Clinical Laboratory Challenges
Larry H. Bernstein, MD, FCAP, Curator
LPBI
CLINICAL LABORATORY NEWS
The Lab and CJD: Safe Handling of Infectious Prion Proteins
Author: Mari DeMarco, PhD // Date: NOV.1.2015 // Source: Clinical Laboratory News
https://www.aacc.org/publications/cln/articles/2015/november/the-lab-and-cjd-safe-handling-of-infectious-prion-proteins

https://www.aacc.org/~/media/images/cln/articles/2015/november/prion-protiens.jpg
Body fluids from individuals with possible Creutzfeldt-Jakob disease (CJD) present distinctive safety challenges for clinical laboratories. Sporadic, iatrogenic, and familial CJD (known collectively as classic CJD), along with variant CJD, kuru, Gerstmann-Sträussler-Scheinker, and fatal familial insomnia, are prion diseases, also known as transmissible spongiform encephalopathies. Prion diseases affect the central nervous system, and from the onset of symptoms follow a typically rapid progressive neurological decline. While prion diseases are rare, it is not uncommon for the most prevalent form—sporadic CJD—to be included in the differential diagnosis of individuals presenting with rapid cognitive decline. Thus, laboratories may deal with a significant number of possible CJD cases, and should have protocols in place to process specimens, even if a confirmatory diagnosis of CJD is made in only a fraction of these cases.
The Lab’s Role in Diagnosis
Laboratory protocols for handling specimens from individuals with possible, probable, and definitive cases of CJD are important to ensure timely and appropriate patient management. When the differential includes CJD, an attempt should be made to rule-in or out other causes of rapid neurological decline. Laboratories should be prepared to process blood and cerebrospinal fluid (CSF) specimens in such cases for routine analyses.
Definitive diagnosis requires identification of prion aggregates in brain tissue, which can be achieved by immunohistochemistry, a Western blot for proteinase K-resistant prions, and/or by the presence of prion fibrils. Thus, confirmatory diagnosis is typically achieved at autopsy. A probable diagnosis of CJD is supported by elevated concentration of 14-3-3 protein in CSF (a non-specific marker of neurodegeneration), EEG, and MRI findings. Thus, the laboratory may be required to process and send CSF samples to a prion surveillance center for 14-3-3 testing, as well as blood samples for sequencing of the PRNP gene (in inherited cases).
Processing Biofluids
Laboratories should follow standard protective measures when working with biofluids potentially containing abnormally folded prions, such as donning standard personal protective equipment (PPE); avoiding or minimizing the use of sharps; using single-use disposable items; and processing specimens to minimize formation of aerosols and droplets. An additional safety consideration is the use of single-use disposal PPE; otherwise, re-usable items must be either cleaned using prion-specific decontamination methods, or destroyed.
Blood. In experimental models, infectivity has been detected in the blood; however, there have been no cases of secondary transmission of classical CJD via blood product transfusions in humans. As such, blood has been classified, on epidemiological evidence by the World Health Organization (WHO), as containing “no detectible infectivity,” which means it can be processed by routine methods. Similarly, except for CSF, all other body fluids contain no infectivity and can be processed following standard procedures.
In contrast to classic CJD, there have been four cases of suspected secondary transmission of variant CJD via transfused blood products in the United Kingdom. Variant CJD, the prion disease associated with mad cow disease, is unique in its distribution of prion aggregates outside of the central nervous system, including the lymph nodes, spleen, and tonsils. For regions where variant CJD is a concern, laboratories should consult their regulatory agencies for further guidance.
CSF. Relative to highly infectious tissues of the brain, spinal cord, and eye, infectivity has been identified less often in CSF and is considered to have “low infectivity,” along with kidney, liver, and lung tissue. Since CSF can contain infectious material, WHO has recommended that analyses not be performed on automated equipment due to challenges associated with decontamination. Laboratories should perform a risk assessment of their CSF processes, and, if deemed necessary, consider using manual methods as an alternative to automated systems.
Decontamination
The infectious agent in prion disease is unlike any other infectious pathogen encountered in the laboratory; it is formed of misfolded and aggregated prion proteins. This aggregated proteinacious material forms the infectious unit, which is incredibly resilient to degradation. Moreover, in vitro studies have demonstrated that disrupting large aggregates into smaller aggregates increases cytotoxicity. Thus, if the aim is to abolish infectivity, all aggregates must be destroyed. Disinfectant procedures used for viral, bacterial, and fungal pathogens such as alcohol, boiling, formalin, dry heat (<300°C), autoclaving at 121°C for 15 minutes, and ionizing, ultraviolet, or microwave radiation, are either ineffective or variably effective against aggregated prions.
The only means to ensure no risk of residual infectious prions is to use disposable materials. This is not always practical, as, for instance, a biosafety cabinet cannot be discarded if there is a CSF spill in the hood. Fortunately, there are several protocols considered sufficient for decontamination. For surfaces and heat-sensitive instruments, such as a biosafety cabinet, WHO recommends flooding the surface with 2N NaOH or undiluted NaClO, letting stand for 1 hour, mopping up, and rinsing with water. If the surface cannot tolerate NaOH or NaClO, thorough cleaning will remove most infectivity by dilution. Laboratories may derive some additional benefit by using one of the partially effective methods discussed previously. Non-disposable heat-resistant items preferably should be immersed in 1N NaOH, heated in a gravity displacement autoclave at 121°C for 30 min, cleaned and rinsed in water, then sterilized by routine methods. WHO has outlined several alternate decontamination methods. Using disposable cover sheets is one simple solution to avoid contaminating work surfaces and associated lengthy decontamination procedures.
