggregation itself is seen as pathological.
Pyrroloquinline quinone (PQQ) is known to bind to some of these α-synuclein peptides directly via forming a schiff basewith the lysine amino acids in the peptides[13] similar to both EGCG (Green Tea Catechins) and baicalein (skullcap)[13]although baicalein seems relatively more potent.[141] This direct binding also reduces formation of truncated α-synuclein[142] (which accelerate the formation of larger aggregates[143]) and the larger protein aggregates themselves[13] by around 14.8-50% at 280µM.[142] This may indirectly reduce the cytotoxicity that is seen with large aggregates,[13] although PQQ seems to be capable of reducing cytotoxicity from pre-formed aggregates independent of the aforementioned binding.[142]
Protein aggregates tend to occur normally in the brain, and their aggregation is accelerated and seem to be central to the development of Parkinson’s Disease. PQQ appears to physically bind to these proteins in vitro to prevent the aggregation, but it occurs at a very high concentration and it does not seem likely to occur with respectable potency following oral supplementation
6-hydroxydopamine (6-OHDA), a metabolite of dopamine which is known to cause oxidative damage to dopaminergic neurons and detected at higher levels in persons with Parkinson’s,[144] may have its toxicity attenuated with coincubation of PQQ.[145] Oxidative neurotoxicity and DNA fragmentation induced by 6-hydroxydopamine was reduced in a concentration dependent manner with concentrations of 300nM showing efficacy, yet this protective effect was not seen with Vitamin C or Vitamin E, two other anti-oxidants tested at concentrations up to 100µM.[145]
Elsewhere in isolated neurons, the protein DJ-1 (plays roles in oxidative protection[146][147] and mutations in it underly some genetic cases of early onset Parkinson’s Disease[148]) does not have its expression altered by PQQ[149] but 15µM PQQ appeared to preserve cell survival in the presence of oxidants by preserving the actions of DJ-1;[149] excessive oxidation of DJ-1 at C106 ablates its antioxidant potential[150] and PQQ appears to prevent this from occurring despite no direct binding.[149]
There may be some protective effects at the level of dopaminergic neurons with PQQ that is not related to preventing the formation of protein aggregation, and although this happens at a much more respectable (lower) concentration it is still uncertain if this applies to oral supplementation of PQQ
14.2. Alzheimer’s Disease
Pyrroloquinline quinone appears to inhibit the formation of amyloid fibrils (Aβ1-42; full inhibition at 70μM PQQ[151]), and although it can also bind to α-synuclein this binding does not indirectly inhibit Aβ1-42 aggregation.[13]
and to reduce the cytotoxicity of these fibrils on neuronal cells.[152]
- Nutrient-Nutrient Interactions
15.1. Glutathione
PQQ has been shown to be cytotoxic to U937 leukemia cells, but not NIH3T3 nor L929 cells (but was observed in EL-4), in a dose-dependent manner with most significance at 20-50uM.[131] Catalase treatment neutralized these effects, as they appear to be secondary to hydrogen peroxide production in cells which PQQ has been repeatedly shown to induce.[132]Superoxide dismutase had no effect on PQQ cytotoxicity, while glutathione or N-AcetylCysteine increased cytotoxicity 2-5fold without affecting the cells on their own (and thus working via PQQ by increasing H2O2 production form PQQ 1.5-2fold).[131] PQQ by itself decreased intracellular glutathione levels, and when glutathione was depleted (via BSO, an inhibitor of γ-glutamylcysteine synthetase) the apoptosis of cells morphed into necrosis, and this necrosis was still mediated by H2O2 due to being inhibited by catalase.[131]
Glutathione can be increased by cysteine containing supplements including N-AcetylCysteine or Whey Protein
In cancer cells susceptible to PQQ’s induction of H2O2, adding glutathione to the cell by consuming Cysteine-containing supplements can augment the efficacy of PQQ
- Safety and Toxicology
16.1. General
PQQ has been associated with renal tubule inflammation at the dose of 11-12mg/kg bodyweight in rats after injections, and some symptoms of both renal and hepatic toxicity are seen with injections of 20mg/kg in rats.[110][153] Acute death from PQQ injections between doses of 500-1000mg/kg bodyweight has been recorded in rats.[10][153]
11-12mg/kg bodyweight, based on rudimentary body surface area conversions, is approximately 120-131mg/PQQ daily (although injections) if extrapolated to humans.