With standard PPE—augmented by a few additional safety measures and prion-specific decontamination procedures—laboratories can safely manage biofluid testing in cases of prion disease.
The Microscopic World Inside Us
Emerging Research Points to Microbiome’s Role in Health and Disease
Author: Deborah Levenson // Date: NOV.1.2015 // Source: Clinical Laboratory News
https://www.aacc.org/publications/cln/articles/2015/november/the-microscopic-world-inside-us-emerging-research-points-to-microbiomes-role-in-health-and-disease

https://www.aacc.org/~/media/images/cln/articles/2015/november/tsrf157067927.jpg
Thousands of species of microbes—bacteria, viruses, fungi, and protozoa—inhabit every internal and external surface of the human body. Collectively, these microbes, known as the microbiome, outnumber the body’s human cells by about 10 to 1 and include more than 1,000 species of microorganisms and several million genes residing in the skin, respiratory system, urogenital, and gastrointestinal tracts. The microbiome’s complicated relationship with its human host is increasingly considered so crucial to health that researchers sometimes call it “the forgotten organ.”
Disturbances to the microbiome can arise from nutritional deficiencies, antibiotic use, and antiseptic modern life. Imbalances in the microbiome’s diverse microbial communities, which interact constantly with cells in the human body, may contribute to chronic health conditions, including diabetes, asthma and allergies, obesity and the metabolic syndrome, digestive disorders including irritable bowel syndrome (IBS), and autoimmune disorders like multiple sclerosis and rheumatoid arthritis, research shows.
While study of the microbiome is a growing research enterprise that has attracted enthusiastic media attention and venture capital, its findings are largely preliminary. But some laboratorians are already developing a greater appreciation for the microbiome’s contributions to human biochemistry and are considering a future in which they expect to measure changes in the microbiome to monitor disease and inform clinical practice.
Pivot Toward the Microbiome
Following the National Institutes of Health (NIH) Human Genome Project, many scientists noted the considerable genetic signal from microbes in the body and the existence of technology to analyze these microorganisms. That realization led NIH to establish the Human Microbiome Project in 2007, said Lita Proctor, PhD, its program director. In the project’s first phase, researchers studied healthy adults to produce a reference set of microbiomes and a resource of metagenomic sequences of bacteria in the airways, skin, oral cavities, and the gastrointestinal and vaginal tracts, plus a catalog of microbial genome sequences of reference strains. Researchers also evaluated specific diseases associated with disturbances in the microbiome, including gastrointestinal diseases such as Crohn’s disease, ulcerative colitis, IBS, and obesity, as well as urogenital conditions, those that involve the reproductive system, and skin diseases like eczema, psoriasis, and acne.
Phase 1 studies determined the composition of many parts of the microbiome, but did not define how that composition affects health or specific disease. The project’s second phase aims to “answer the question of what microbes actually do,” explained Proctor. Researchers are now examining properties of the microbiome including gene expression, protein, and human and microbial metabolite profiles in studies of pregnant women at risk for preterm birth, the gut hormones of patients at risk for IBS, and nasal microbiomes of patients at risk for type 2 diabetes.
Promising Lines of Research
Cystic fibrosis and microbiology investigator Michael Surette, PhD, sees promising microbiome research not just in terms of evidence of its effects on specific diseases, but also in what drives changes in the microbiome. Surette is Canada research chair in interdisciplinary microbiome research in the Farncombe Family Digestive Health Research Institute at McMaster University
in Hamilton, Ontario.
One type of study on factors driving microbiome change examines how alterations in composition and imbalances in individual patients relate to improving or worsening disease. “IBS, cystic fibrosis, and chronic obstructive pulmonary disease all have periods of instability or exacerbation,” he noted. Surette hopes that one day, tests will provide clinicians the ability to monitor changes in microbial composition over time and even predict when a patient’s condition is about to deteriorate. Monitoring perturbations to the gut microbiome might also help minimize collateral damage to the microbiome during aggressive antibiotic therapy for hospitalized patients, he added.
Monitoring changes to the microbiome also might be helpful for “culture negative” patients, who now may receive multiple, unsuccessful courses of different antibiotics that drive antibiotic resistance. Frustration with standard clinical biology diagnosis of lung infections in cystic fibrosis patients first sparked Surette’s investigations into the microbiome. He hopes that future tests involving the microbiome might also help asthma patients with neutrophilia, community-acquired pneumonia patients who harbor complex microbial lung communities lacking obvious pathogens, and hospitalized patients with pneumonia or sepsis. He envisions microbiome testing that would look for short-term changes indicating whether or not a drug is effective.
Companion Diagnostics
Daniel Peterson, MD, PhD, an assistant professor of pathology at Johns Hopkins University School of Medicine in Baltimore, believes the future of clinical testing involving the microbiome lies in companion diagnostics for novel treatments, and points to companies that are already developing and marketing tests that will require such assays.
Examples of microbiome-focused enterprises abound, including Genetic Analysis, based in Oslo, Norway, with its high-throughput test that uses 54 probes targeted to specific bacteria to measure intestinal gut flora imbalances in inflammatory bowel disease and irritable bowel syndrome patients. Paris, France-based Enterome is developing both novel drugs and companion diagnostics for microbiome-related diseases such as IBS and some metabolic diseases. Second Genome, based in South San Francisco, has developed an experimental drug, SGM-1019, that the company says blocks damaging activity of the microbiome in the intestine. Cambridge, Massachusetts-based Seres Therapeutics has received Food and Drug Administration orphan drug designation for SER-109, an oral therapeutic intended to correct microbial imbalances to prevent recurrent Clostridium difficile infection in adults.