One human study using 20mg PQQ alone or in combination with 300mg CoQ10 noted that there were no toxicological signs or symptoms associated with treatment over a 12 week period,[106] and consumption of up to 0.3mg/kg PQQ (around 20mg for a 150lb person) for one week has been noted to be safe.[28]
Chronic toxicity to the kidneys and liver may be achieved at a relatively low dose, although acute death requires a very high and unpractical dose. Until more evidence surfaces, it would be prudent to avoid superloading
16.2. Genotoxicity
In an Ames test (TA1535, TA1537, TA98, and TA100 strains), 10-5000μg PQQ per plate (without metabolic activation) and 156-5000μg per plate (with activation) has failed to show appreciable genotoxic effects.[154]
In lung fibroblasts derived from chinese hamsters, 12.5-400μg/mL (no metabolic activation) and 117.2-3750μg/mL (with activation; highest concentration being 10mM) and the latter concentration in isolated lymphocytes failed to exert appreciable genotoxic effects as assessed by structural abberations and polyploidy.[154]
The aforementioned disodium salt of PQQ has failed to acutely exert genotoxic effects in mice (up to 2,000mg/kg) as assessed by a micronucleus assay and in bone marrow erythrocytes.[154]
No genotoxiticity has been noted with the disodium salt of PQQ
Scientific Support & Reference Citations
References
- HAUGE JG GLUCOSE DEHYDROGENASE OF BACTERIUM ANITRATUM: AN ENZYME WITH A NOVEL PROSTHETIC GROUP . J Biol Chem. (1964)
- Anthony C Pyrroloquinoline quinone (PQQ) and quinoprotein enzymes . Antioxid Redox Signal. (2001)
- Kumazawa T1, et al Levels of pyrroloquinoline quinone in various foods . Biochem J. (1995)
- Noji N, et al Simple and sensitive method for pyrroloquinoline quinone (PQQ) analysis in various foods using liquid chromatography/electrospray-ionization tandem mass spectrometry . J Agric Food Chem. (2007)
- Mitchell AE, et al Characterization of pyrroloquinoline quinone amino acid derivatives by electrospray ionization mass spectrometry and detection in human milk . Anal Biochem. (1999)
- Mitchell AE, et al Characterization of Pyrroloquinoline Quinone Amino Acid Derivatives by Electrospray Ionization Mass Spectrometry and Detection in Human Milk . Anal Biochem. (1999)
- Steinberg F, et al Pyrroloquinoline quinone improves growth and reproductive performance in mice fed chemically defined diets . Exp Biol Med (Maywood). (2003)
- Ikemoto K, Sakamoto H, Nakano M Crystal structure and characterization of pyrroloquinoline quinone disodium trihydrate . Chem Cent J. (2012)
- Ishida T, et al Molecular and crystal structure of PQQ (methoxatin), a novel coenzyme of quinoproteins: extensive stacking character and metal ion interaction . J Am Chem Soc. (1989)
- Rucker R, Chowanadisai W, Nakano M Potential physiological importance of pyrroloquinoline quinone . Altern Med Rev. (2009)
- Kim J, et al Pyrroloquinoline quinone inhibits the fibrillation of amyloid proteins . Prion. (2010)
- Mukai K, Ouchi A, Nakano M Kinetic study of the quenching reaction of singlet oxygen by Pyrroloquinolinequinol (PQQH(2), a reduced form of Pyrroloquinolinequinone) in micellar solution .J Agric Food Chem. (2011)
- Yoshida W, et al Partial peptide of α-synuclein modified with small-molecule inhibitors specifically inhibits amyloid fibrillation of α-synuclein . Int J Mol Sci. (2013)
- Adachi O, et al Adduct formation of pyrroloquinoline quinone and amino acid . Biofactors. (1988)
- Zhang Y, Rosenberg PA The essential nutrient pyrroloquinoline quinone may act as a neuroprotectant by suppressing peroxynitrite formation . Eur J Neurosci. (2002)
- Kasahara T, Kato T Nutritional biochemistry: A new redox-cofactor vitamin for mammals . Nature. (2003)
- Felton LM, Anthony C Biochemistry: role of PQQ as a mammalian enzyme cofactor . Nature. (2005)
- Rucker R, et al Biochemistry: is pyrroloquinoline quinone a vitamin . Nature. (2005)
- Wang L, et al Cloning and characterization of a novel human homolog* of mouse U26, a putative PQQ-dependent AAS dehydrogenase . Mol Biol Rep. (2005)
- Bauerly KA, et al Pyrroloquinoline quinone nutritional status alters lysine metabolism and modulates mitochondrial DNA content in the mouse and rat . Biochim Biophys Acta. (2006)
- Stites TE, Mitchell AE, Rucker RB Physiological importance of quinoenzymes and the O-quinone family of cofactors . J Nutr. (2000)
- Goodwin PM, Anthony C The Biochemistry, Physiology and Genetics of PQQ and PQQ-containing Enzymes . Adv Microb Physiol. (1998)
- Puehringer S, Metlitzky M, Schwarzenbacher R The pyrroloquinoline quinone biosynthesis pathway revisited: a structural approach . BMC Biochem. (2008)
- Flückiger R, et al Characterization of the glycine-dependent redox-cycling activity in animal fluids and tissues using specific inhibitors and activators: evidence for presence of PQQ .Biochem Biophys Res Commun. (1993)
- Flückiger R, Paz MA, Gallop PM Redox-cycling detection of dialyzable pyrroloquinoline quinone and quinoproteins . Methods Enzymol. (1995)
- Stites T, et al Pyrroloquinoline quinone modulates mitochondrial quantity and function in mice . J Nutr. (2006)
- Bauerly K, et al Altering pyrroloquinoline quinone nutritional status modulates mitochondrial, lipid, and energy metabolism in rats . PLoS One. (2011)
- Harris CB, et al Dietary pyrroloquinoline quinone (PQQ) alters indicators of inflammation and mitochondrial-related metabolism in human subjects . J Nutr Biochem. (2013)
- Paz MA, et al The catalysis of redox cycling by pyrroloquinoline quinone (PQQ), PQQ derivatives, and isomers and the specificity of inhibitors . Anal Biochem. (1996)
- Paz MA, et al Specific detection of quinoproteins by redox-cycling staining . J Biol Chem. (1991)
- Salisbury SA, et al A novel coenzyme from bacterial primary alcohol dehydrogenase . Nature. (1979)
- Dooley DM, et al The generation of an organic free radical in substrate-reduced pig kidney diamine oxidase-cyanide . FEBS Lett. (1987)
- van der Meer RA, et al Primary structure of a pyrroloquinoline quinone (PQQ) containing peptide isolated from porcine kidney diamine oxidase . Biochem Biophys Res Commun. (1989)
- Groen BW, van der Meer RA, Duine JA Evidence for PQQ as cofactor in 3,4-dihydroxyphenylalanine (dopa) decarboxylase of pig kidney . FEBS Lett. (1988)
- van der Meer RA, Jongejan JA, Duine JA Dopamine beta-hydroxylase from bovine adrenal medulla contains covalently-bound pyrroloquinoline quinone . FEBS Lett. (1988)
- Robertson JG, et al Spectral studies of bovine dopamine beta-hydroxylase. Absence of covalently bound pyrroloquinoline quinone . J Biol Chem. (1989)
- Duine JA PQQ in plants (and animals) . Trends Biochem Sci. (1991)
- Klinman JP, et al Status of the cofactor identity in copper oxidative enzymes . FEBS Lett. (1991)
- Kamata H, Hirata H Redox regulation of cellular signalling . Cell Signal. (1999)
- Ouchi A1, et al Kinetic study of the antioxidant activity of pyrroloquinolinequinol (PQQH(2), a reduced form of pyrroloquinolinequinone) in micellar solution . J Agric Food Chem. (2009)
- Kimura K, et al Pyrroloquinoline quinone stimulates epithelial cell proliferation by activating epidermal growth factor receptor through redox cycling . Free Radic Biol Med. (2012)
- Takada M, et al Pyrroloquinoline quinone, a novel protein tyrosine phosphatase 1B inhibitor, activates insulin signaling in C2C12 myotubes and improves impaired glucose tolerance in diabetic KK-A(y) mice . Biochem Biophys Res Commun. (2012)
- Arnér ES, Holmgren A Physiological functions of thioredoxin and thioredoxin reductase . Eur J Biochem. (2000)
- Xu J, Arnér ES Pyrroloquinoline quinone modulates the kinetic parameters of the mammalian selenoprotein thioredoxin reductase 1 and is an inhibitor of glutathione reductase . Biochem Pharmacol. (2012)
- Cenas N, et al Interactions of nitroaromatic compounds with the mammalian selenoprotein thioredoxin reductase and the relation to induction of apoptosis in human cancer cells . J Biol Chem. (2006)
- Brigelius-Flohé R, et al The yin and yang of nrf2-regulated selenoproteins in carcinogenesis . Int J Cell Biol. (2012)
- Locy ML, et al Thioredoxin reductase inhibition elicits nrf2-mediated responses in clara cells: implications for oxidant-induced lung injury . Antioxid Redox Signal. (2012)
- Suvorova ES, et al Cytoprotective Nrf2 pathway is induced in chronically txnrd 1-deficient hepatocytes . PLoS One. (2009)
- Tchaparian E, et al Identification of transcriptional networks responding to pyrroloquinoline quinone dietary supplementation and their influence on thioredoxin expression, and the JAK/STAT and MAPK pathways . Biochem J. (2010)
- Puigserver P Tissue-specific regulation of metabolic pathways through the transcriptional coactivator PGC1-alpha . Int J Obes (Lond). (2005)
- Chowanadisai W, et al Pyrroloquinoline quinone stimulates mitochondrial biogenesis through cAMP response element-binding protein phosphorylation and increased PGC-1alpha expression . J Biol Chem. (2010)
- Steinberg FM, Gershwin ME, Rucker RB Dietary pyrroloquinoline quinone: growth and immune response in BALB/c mice . J Nutr. (1994)
- Wasserman K, et al Lactate, pyruvate, and lactate-to-pyruvate ratio during exercise and recovery . J Appl Physiol (1985). (1985)
- Guan M, et al Systemic perturbations of key metabolites in diabetic rats during the evolution of diabetes studied by urine metabonomics . PLoS One. (2013)
- Terán-García M, Ibarra I, Velázquez A Urinary organic acids in infant malnutrition . Pediatr Res. (1998)
- Kumazawa T, et al Activation of Ras signaling pathways by pyrroloquinoline quinone in NIH3T3 mouse fibroblasts . Int J Mol Med. (2007)
- Ishii T, et al Pro-oxidant action of pyrroloquinoline quinone: characterization of protein oxidative modifications . Biosci Biotechnol Biochem. (2010)
- Lee SR, et al Reversible inactivation of protein-tyrosine phosphatase 1B in A431 cells stimulated with epidermal growth factor . J Biol Chem. (1998)
- Rhee SG, et al Hydrogen peroxide: a key messenger that modulates protein phosphorylation through cysteine oxidation . Sci STKE. (2000)
- Huyer G, et al Mechanism of inhibition of protein-tyrosine phosphatases by vanadate and pervanadate . J Biol Chem. (1997)
- Goldstein BJ Protein-tyrosine phosphatase 1B (PTP1B): a novel therapeutic target for type 2 diabetes mellitus, obesity and related states of insulin resistance . Curr Drug Targets Immune Endocr Metabol Disord. (2001)
- Smidt CR, et al Intestinal absorption and tissue distribution of {14C}pyrroloquinoline quinone in mice . Proc Soc Exp Biol Med. (1991)
- VanEngelen MR, et al Uranium exerts acute toxicity by binding to pyrroloquinoline quinone cofactor . Environ Sci Technol. (2011)
- Schürer G Is the calcium-ion catalysis of biological reoxidation of reduced PQQ purely electrostatic? . Chem Commun. (1998)
- Zheng YJ, Bruice TC Conformation of coenzyme pyrroloquinoline quinone and role of Ca2+ in the catalytic mechanism of quinoprotein methanol dehydrogenase . Proc Natl Acad Sci U S A. (1997)
- Aizenman E, Hartnett KA, Reynolds IJ Oxygen free radicals regulate NMDA receptor function via a redox modulatory site . Neuron. (1990)
- Aizenman E, Lipton SA, Loring RH Selective modulation of NMDA responses by reduction and oxidation . Neuron. (1989)