One promising clinical use of the microbiome is fecal transplantation, which both prospective and retrospective studies have shown to be effective in patients with C. difficile infections who do not respond to front-line therapies, said James Versalovic, MD, PhD, director of Texas Children’s Hospital Microbiome Center and professor of pathology at Baylor College of Medicine in Houston. “Fecal transplants and other microbiome replacement strategies can radically change the composition of the microbiome in hours to days,” he explained.
But NIH’s Proctor discourages too much enthusiasm about fecal transplant. “Natural products like stool can have [side] effects,” she pointed out. “The [microbiome research] field needs to mature and we need to verify outcomes before anything becomes routine.”
Hurdles for Lab Testing
While he is hopeful that labs someday will use the microbiome to produce clinically useful information, Surette pointed to several problems that must be solved beforehand. First, molecular methods commonly used right now should be more quantitative and accurate. Additionally, research on the microbiome encompasses a wide variety of protocols, some of which are better at extracting particular types of bacteria and therefore can give biased views of communities living in the body. Also, tests may need to distinguish between dead and live microbes. Another hurdle is that labs using varied bioinfomatic methods may produce different results from the same sample, a problem that Surette sees as ripe for a solution from clinical laboratorians, who have expertise in standardizing robust protocols and in automating tests.
One way laboratorians can prepare for future, routine microbiome testing is to expand their notion of clinical chemistry to include both microbial and human biochemistry. “The line between microbiome science and clinical science is blurring,” said Versalovic. “When developing future assays to detect biochemical changes in disease states, we must consider the contributions of microbial metabolites and proteins and how to tailor tests to detect them.” In the future, clinical labs may test for uniquely microbial metabolites in various disease states, he predicted.
Automated Review of Mass Spectrometry Results
Can We Achieve Autoverification?
Author: Katherine Alexander and Andrea R. Terrell, PhD // Date: NOV.1.2015 // Source:Clinical Laboratory News
https://www.aacc.org/publications/cln/articles/2015/november/automated-review-of-mass-spectrometry-results-can-we-achieve-autoverification
Paralleling the upswing in prescription drug misuse, clinical laboratories are receiving more requests for mass spectrometry (MS) testing as physicians rely on its specificity to monitor patient compliance with prescription regimens. However, as volume has increased, reimbursement has declined, forcing toxicology laboratories both to increase capacity and lower their operational costs—without sacrificing quality or turnaround time. Now, new solutions are available enabling laboratories to bring automation to MS testing and helping them with the growing demand for toxicology and other testing.
What is the typical MS workflow?
A typical workflow includes a long list of manual steps. By the time a sample is loaded onto the mass spectrometer, it has been collected, logged into the lab information management system (LIMS), and prepared for analysis using a variety of wet chemistry techniques.
Most commercial clinical laboratories receive enough samples for MS analysis to batch analyze those samples. A batch consists of a calibrator(s), quality control (QC) samples, and patient/donor samples. Historically, the method would be selected (i.e. “analysis of opiates”), sample identification information would be entered manually into the MS software, and the instrument would begin analyzing each sample. Upon successful completion of the batch, the MS operator would view all of the analytical data, ensure the QC results were acceptable, and review each patient/donor specimen, looking at characteristics such as peak shape, ion ratios, retention time, and calculated concentration.
The operator would then post acceptable results into the LIMS manually or through an interface, and unacceptable results would be rescheduled or dealt with according to lab-specific protocols. In our laboratory we perform a final certification step for quality assurance by reviewing all information about the batch again, prior to releasing results for final reporting through the LIMS.
What problems are associated with this workflow?
The workflow described above results in too many highly trained chemists performing manual data entry and reviewing perfectly acceptable analytical results. Lab managers would prefer that MS operators and certifying scientists focus on troubleshooting problem samples rather than reviewing mounds of good data. Not only is the current process inefficient, it is mundane work prone to user errors. This risks fatigue, disengagement, and complacency by our highly skilled scientists.
Importantly, manual processes also take time. In most clinical lab environments, turnaround time is critical for patient care and industry competitiveness. Lab directors and managers are looking for solutions to automate mundane, error-prone tasks to save time and costs, reduce staff burnout, and maintain high levels of quality.
How can software automate data transfer from MS systems to LIMS?
Automation is not a new concept in the clinical lab. Labs have automated processes in shipping and receiving, sample preparation, liquid handling, and data delivery to the end user. As more labs implement MS, companies have begun to develop special software to automate data analysis and review workflows.
In July 2011, AIT Labs incorporated ASCENT into our workflow, eliminating the initial manual peak review step. ASCENT is an algorithm-based peak picking and data review system designed specifically for chromatographic data. The software employs robust statistical and modeling approaches to the raw instrument data to present the true signal, which often can be obscured by noise or matrix components.
The system also uses an exponentially modified Gaussian (EMG) equation to apply a best-fit model to integrated peaks through what is often a noisy signal. In our experience, applying the EMG results in cleaner data from what might appear to be poor chromatography ultimately allows us to reduce the number of samples we might otherwise rerun.
How do you validate the quality of results?
We’ve developed a robust validation protocol to ensure that results are, at minimum, equivalent to results from our manual review. We begin by building the assay in ASCENT, entering assay-specific information from our internal standard operating procedure (SOP). Once the assay is configured, validation proceeds with parallel batch processing to compare results between software-reviewed data and staff-reviewed data. For new implementations we run eight to nine batches of 30–40 samples each; when we are modifying or upgrading an existing implementation we run a smaller number of batches. The parallel batches should contain multiple positive and negative results for all analytes in the method, preferably spanning the analytical measurement range of the assay.