- Tang LH, Aizenman E The modulation of N-methyl-D-aspartate receptors by redox and alkylating reagents in rat cortical neurones in vitro . J Physiol. (1993)
- Aizenman E, et al Further evidence that pyrroloquinoline quinone interacts with the N-methyl-D-aspartate receptor redox site in rat cortical neurons in vitro . Neurosci Lett. (1994)
- Aizenman E, et al Interaction of the putative essential nutrient pyrroloquinoline quinone with the N-methyl-D-aspartate receptor redox modulatory site . J Neurosci. (1992)
- Scanlon JM, Aizenman E, Reynolds IJ Effects of pyrroloquinoline quinone on glutamate-induced production of reactive oxygen species in neurons . Eur J Pharmacol. (1997)
- Zhang Q, et al Pyrroloquinoline quinone rescues hippocampal neurons from glutamate-induced cell death through activation of Nrf2 and up-regulation of antioxidant genes . Genet Mol Res. (2012)
- Zhang Q, et al Pyrroloquinoline quinine protects rat brain cortex against acute glutamate-induced neurotoxicity . Neurochem Res. (2013)
- Zhang Q, et al The neuroprotective action of pyrroloquinoline quinone against glutamate-induced apoptosis in hippocampal neurons is mediated through the activation of PI3K/Akt pathway. Toxicol Appl Pharmacol. (2011)
- Yamaguchi K, et al Stimulation of nerve growth factor production by pyrroloquinoline quinone and its derivatives in vitro and in vivo . Biosci Biotechnol Biochem. (1993)
- Urakami T, et al Synthesis of esters of coenzyme PQQ and IPQ, and stimulation of nerve growth factor production . Biofactors. (1995-1996)
- Yamaguchi K, et al Cyclooxygenase induction is essential for NGF synthesis enhancement by NGF inducers in L-M cells . Biosci Biotechnol Biochem. (1996)
- He B, Liu SQ, Li HH The roles of PI3K/Akt pathway in proliferation of Schwann cells promoted by pyrroloquinoline quinone . Zhonghua Zheng Xing Wai Ke Za Zhi. (2010)
- Hatanaka M, et al 15d-prostaglandin J2 enhancement of nerve growth factor-induced neurite outgrowth is blocked by the chemoattractant receptor- homologous molecule expressed on T-helper type 2 cells (CRTH2) antagonist CAY10471 in PC12 cells . J Pharmacol Sci. (2010)
- Bulló M, Peeraully MR, Trayhurn P Stimulation of NGF expression and secretion in 3T3-L1 adipocytes by prostaglandins PGD2, PGJ2, and Delta12-PGJ2 . Am J Physiol Endocrinol Metab. (2005)
- Toyomoto M, et al Prostaglandins are powerful inducers of NGF and BDNF production in mouse astrocyte cultures . FEBS Lett. (2004)
- Satoh T, et al Prostaglandin J2 and its metabolites promote neurite outgrowth induced by nerve growth factor in PC12 cells . Biochem Biophys Res Commun. (1999)
- Murase K, et al Stimulation of nerve growth factor synthesis/secretion in mouse astroglial cells by coenzymes . Biochem Mol Biol Int. (1993)
- Liu S, et al Enhanced rat sciatic nerve regeneration through silicon tubes filled with pyrroloquinoline quinone . Microsurgery. (2005)
- Satake K, et al Nitric oxide via macrophage iNOS induces apoptosis following traumatic spinal cord injury . Brain Res Mol Brain Res. (2000)
- Xu J, et al iNOS and nitrotyrosine expression after spinal cord injury . J Neurotrauma. (2001)
- Hirakawa A, et al Pyrroloquinoline quinone attenuates iNOS gene expression in the injured spinal cord . Biochem Biophys Res Commun. (2009)
- Gong D, et al Effect of pyrroloquinoline quinone on neuropathic pain following chronic constriction injury of the sciatic nerve in rats . Eur J Pharmacol. (2012)
- Zhang Y, Rosenberg PA The essential nutrient pyrroloquinoline quinone may act as a neuroprotectant by suppressing peroxynitrite formation . Eur J Neurosci. (2002)
- Urakami T, et al Synthesis of monoesters of pyrroloquinoline quinone and imidazopyrroloquinoline, and radical scavenging activities using electron spin resonance in vitro and pharmacological activity in vivo . J Nutr Sci Vitaminol (Tokyo). (1997)
- Gallop PM, et al Is the antioxidant, anti-inflammatory putative new vitamin, PQQ, involved with nitric oxide in bone metabolism . Connect Tissue Res. (1993)
- Chapman AG Glutamate receptors in epilepsy . Prog Brain Res. (1998)
- Bradford HF Glutamate, GABA and epilepsy . Prog Neurobiol. (1995)
- Sanchez RM, et al Novel role for the NMDA receptor redox modulatory site in the pathophysiology of seizures . J Neurosci. (2000)
- Hoshi Y, Tamura M Dynamic changes in cerebral oxygenation in chemically induced seizures in rats: study by near-infrared spectrophotometry . Brain Res. (1993)
- Fujikawa DG, et al Generalized seizures deplete brain energy reserves in normoxemic newborn monkeys . Brain Res. (1988)
- Dannhardt G, Kohl BK The glycine site on the NMDA receptor: structure-activity relationships and possible therapeutic applications . Curr Med Chem. (1998)
- Kornhuber J, Weller M Psychotogenicity and N-methyl-D-aspartate receptor antagonism: implications for neuroprotective pharmacotherapy . Biol Psychiatry. (1997)
- Jensen FE, et al The putative essential nutrient pyrroloquinoline quinone is neuroprotective in a rodent model of hypoxic/ischemic brain injury . Neuroscience. (1994)
- Zhang Y, Feustel PJ, Kimelberg HK Neuroprotection by pyrroloquinoline quinone (PQQ) in reversible middle cerebral artery occlusion in the adult rat . Brain Res. (2006)
- Zhang L, et al The neuroprotective effect of pyrroloquinoline quinone on traumatic brain injury . J Neurotrauma. (2012)
- Ohwada K, et al Pyrroloquinoline Quinone (PQQ) Prevents Cognitive Deficit Caused by Oxidative Stress in Rats . J Clin Biochem Nutr. (2008)
- Zhang P, et al Protection of pyrroloquinoline quinone against methylmercury-induced neurotoxicity via reducing oxidative stress . Free Radic Res. (2009)
- Zhang P, et al In vitro protective effects of pyrroloquinoline quinone on methylmercury-induced neurotoxicity . Environ Toxicol Pharmacol. (2009)
- Takatsu H, et al Effect of vitamin E on learning and memory deficit in aged rats . J Nutr Sci Vitaminol (Tokyo). (2009)
- Nakano M, et al Effect of Pyrroloquinoline Quinone (PQQ) on mental status of Middle-Aged and Elderly Persons . Food Style. (2009)
- Nakano M, et al Effects of Oral Supplementation with Pyrroloquinoline Quinone on Stress, Fatigue, and Sleep . Funct Foods Health Dis. (2012)
- Zhu BQ, et al Pyrroloquinoline quinone (PQQ) decreases myocardial infarct size and improves cardiac function in rat models of ischemia and ischemia/reperfusion . Cardiovasc Drugs Ther. (2004)
- Tao R, et al Pyrroloquinoline quinone preserves mitochondrial function and prevents oxidative injury in adult rat cardiac myocytes . Biochem Biophys Res Commun. (2007)
- Zhu BQ, et al Comparison of pyrroloquinoline quinone and/or metoprolol on myocardial infarct size and mitochondrial damage in a rat model of ischemia/reperfusion injury . J Cardiovasc Pharmacol Ther. (2006)
- Singh SK, et al The connection between C-reactive protein and atherosclerosis . Ann Med. (2008)
- Stella C, et al Susceptibility of human metabolic phenotypes to dietary modulation . J Proteome Res. (2006)
- Muoio DM, Koves TR Skeletal muscle adaptation to fatty acid depends on coordinated actions of the PPARs and PGC1 alpha: implications for metabolic disease . Appl Physiol Nutr Metab. (2007)
- Odkhuu E, et al Inhibition of receptor activator of nuclear factor-κB ligand (RANKL)-induced osteoclast formation by pyrroloquinoline quinine (PQQ) . Immunol Lett. (2012)
- Zhao Q, et al NFATc1: functions in osteoclasts . Int J Biochem Cell Biol. (2010)
- Takayanagi H, et al Induction and activation of the transcription factor NFATc1 (NFAT2) integrate RANKL signaling in terminal differentiation of osteoclasts . Dev Cell. (2002)
- Ikeda F, et al Critical roles of c-Jun signaling in regulation of NFAT family and RANKL-regulated osteoclast differentiation . J Clin Invest. (2004)
- Wagner EF, Eferl R Fos/AP-1 proteins in bone and the immune system . Immunol Rev. (2005)
- Abraham AK, et al Mechanisms of interferon-beta effects on bone homeostasis . Biochem Pharmacol. (2009)
- Takayanagi H, et al RANKL maintains bone homeostasis through c-Fos-dependent induction of interferon-beta . Nature. (2002)
- Takayanagi H, et al Interplay between interferon and other cytokine systems in bone metabolism . Immunol Rev. (2005)
- Tremblay F, et al Role of dietary proteins and amino acids in the pathogenesis of insulin resistance . Annu Rev Nutr. (2007)
- Omata J, et al Influence of adding pyrroloquinoline quinone to parenteral nutrition on gut-associated lymphoid tissue . JPEN J Parenter Enteral Nutr. (2011)
- Hogg N1, et al Production of hydroxyl radicals from the simultaneous generation of superoxide and nitric oxide . Biochem J. (1992)
- Rein D, et al Epicatechin in human plasma: in vivo determination and effect of chocolate consumption on plasma oxidation status . J Nutr. (2000)
- Xiong XH, et al Production and radioprotective effects of pyrroloquinoline quinone . Int J Mol Sci. (2011)
- Tsuchida T, et al The protective effect of pyrroloquinoline quinone and its derivatives against carbon tetrachloride-induced liver injury of rats . J Gastroenterol Hepatol. (1993)
- Adachi O, et al New developments in oxidative fermentation . Appl Microbiol Biotechnol. (2003)
- Matsushita K, et al Escherichia coli is unable to produce pyrroloquinoline quinone (PQQ) . Microbiology. (1997)
- Smidt CR, et al Does the intestinal microflora synthesize pyrroloquinoline quinone . Biofactors. (1991)
- Shankar BS, et al Role of glutathione in augmenting the anticancer activity of pyrroloquinoline quinone (PQQ) . Redox Rep. (2010)
- He K, et al Antioxidant and pro-oxidant properties of pyrroloquinoline quinone (PQQ): implications for its function in biological systems . Biochem Pharmacol. (2003)
- Sato K, Toriyama M Effect of pyrroloquinoline quinone (PQQ) on melanogenic protein expression in murine B16 melanoma . J Dermatol Sci. (2009)
- Kosano H, et al Pyrroloquinoline quinone (PQQ) inhibits the expression of tyrosinase mRNA by alpha-melanocyte stimulating hormone in murine B16 melanoma cells . Life Sci. (1995)
- Irizarry MC, et al Nigral and cortical Lewy bodies and dystrophic nigral neurites in Parkinson’s disease and cortical Lewy body disease contain alpha-synuclein immunoreactivity . J Neuropathol Exp Neurol. (1998)
- Wakabayashi K, et al NACP, a presynaptic protein, immunoreactivity in Lewy bodies in Parkinson’s disease . Neurosci Lett. (1997)
- Spillantini MG, et al Alpha-synuclein in Lewy bodies . Nature. (1997)
- Sode K, et al Engineered alpha-synuclein prevents wild type and familial Parkin variant fibril formation . Biochem Biophys Res Commun. (2005)
- Moussa CE, et al Differential cytotoxicity of human wild type and mutant alpha-synuclein in human neuroblastoma SH-SY5Y cells in the presence of dopamine . Biochemistry. (2004)
- Kim TD, Paik SR, Yang CH Structural and functional implications of C-terminal regions of alpha-synuclein . Biochemistry. (2002)
- Zhu M, et al The flavonoid baicalein inhibits fibrillation of alpha-synuclein and disaggregates existing fibrils . J Biol Chem. (2004)
- Kim J, et al The inhibitory effect of pyrroloquinoline quinone on the amyloid formation and cytotoxicity of truncated alpha-synuclein . Mol Neurodegener. (2010)
- Liu CW, et al A precipitating role for truncated alpha-synuclein and the proteasome in alpha-synuclein aggregation: implications for pathogenesis of Parkinson disease . J Biol Chem. (2005)