The next step is to compare the results and calculate the percent difference between the data review methods. We require that two-thirds of the automated results fall within 20% of the manually reviewed result. In addition to validating patient sample correlation, we also test numerous quality assurance rules that should initiate a flag for further review.
What are the biggest challenges during implementation and continual improvement initiatives?
On the technological side, our largest hurdle was loading the sequence files into ASCENT. We had created an in-house mechanism for our chemists to upload the 96-well plate map for their batch into the MS software. We had some difficulty transferring this information to ASCENT, but once we resolved this issue, the technical workflow proceeded fairly smoothly.
The greater challenge was changing our employees’ mindset from one of fear that automation would displace them, to a realization that learning this new technology would actually make them more valuable. Automating a non-mechanical process can be a difficult concept for hands-on scientists, so managers must be patient and help their employees understand that this kind of technology leverages the best attributes of software and people to create a powerful partnership.
We recommend that labs considering automated data analysis engage staff in the validation and implementation to spread the workload and the knowledge. As is true with most technology, it is best not to rely on just one or two super users. We also found it critical to add supervisor level controls on data file manipulation, such as removing a sample that wasn’t run from the sequence table. This can prevent inadvertent deletion of a file, requiring reinjection of the entire batch!
Understanding Fibroblast Growth Factor 23
Author: Damien Gruson, PhD // Date: OCT.1.2015 // Source: Clinical Laboratory News
https://www.aacc.org/publications/cln/articles/2015/october/understanding-fibroblast-growth-factor-23
What is the relationship of FGF-23 to heart failure?
A Heart failure (HF) is an increasingly common syndrome associated with high morbidity, elevated hospital readmission rates, and high mortality. Improving diagnosis, prognosis, and treatment of HF requires a better understanding of its different sub-phenotypes. As researchers gained a comprehensive understanding of neurohormonal activation—one of the hallmarks of HF—they discovered several biomarkers, including natriuretic peptides, which now are playing an important role in sub-phenotyping HF and in driving more personalized management of this chronic condition.
Like the natriuretic peptides, fibroblast growth factor 23 (FGF-23) could become important in risk-stratifying and managing HF patients. Produced by osteocytes, FGF-23 is a key regulator of phosphorus homeostasis. It binds to renal and parathyroid FGF-Klotho receptor heterodimers, resulting in phosphate excretion, decreased 1-α-hydroxylation of 25-hydroxyvitamin D, and decreased parathyroid hormone (PTH) secretion. The relationship to PTH is important because impaired homeostasis of cations and decreased glomerular filtration rate might contribute to the rise of FGF-23. The amino-terminal portion of FGF-23 (amino acids 1-24) serves as a signal peptide allowing secretion into the blood, and the carboxyl-terminal portion (aa 180-251) participates in its biological action.
How might FGF-23 improve HF risk assessment?
Studies have shown that FGF-23 is related to the risk of cardiovascular diseases and mortality. It was first demonstrated that FGF-23 levels were independently associated with left ventricular mass index and hypertrophy as well as mortality in patients with chronic kidney disease (CKD). FGF-23 also has been associated with left ventricular dysfunction and atrial fibrillation in coronary artery disease subjects, even in the absence of impaired renal function.
FGF-23 and FGF receptors are both expressed in the myocardium. It is possible that FGF-23 has direct effects on the heart and participates in the physiopathology of cardiovascular diseases and HF. Experiments have shown that for in vitro cultured rat cardiomyocytes, FGF-23 stimulates pathological hypertrophy by activating the calcineurin-NFAT pathway—and in wild-type mice—the intra-myocardial or intravenous injection of FGF-23 resulted in left ventricular hypertrophy. As such, FGF-23 appears to be a potential stimulus of myocardial hypertrophy, and increased levels may contribute to the worsening of heart failure and long-term cardiovascular death.
Researchers have documented that HF patients have elevated FGF-23 circulating levels. They have also found a significant correlation between plasma levels of FGF-23 and B-type natriuretic peptide, a biomarker related to ventricular stretch and cardiac hypertrophy, in patients with left ventricular hypertrophy. As such, measuring FGF-23 levels might be a useful tool to predict long-term adverse cardiovascular events in HF patients.
Interestingly, researchers have documented a significant relationship between FGF-23 and PTH in both CKD and HF patients. As PTH stimulates FGF-23 expression, it could be that in HF patients, increased PTH levels increase the bone expression of FGF-23, which enhances its effects on the heart.
The Past, Present, and Future of Western Blotting in the Clinical Laboratory
Author: Curtis Balmer, PhD // Date: OCT.1.2015 // Source: Clinical Laboratory News
https://www.aacc.org/publications/cln/articles/2015/october/the-past-present-and-future-of-western-blotting-in-the-clinical-laboratory
Much of the discussion about Western blotting centers around its performance as a biological research tool. This isn’t surprising. Since its introduction in the late 1970s, the Western blot has been adopted by biology labs of virtually every stripe, and become one of the most widely used techniques in the research armamentarium. However, Western blotting has also been employed in clinical laboratories to aid in the diagnosis of various diseases and disorders—an equally important and valuable application. Yet there has been relatively little discussion of its use in this context, or of how advances in Western blotting might affect its future clinical use.
Highlighting the clinical value of Western blotting, Stanley Naides, MD, medical director of Immunology at Quest Diagnostics observed that, “Western blotting has been a very powerful tool in the laboratory and for clinical diagnosis. It’s one of many various methods that the laboratorian brings to aid the clinician in the diagnosis of disease, and the selection and monitoring of therapy.” Indeed, Western blotting has been used at one time or the other to aid in the diagnosis of infectious diseases including hepatitis C (HCV), HIV, Lyme disease, and syphilis, as well as autoimmune disorders such as paraneoplastic disease and myositis conditions.