- Andrew R, et al The determination of hydroxydopamines and other trace amines in the urine of parkinsonian patients and normal controls . Neurochem Res. (1993)
- Hara H, Hiramatsu H, Adachi T Pyrroloquinoline quinone is a potent neuroprotective nutrient against 6-hydroxydopamine-induced neurotoxicity . Neurochem Res. (2007)
- Yokota T, et al Down regulation of DJ-1 enhances cell death by oxidative stress, ER stress, and proteasome inhibition . Biochem Biophys Res Commun. (2003)
- Lev N, et al Oxidative insults induce DJ-1 upregulation and redistribution: implications for neuroprotection . Neurotoxicology. (2008)
- Bonifati V, et al Mutations in the DJ-1 gene associated with autosomal recessive early-onset parkinsonism . Science. (2003)
- Nunome K, et al Pyrroloquinoline quinone prevents oxidative stress-induced neuronal death probably through changes in oxidative status of DJ-1 . Biol Pharm Bull. (2008)
- Zhou W, et al The oxidation state of DJ-1 regulates its chaperone activity toward alpha-synuclein . J Mol Biol. (2006)
- Kobayashi M, et al Pyrroloquinoline quinone (PQQ) prevents fibril formation of alpha-synuclein . Biochem Biophys Res Commun. (2006)
- Zhang JJ, Zhang RF, Meng XK Protective effect of pyrroloquinoline quinone against Abeta-induced neurotoxicity in human neuroblastoma SH-SY5Y cells . Neurosci Lett. (2009)
- Watanabe A, et al Nephrotoxicity of pyrroloquinoline quinone in rats . Hiroshima J Med Sci. (1989)
- Nakano M, et al Genotoxicity of pyrroloquinoline quinone (PQQ) disodium salt (BioPQQ™) . Regul Toxicol Pharmacol. (2013)
(Users who contributed to this page include shrillthrill, GregoryLopez, KurtisFrank, KamalPatel )
Pyrroloquinoline quinone
Pyrroloquinoline quinone (PQQ) was discovered by J.G. Hauge as the third redox cofactor after nicotinamide and flavin in bacteria (although he hypothesised that it was naphthoquinone).[1] Anthony and Zatman also found the unknown redox cofactor in alcohol dehydrogenase and named it methoxatin.[2] In 1979, Salisbury and colleagues[3] as well as Duine and colleagues[4] extracted this prosthetic group from methanol dehydrogenase of methylotrophs and identified its molecular structure. Adachi and colleagues identified that PQQ was also found in Acetobacter.[5]
These enzymes containing PQQ are called quinoproteins. Glucose dehydrogenase, one of the quinoproteins, is used as a glucose sensor. Subsequently, PQQ was found to stimulate growth in bacteria.[6] In addition, antioxidant and neuroprotective effects were also found.[7]
https://en.wikipedia.org/wiki/Pyrroloquinoline_quinone
Research in animals[edit]
Mitochondrial biogenesis in mice[edit]
In 2010, researchers at the University of California at Davis released a peer-reviewed publication showing that PQQ’s critical role in growth and development stems from its unique ability to activate cell signaling pathways directly involved in cellular energy metabolism, development, and function. The study demonstrated that PQQ not only protects mouse hepatocyte mitochondria from oxidative stress—it promotes the spontaneous generation of new mitochondria within aging cells, a process known asmitochondrial biogenesis.[8]
The team of researchers at the University of California analyzed PQQ’s influence over cell signaling pathways involved in the generation of new mitochondria and found that there are three mouse proteins activated by PQQ that cause cells to undergo spontaneous mitochondrial biogenesis: peroxisome proliferator-activated receptor gamma coactivator 1-alpha, cAMP response element-binding protein, and the DJ-1 protein.[8]
Cardioprotection in rat models[edit]
Damage from a heart attack, like a stroke, is inflicted via ischemic reperfusion injury. PQQ administration reduces the size of damaged areas in animal models of acute heart attack (myocardial infarction). Significantly, this occurs irrespective of whether the chemical is given before or after the ischemic event itself, suggesting that administration within the first hours of medical response may offer benefits to heart attack victims.[9]
Researchers at the University of California at San Francisco investigated this potential, comparing PQQ with the beta blocker metoprolol—a standard post-MI clinical treatment. Independently, both treatments reduced the size of the damaged areas and protected against heart muscle dysfunction. When given together, the left ventricle’s pumping pressure was enhanced. The combination of PQQ with metoprolol also increased mitochondrial energy-producing functions—but the effect was modest compared with PQQ alone. Only PQQ favorably reduced lipid peroxidation. These results led the researchers to conclude that “PQQ is superior to metoprolol in protecting mitochondria from ischemia/reperfusion oxidative damage.” [10]
Subsequent research has also demonstrated that PQQ helps heart muscle cells resist acute oxidative stress by preserving and enhancing mitochondrial function.[11]
Radiation poisoning in mice[edit]
In a study of gamma radiation poisoning in mice, 4mg/kg of PQQ improved 30-day survival from 2/20 to 12/20 at an 8 Gy dose.[12]
Neuroprotection[edit]
PQQ is a neuroprotective compound that has been shown in a small number of preliminary studies to protect memory and cognition in aging animals and humans.[13][14] It has been shown to reverse cognitive impairment caused by chronic oxidative stress in animal models and improve performance on memory tests.[15] PQQ supplementation stimulates the production and release of nerve growth factors in cells that support neurons in the brain,[16] a possible mechanism for the improvement of memory function it appears to produce in aging humans and rats.