However, Naides was quick to point out that the choice of assays to use clinically is based on their demonstrated sensitivity and performance, and that the search for something better is never-ending. “We’re constantly looking for methods that improve detection of our target [protein],” Naides said. “There have been a number of instances where we’ve moved away from Western blotting because another method proves to be more sensitive.” But this search can also lead back to Western blotting. “We’ve gone away from other methods because there’s been a Western blot that’s been developed that’s more sensitive and specific. There’s that constant movement between methods as new tests are developed.”
In recent years, this quest has been leading clinical laboratories away from Western blotting toward more sensitive and specific diagnostic assays, at least for some diseases. Using confirmatory diagnosis of HCV infection as an example, Sai Patibandla, PhD, director of the immunoassay group at Siemens Healthcare Diagnostics, explained that movement away from Western blotting for confirmatory diagnosis of HCV infection began with a technical modification called Recombinant Immunoblotting Assay (RIBA). RIBA streamlines the conventional Western blot protocol by spotting recombinant antigen onto strips which are used to screen patient samples for antibodies against HCV. This approach eliminates the need to separate proteins and transfer them onto a membrane.
The RIBA HCV assay was initially manufactured by Chiron Corporation (acquired by Novartics Vaccines and Diagnostics in 2006). It received Food and Drug Administration (FDA) approval in 1999, and was marketed as Chiron RIBA HCV 3.0 Strip Immunoblot Assay. Patibandla explained that, at the time, the Chiron assay “…was the only FDA-approved confirmatory testing for HCV.” In 2013 the assay was discontinued and withdrawn from the market due to reports that it was producing false-positive results.
Since then, clinical laboratories have continued to move away from Western blot-based assays for confirmation of HCV in favor of the more sensitive technique of nucleic acid testing (NAT). “The migration is toward NAT for confirmation of HCV [diagnosis]. We don’t use immunoblots anymore. We don’t even have a blot now to confirm HCV,” Patibandla said.
Confirming HIV infection has followed a similar path. Indeed, in 2014 the Centers for Disease Control and Prevention issued updated recommendations for HIV testing that, in part, replaced Western blotting with NAT. This change was in response to the recognition that the HIV-1 Western blot assay was producing false-negative or indeterminable results early in the course of HIV infection.
At this juncture it is difficult to predict if this trend away from Western blotting in clinical laboratories will continue. One thing that is certain, however, is that clinicians and laboratorians are infinitely pragmatic, and will eagerly replace current techniques with ones shown to be more sensitive, specific, and effective. This raises the question of whether any of the many efforts currently underway to improve Western blotting will produce an assay that exceeds the sensitivity of currently employed techniques such as NAT.
Some of the most exciting and groundbreaking work in this area is being done by Amy Herr, PhD, a professor of bioengineering at University of California, Berkeley. Herr’s group has taken on some of the most challenging limitations of Western blotting, and is developing techniques that could revolutionize the assay. For example, the Western blot is semi-quantitative at best. This weakness dramatically limits the types of answers it can provide about changes in protein concentrations under various conditions.
To make Western blotting more quantitative, Herr’s group is, among other things, identifying losses of protein sample mass during the assay protocol. About this, Herr explains that the conventional Western blot is an “open system” that involves lots of handling of assay materials, buffers, and reagents that makes it difficult to account for protein losses. Or, as Kevin Lowitz, a senior product manager at Thermo Fisher Scientific, described it, “Western blot is a [simple] technique, but a really laborious one, and there are just so many steps and so many opportunities to mess it up.”
Herr’s approach is to reduce the open aspects of Western blot. “We’ve been developing these more closed systems that allow us at each stage of the assay to account for [protein mass] losses. We can’t do this exactly for every target of interest, but it gives us a really good handle [on protein mass losses],” she said. One of the major mechanisms Herr’s lab is using to accomplish this is to secure proteins to the blot matrix with covalent bonding rather than with the much weaker hydrophobic interactions that typically keep the proteins in place on the membrane.
Herr’s group also has been developing microfluidic platforms that allow Western blotting to be done on single cells, “In our system we’re doing thousands of independent Westerns on single cells in four hours. And, hopefully, we’ll cut that down to one hour over the next couple years.”
Other exciting modifications that stand to dramatically increase the sensitivity, quantitation, and through-put of Western blotting also are being developed and explored. For example, the use of capillary electrophoresis—in which proteins are conveyed through a small electrolyte-filled tube and separated according to size and charge before being dropped onto a blotting membrane—dramatically reduces the amount of protein required for Western blot analysis, and thereby allows Westerns to be run on proteins from rare cells or for which quantities of sample are extremely limited.
Jillian Silva, PhD, an associate specialist at the University of California, San Francisco Helen Diller Family Comprehensive Cancer Center, explained that advances in detection are also extending the capabilities of Western blotting. “With the advent of fluorescence detection we have a way to quantitate Westerns, and it is now more quantitative than it’s ever been,” said Silva.
Whether or not these advances produce an assay that is adopted by clinical laboratories remains to be seen. The emphasis on Western blotting as a research rather than a clinical tool may bias advances in favor of the needs and priorities of researchers rather than clinicians, and as Patibandla pointed out, “In the research world Western blotting has a certain purpose. [Researchers] are always coming up with new things, and are trying to nail down new proteins, so you cannot take Western blotting away.” In contrast, she suggested that for now, clinical uses of Western blotting remain “limited.”
Adapting Next Generation Technologies to Clinical Molecular Oncology Service
Author: Ronald Carter, PhD, DVM // Date: OCT.1.2015 // Source: Clinical Laboratory News
https://www.aacc.org/publications/cln/articles/2015/october/adapting-next-generation-technologies-to-clinical-molecular-oncology-service
Next generation technologies (NGT) deliver huge improvements in cost efficiency, accuracy, robustness, and in the amount of information they provide. Microarrays, high-throughput sequencing platforms, digital droplet PCR, and other technologies all offer unique combinations of desirable performance.
As stronger evidence of genetic testing’s clinical utility influences patterns of patient care, demand for NGT testing is increasing. This presents several challenges to clinical laboratories, including increased urgency, clinical importance, and breadth of application in molecular oncology, as well as more integration of genetic tests into synoptic reporting. Laboratories need to add NGT-based protocols while still providing old tests, and the pace of change is increasing.What follows is one viewpoint on the major challenges in adopting NGTs into diagnostic molecular oncology service.
Choosing a Platform
Instrument selection is a critical decision that has to align with intended test applications, sequencing chemistries, and analytical software. Although multiple platforms are available, a mainstream standard has not emerged. Depending on their goals, laboratories might set up NGTs for improved accuracy of mutation detection, massively higher sequencing capacity per test, massively more targets combined in one test (multiplexing), greater range in sequencing read length, much lower cost per base pair assessed, and economy of specimen volume.
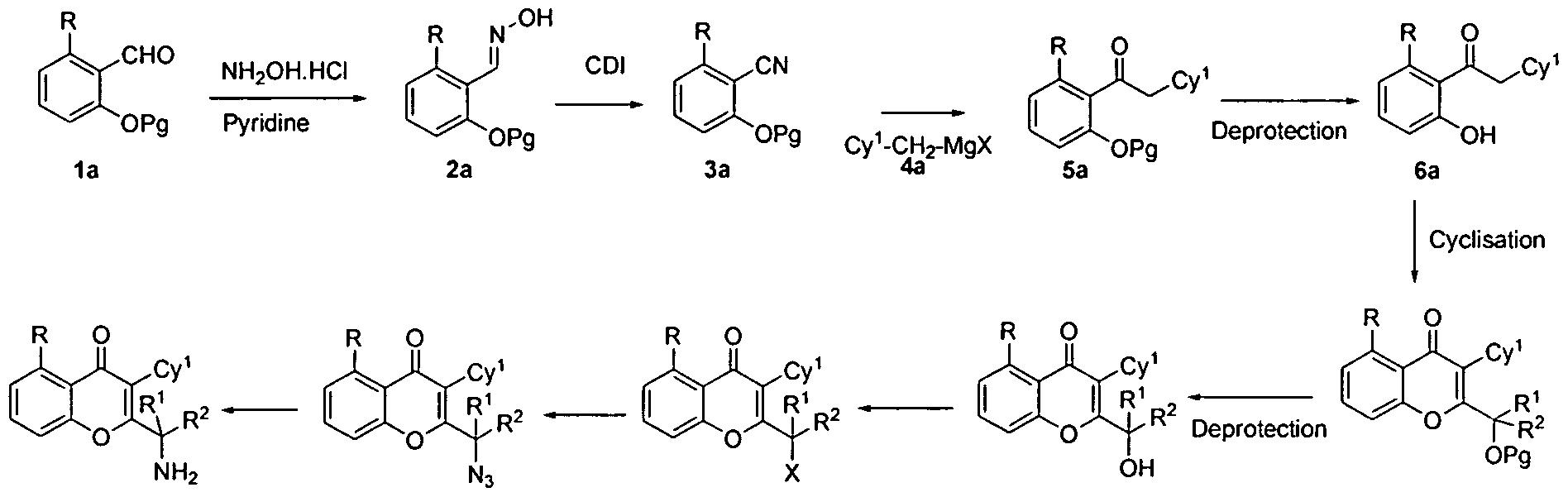
When high-throughput instruments first made their appearance, laboratories paid more attention to the accuracy of base-reading: Less accurate sequencing meant more data cleaning and resequencing (1). Now, new instrument designs have narrowed the differences, and test chemistry can have a comparatively large impact on analytical accuracy (Figure 1). The robustness of technical performance can also vary significantly depending upon specimen type. For example, LifeTechnologies’ sequencing platforms appear to be comparatively more tolerant of low DNA quality and concentration, which is an important consideration for fixed and processed tissues.

https://www.aacc.org/~/media/images/cln/articles/2015/october/carter_fig1_cln_oct15_ed.jpg
Figure 1 Comparison of Sequencing Chemistries
Sequence pile-ups of the same target sequence (2 large genes), all performed on the same analytical instrument. Results from 4 different chemistries, as designed and supplied by reagent manufacturers prior to optimization in the laboratory. Red lines represent limits of exons. Height of blue columns proportional to depth of coverage. In this case, the intent of the test design was to provide high depth of coverage so that reflex Sanger sequencing would not be necessary. Courtesy B. Sadikovic, U. of Western Ontario.
In addition, batching, robotics, workload volume patterns, maintenance contracts, software licenses, and platform lifetime affect the cost per analyte and per specimen considerably. Royalties and reagent contracts also factor into the cost of operating NGT: In some applications, fees for intellectual property can represent more than 50% of the bench cost of performing a given test, and increase substantially without warning.
Laboratories must also deal with the problem of obsolescence. Investing in a new platform brings the angst of knowing that better machines and chemistries are just around the corner. Laboratories are buying bigger pieces of equipment with shorter service lives. Before NGTs, major instruments could confidently be expected to remain current for at least 6 to 8 years. Now, a major instrument is obsolete much sooner, often within 2 to 3 years. This means that keeping it in service might cost more than investing in a new platform. Lease-purchase arrangements help mitigate year-to-year fluctuations in capital equipment costs, and maximize the value of old equipment at resale.