PQQ has also been shown to safeguard against the self-oxidation of the DJ-1 protein, an early step in the onset of some forms of Parkinson’s disease.[17]
PQQ protects brain cells against oxidative damage following ischemia-reperfusion injury—the inflammation and oxidative damage that result from the sudden return of blood and nutrients to tissues deprived of them by stroke.[18] Reactive nitrogen species (RNS) arise spontaneously following stroke and spinal cord injuries and impose severe stresses on damaged neurons, contributing to subsequent long-term neurological damage.[19] PQQ suppresses RNS in experimentally induced strokes,[20] and provides additional protection following spinal cord injury by blocking inducible nitric oxide synthase (iNOS), a major source of RNS.[21]
In animal models, administration of PQQ immediately prior to induction of stroke significantly reduces the size of the damaged brain area.[22] These observations have been compounded by the observation in vivo that PQQ protects against the likelihood of severe stroke in an experimental animal model for stroke and brain hypoxia.[18]
PQQ also affects some of the brain’s neurotransmitter systems. It protects neurons by modulating the properties of the N-methyl-D-aspartate (NMDA) receptor,[23][24] and so reducing excitotoxicity—the damaging consequence of long-term overstimulation of neurons that is associated with many neurodegenerative diseases and seizures.[25][26][27][28]
PQQ also protects the brain against neurotoxicity induced by other powerful toxins, including mercury[29](a suspected factor in the development of Alzheimer’s disease[30]) and oxidopamine[31] (a potent neurotoxin used by scientists to induce Parkinsonism in laboratory animals by destroying dopaminergic and noradrenergic neurons.[32])
PQQ prevents aggregation of alpha-synuclein, a protein associated with Parkinson’s disease.[33] PQQ also protects nerve cells from the toxic effects of the amyloid-beta protein linked with Alzheimer’s disease,[34]and reduces the formation of new amyloid beta aggregates.[35]
Controversy[edit]
Although Nature Magazine published the 2003 paper by Kasahara and Kato which essentially stated that PQQ was a new vitamin, they also subsequently published, in 2005, an article by Chris Anthony and his colleague L.M. Fenton of the University of Southhampton which states that the 2003 Kasahara and Kato paper drew incorrect and unsubstantiated conclusions.[36] On his website,[37] Anthony discusses the Nature Magazine publications:
When I pointed out to the journal Nature that their high reputation was being used to justify investments of millions of dollars in the development of PQQ as a vitamin, they investigated the original paper, agreed with our objections and published our argument against it (Felton & Anthony, Nature Vol. 433, 2005). They also published (alongside ours) a paper by Rucker disagreeing with the conclusions of Kasahara and Kato on nutritional grounds, concluding “that insufficient information is available so far to state that PQQ uniquely performs an essential vitamin function in animals”.
Anthony further states on his website that “No mammalian PQQ-containing enzyme (quinoprotein) has been described” and that PQQ therefore cannot be called a “vitamin”. The latter statement is an exaggeration, since there is one mammalian enzyme which appears to use PQQ as a cofactor:[38]
References[edit]
- Jump up^ Hauge JG (1964). “Glucose dehydrogenase of bacterium anitratum: an enzyme with a novel prosthetic group”. J Biol Chem 239: 3630–9. PMID 14257587.
- Jump up^ Anthony C, Zatman LJ (1967). “The microbial oxidation of methanol. The prosthetic group of the alcohol dehydrogenase of Pseudomonas sp. M27: a new oxidoreductase prosthetic group”. Biochem J 104 (3): 960–9. PMC 1271238. PMID 6049934.
- Jump up^ Salisbury SA, Forrest HS, Cruse WB, Kennard O (1979). “A novel coenzyme from bacterial primary alcohol dehydrogenases”. Nature 280 (5725): 843–4. doi:10.1038/280843a0. PMID 471057.
- Jump up^ Westerling J, Frank J, Duine JA (1979). “The prosthetic group of methanol dehydrogenase from Hyphomicrobium X: electron spin resonance evidence for a quinone structure”. Biochem Biophys Res Commun87 (3): 719–24. doi:10.1016/0006-291X(79)92018-7. PMID 222269.
- Jump up^ Ameyama M, Matsushita K, Ohno Y, Shinagawa E, Adachi O (1981). “Existence of a novel prosthetic group, PQQ, in membrane-bound, electron transport chain-linked, primary dehydrogenases of oxidative bacteria”.FEBS Lett 130 (2): 179–83. doi:10.1016/0014-5793(81)81114-3. PMID 6793395.
- Jump up^ Ameyama M, Matsushita K, Shinagawa E, Hayashi M, Adachi O (1988). “Pyrroloquinoline quinone: excretion by methylotrophs and growth stimulation for microorganisms”. BioFactors 1 (1): 51–3. PMID 2855583.
- Jump up^ Rucker R, Chowanadisai W, Nakano M. (2009). “Potential physiological importance of pyrroloquinoline quinone”. Altern Med Rev. 14 (3): 179–83.
- ^ Jump up to:a b Chowanadisai, W.; Bauerly, K. A.; Tchaparian, E.; Wong, A.; Cortopassi, G. A.; Rucker, R. B. (January 2010). “Pyrroloquinoline quinone stimulates mitochondrial biogenesis through cAMP response element-binding protein phosphorylation and increased PGC-1alpha expression”. Journal of Biological Chemistry 285 (1): 142–152. doi:10.1074/jbc.M109.030130. PMC 2804159. PMID 19861415.
- Jump up^ Zhu, B. Q.; Zhou, H. Z.; Teerlink, J. R.; Karliner, J. S. (November 2004). “Pyrroloquinoline quinone (PQQ) decreases myocardial infarct size and improves cardiac function in rat models of ischemia and ischemia/reperfusion”. Cardiovascular Drugs and Therapy 18 (6): 421–431. doi:10.1007/s10557-004-6219-x.PMID 15770429.
- Jump up^ Zhu, B. -Q.; Simonis, U.; Cecchini, G.; Zhou, H. -Z.; Li, L.; Teerlink, J. R.; Karliner, J. S. (June 2006). “Comparison of pyrroloquinoline quinone and/or metoprolol on myocardial infarct size and mitochondrial damage in a rat model of ischemia/reperfusion injury”. Journal of Cardiovascular Pharmacology and Therapeutics 11 (2): 119–128. doi:10.1177/1074248406288757. PMID 16891289.
- Jump up^ Tao, R; Karliner, J; Simonis, U; Zheng, J; Zhang, J; Honbo, N; Alano, C (2007). “Pyrroloquinoline quinone preserves mitochondrial function and prevents oxidative injury in adult rat cardiac myocytes”. Biochemical and Biophysical Research Communications 363 (2): 257–62. doi:10.1016/j.bbrc.2007.08.041. PMC 2844438.PMID 17880922.
- Jump up^ Xiong, X. H.; Zhao, Y; Ge, X; Yuan, S. J.; Wang, J. H.; Zhi, J. J.; Yang, Y. X.; Du, B. H.; Guo, W. J.; Wang, S. S.; Yang, D. X.; Zhang, W. C. (2011). “Production and radioprotective effects of pyrroloquinoline quinone”. International Journal of Molecular Sciences 12 (12): 8913–23. doi:10.3390/ijms12128913.PMC 3257108. PMID 22272111.
- Jump up^ Takatsu, H; Owada, K; Abe, K; Nakano, M; Urano, S (2009). “Effect of vitamin E on learning and memory deficit in aged rats”. Journal of nutritional science and vitaminology 55 (5): 389–93. doi:10.3177/jnsv.55.389.PMID 19926923.
- Jump up^ Nakano M, Ubukata K, Yamamoto T, Yamaguchi H. (2009). “Effect of pyrroloquinoline quinone (PQQ) on mental status of middle-aged and elderly persons”. Food Style 21 13 (7): 50–52.
- Jump up^ Ohwada, K.; Takeda, H.; Yamazaki, M.; Isogai, H.; Nakano, M.; Shimomura, M.; Fukui, K.; Urano, S. (January 2008). “Pyrroloquinoline quinone (PQQ) prevents cognitive deficit caused by oxidative stress in rats”. Journal of Clinical Biochemistry and Nutrition 42 (1): 29–34. doi:10.3164/jcbn.2008005.PMC 2212345. PMID 18231627.
- Jump up^ Murase, K; Hattori, A; Kohno, M; Hayashi, K (1993). “Stimulation of nerve growth factor synthesis/secretion in mouse astroglial cells by coenzymes”. Biochemistry and molecular biology international 30 (4): 615–21.PMID 8401318.
- Jump up^ Nunome, K; Miyazaki, S; Nakano, M; Iguchi-Ariga, S; Ariga, H (2008). “Pyrroloquinoline quinone prevents oxidative stress-induced neuronal death probably through changes in oxidative status of DJ-1”. Biological & Pharmaceutical Bulletin 31 (7): 1321–6. doi:10.1248/bpb.31.1321. PMID 18591768.
- ^ Jump up to:a b Jensen, FE; Gardner, GJ; Williams, AP; Gallop, PM; Aizenman, E; Rosenberg, PA (1994). “The putative essential nutrient pyrroloquinoline quinone is neuroprotective in a rodent model of hypoxic/ischemic brain injury”. Neuroscience 62 (2): 399–406. doi:10.1016/0306-4522(94)90375-1. PMID 7830887.
- Jump up^ Ono, K.; Suzuki, H.; Sawada, M. (2010-10-05). “Delayed neural damage is induced by iNOS-expressing microglia in a brain injury model”. Neuroscience Letters 473 (2): 146–150. doi:10.1016/j.neulet.2010.02.041.PMID 20178828.
- Jump up^ Zhang, Y; Rosenberg, PA (2002). “The essential nutrient pyrroloquinoline quinone may act as a neuroprotectant by suppressing peroxynitrite formation”. The European Journal of Neuroscience 16 (6): 1015–24. doi:10.1046/j.1460-9568.2002.02169.x. PMID 12383230.
PQQ and Statin Damage
By Dr. Duane Graveline MD, MPH
Those of you who have been following my research during the past two years will know that I consider mitochondrial DNA damage as the ultimate result for some of statin drug intake.
Through mevalonate blockade, statins directly inhibit CoQ10 synthesis making mitochondrial damage and mutation all but inevitable. Furthermore, the inhibitory effect of statins on dolichol synthesis makes repair of DNA damage all the more difficult because of dolichol’s vital role in glycoprotein (glycohydrolase) synthesis.
Recently I have learned of another biochemical substance that also is implicated in this process of mitochondrial maintenance. The name of this biochemical is pyrroloquinoline quinone with the shorthand version being PQQ.
This substance has been discovered only in the past decade with its vital role in mitochondrial support having been documented only in the past several years. From what I have read of this substance, trying to get beyond the hype, it is worth considering for those of us who have been damaged by statins, whether by cognitive dysfunction, permanent myopathy, ALS like symptoms, or peripheral neuropathy.
Dietary sources of PQQ include many fruits and vegetables and egg yolk. Natto ( fermented soybeans ) has the highest concentration but parsley, green peppers, papaya, kiwi fruit and spinach are all good sources. PQQ is also available as a dietary supplement. Human trials and studies will need to be performed to support any claims for the benefits of PQQ supplementation.
One promotion for PQQ begins with, “The more functional mitochondria you have in your cells, the greater your overall health and durability,” which is the premise of my new e-book, The Dark Side of Statins, so my interest in this substance is obvious.
The problem is that as we age, our mitochondria degrade and become dysfunctional. Compared with nuclear DNA, mitochondrial DNA is left almost entirely exposed to the ravages of free radicals. It attaches directly to the inner membrane where the mitochondria’s furnace rages continuously.
Statin drugs directly hasten this process of mitochondrial DNA degradation by direct inhibition of CoQ10 and dolichol synthesis. The ultimate cause of statin associated adverse reactions is this progressive deterioration of mitochondrial DNA. PQQ is being touted not only for its extra anti-oxidant protection in the fight against free radicals but also for its potential use for mitochondrial genesis
https://youtu.be/-PA-buwI3q4
https://youtu.be/-PA-buwI3q4?t=406
https://youtu.be/-PA-buwI3q4?t=455
https://youtu.be/j1FmK4582mA
This is part 1 (of nine parts) of the Preventing and Reversing Alzheimer’s Disease presentation, an earlier version of which was presented to the San Francisco bay area Smart Life Forum in January of 2009. This part covers the verbal introduction and the falling-dominoes illustration of the Alzheimer’s cascade
https://youtu.be/hQipKkFppzI
https://youtu.be/oX6RG6ky0yU
This is part three of the Prevention and Reversal of Alzheimer’s Disease presentation. This part covers the Alzheimer’s Map (schematic), mitochondria, and creatine kinase (the first domino in the Alzheimer’s disease cascade).
https://youtu.be/2dutY1zUM7k
https://youtu.be/kktDaCRwnFM
This is part six of the Prevention and Reversal of Alzheimer’s Disease presentation. This part covers the antioxidant defense system, glutathione (the “star of the movie”), and the brain’s phosphorylation cycle (the brains “biorhythm).
https://youtu.be/kktDaCRwnFM?t=81
https://youtu.be/kktDaCRwnFM?t=118
https://youtu.be/iC9GOU78OwU
Read Full Post »