One Size Still Does Not Fit All
Laboratories face numerous technical considerations to optimize sequencing protocols, but the test has to be matched to the performance criteria needed for the clinical indication (2). For example, measuring response to treatment depends first upon the diagnostic recognition of mutation(s) in the tumor clone; the marker(s) then have to be quantifiable and indicative of tumor volume throughout the course of disease (Table 1).
As a result, diagnostic tests need to cover many different potential mutations, yet accurately identify any clinically relevant mutations actually present. On the other hand, tests for residual disease need to provide standardized, sensitive, and accurate quantification of a selected marker mutation against the normal background. A diagnostic panel might need 1% to 3% sensitivity across many different mutations. But quantifying early response to induction—and later assessment of minimal residual disease—needs a test that is reliably accurate to the 10-4 or 10-5 range for a specific analyte.
Covering all types of mutations in one diagnostic test is not yet possible. For example, subtyping of acute myeloid leukemia is both old school (karyotype, fluorescent in situ hybridization, and/or PCR-based or array-based testing for fusion rearrangements, deletions, and segmental gains) and new school (NGT-based panel testing for molecular mutations).
Chemistries that cover both structural variants and copy number variants are not yet in general use, but the advantages of NGTs compared to traditional methods are becoming clearer, such as in colorectal cancer (3). Researchers are also using cell-free DNA (cfDNA) to quantify residual disease and detect resistance mutations (4). Once a clinically significant clone is identified, enrichment techniques help enable extremely sensitive quantification of residual disease (5).
Validation and Quality Assurance
Beyond choosing a platform, two distinct challenges arise in bringing NGTs into the lab. The first is assembling the resources for validation and quality assurance. The second is keeping tests up-to-date as new analytes are needed. Even if a given test chemistry has the flexibility to add analytes without revalidating the entire panel, keeping up with clinical advances is a constant priority.
Due to their throughput and multiplexing capacities, NGT platforms typically require considerable upfront investment to adopt, and training staff to perform testing takes even more time. Proper validation is harder to document: Assembling positive controls, documenting test performance criteria, developing quality assurance protocols, and conducting proficiency testing are all demanding. Labs meet these challenges in different ways. Laboratory-developed tests (LDTs) allow self-determined choice in design, innovation, and control of the test protocol, but can be very expensive to set up.
Food and Drug Administration (FDA)-approved methods are attractive but not always an option. More FDA-approved methods will be marketed, but FDA approval itself brings other trade-offs. There is a cost premium compared to LDTs, and the test methodologies are locked down and not modifiable. This is particularly frustrating for NGTs, which have the specific attraction of extensive multiplexing capacity and accommodating new analytes.
IT and the Evolution of Molecular Oncology Reporting Standards
The options for information technology (IT) pipelines for NGTs are improving rapidly. At the same time, recent studies still show significant inconsistencies and lack of reproducibility when it comes to interpreting variants in array comparative genomic hybridization, panel testing, tumor expression profiling, and tumor genome sequencing. It can be difficult to duplicate published performances in clinical studies because of a lack of sufficient information about the protocol (chemistry) and software. Building bioinformatics capacity is a key requirement, yet skilled people are in short supply and the qualifications needed to work as a bioinformatician in a clinical service are not yet clearly defined.
Tumor biology brings another level of complexity. Bioinformatic analysis must distinguish tumor-specific variants from genomic variants. Sequencing of paired normal tissue is often performed as a control, but virtual normal controls may have intriguing advantages (6). One of the biggest challenges is to reproducibly interpret the clinical significance of interactions between different mutations, even with commonly known, well-defined mutations (7). For multiple analyte panels, such as predictive testing for breast cancer, only the performance of the whole panel in a population of patients can be compared; individual patients may be scored into different risk categories by different tests, all for the same test indication.
In large scale sequencing of tumor genomes, which types of mutations are most informative in detecting, quantifying, and predicting the behavior of the tumor over time? The amount and complexity of mutation varies considerably across different tumor types, and while some mutations are more common, stable, and clinically informative than others, the utility of a given tumor marker varies in different clinical situations. And, for a given tumor, treatment effect and metastasis leads to retesting for changes in drug sensitivities.
These complexities mean that IT must be designed into the process from the beginning. Like robotics, IT represents a major ancillary decision. One approach many labs choose is licensed technologies with shared databases that are updated in real time. These are attractive, despite their cost and licensing fees. New tests that incorporate proprietary IT with NGT platforms link the genetic signatures of tumors to clinically significant considerations like tumor classification, recommended methodologies for monitoring response, predicted drug sensitivities, eligible clinical trials, and prognostic classifications. In-house development of such solutions will be difficult, so licensing platforms from commercial partners is more likely to be the norm.
The Commercial Value of Health Records and Test Data
The future of cancer management likely rests on large-scale databases that link hereditary and somatic tumor testing with clinical outcomes. Multiple centers have such large studies underway, and data extraction and analysis is providing increasingly refined interpretations of clinical significance.
Extracting health outcomes to correlate with molecular test results is commercially valuable, as the pharmaceutical, insurance, and healthcare sectors focus on companion diagnostics, precision medicine, and evidence-based health technology assessment. Laboratories that can develop tests based on large-scale integration of test results to clinical utility will have an advantage.
NGTs do offer opportunities for net reductions in the cost of healthcare. But the lag between availability of a test and peer-evaluated demonstration of clinical utility can be considerable. Technical developments arise faster than evidence of clinical utility. For example, immunohistochemistry, estrogen receptor/progesterone receptor status, HER2/neu, and histology are still the major pathological criteria for prognostic evaluation of breast cancer at diagnosis, even though multiple analyte tumor profiling has been described for more than 15 years. Healthcare systems need a more concerted assessment of clinical utility if they are to take advantage of the promises of NGTs in cancer care.
Disruptive Advances
Without a doubt, “disruptive” is an appropriate buzzword in molecular oncology, and new technical advances are about to change how, where, and for whom testing is performed.
• Predictive Testing
Besides cost per analyte, one of the drivers for taking up new technologies is that they enable multiplexing many more analytes with less biopsy material. Single-analyte sequential testing for epidermal growth factor receptor (EGFR), anaplastic lymphoma kinase, and other targets on small biopsies is not sustainable when many more analytes are needed, and even now, a significant proportion of test requests cannot be completed due to lack of suitable biopsy material. Large panels incorporating all the mutations needed to cover multiple tumor types are replacing individual tests in companion diagnostics.
• Cell-Free Tumor DNA
Challenges of cfDNA include standardizing the collection and processing methodologies, timing sampling to minimize the effect of therapeutic toxicity on analytical accuracy, and identifying the most informative sample (DNA, RNA, or protein). But for more and more tumor types, it will be possible to differentiate benign versus malignant lesions, perform molecular subtyping, predict response, monitor treatment, or screen for early detection—all without a surgical biopsy.
cfDNA technologies can also be integrated into core laboratory instrumentation. For example, blood-based EGFR analysis for lung cancer is being developed on the Roche cobas 4800 platform, which will be a significant change from the current standard of testing based upon single tests of DNA extracted from formalin-fixed, paraffin-embedded sections selected by a pathologist (8).
• Whole Genome and Whole Exome Sequencing
Whole genome and whole exome tumor sequencing approaches provide a wealth of biologically important information, and will replace individual or multiple gene test panels as the technical cost of sequencing declines and interpretive accuracy improves (9). Laboratories can apply informatics selectively or broadly to extract much more information at relatively little increase in cost, and the interpretation of individual analytes will be improved by the context of the whole sequence.
• Minimal Residual Disease Testing
Massive resequencing and enrichment techniques can be used to detect minimal residual disease, and will provide an alternative to flow cytometry as costs decline. The challenge is to develop robust analytical platforms that can reliably produce results in a high proportion of patients with a given tumor type, despite using post-treatment specimens with therapy-induced degradation, and a very low proportion of target (tumor) sequence to benign background sequence.
The tumor markers should remain informative for the burden of disease despite clonal evolution over the course of multiple samples taken during progression of the clinical course and treatment. Quantification needs to be accurate and sensitive down to the 10-5 range, and cost competitive with flow cytometry.
• Point-of-Care Test Methodologies
Small, rapid, cheap, and single use point-of-care (POC) sequencing devices are coming. Some can multiplex with analytical times as short as 20 minutes. Accurate and timely testing will be possible in places like pharmacies, oncology clinics, patient service centers, and outreach programs. Whether physicians will trust and act on POC results alone, or will require confirmation by traditional laboratory-based testing, remains to be seen. However, in the simplest type of application, such as a patient known to have a particular mutation, the advantages of POC-based testing to quantify residual tumor burden are clear.
Conclusion
Molecular oncology is moving rapidly from an esoteric niche of diagnostics to a mainstream, required component of integrated clinical laboratory services. While NGTs are markedly reducing the cost per analyte and per specimen, and will certainly broaden the scope and volume of testing performed, the resources required to choose, install, and validate these new technologies are daunting for smaller labs. More rapid obsolescence and increased regulatory scrutiny for LDTs also present significant challenges. Aligning test capacity with approved clinical indications will require careful and constant attention to ensure competitiveness.
References
1. Liu L, Li Y, Li S, et al. Comparison of next-generation sequencing systems. J Biomed Biotechnol 2012; doi:10.1155/2012/251364.
2. Brownstein CA, Beggs AH, Homer N, et al. An international effort towards developing standards for best practices in analysis, interpretation and reporting of clinical genome sequencing results in the CLARITY Challenge. Genome Biol 2014;15:R53.
3. Haley L, Tseng LH, Zheng G, et al. Performance characteristics of next-generation sequencing in clinical mutation detection of colorectal cancers. [Epub ahead of print] Modern Pathol July 31, 2015 as doi:10.1038/modpathol.2015.86.
4. Butler TM, Johnson-Camacho K, Peto M, et al. Exome sequencing of cell-free DNA from metastatic cancer patients identifies clinically actionable mutations distinct from primary disease. PLoS One 2015;10:e0136407.
5. Castellanos-Rizaldos E, Milbury CA, Guha M, et al. COLD-PCR enriches low-level variant DNA sequences and increases the sensitivity of genetic testing. Methods Mol Biol 2014;1102:623–39.
6. Hiltemann S, Jenster G, Trapman J, et al. Discriminating somatic and germline mutations in tumor DNA samples without matching normals. Genome Res 2015;25:1382–90.
7. Lammers PE, Lovly CM, Horn L. A patient with metastatic lung adenocarcinoma harboring concurrent EGFR L858R, EGFR germline T790M, and PIK3CA mutations: The challenge of interpreting results of comprehensive mutational testing in lung cancer. J Natl Compr Canc Netw 2015;12:6–11.
8. Weber B, Meldgaard P, Hager H, et al. Detection of EGFR mutations in plasma and biopsies from non-small cell lung cancer patients by allele-specific PCR assays. BMC Cancer 2014;14:294.
9. Vogelstein B, Papadopoulos N, Velculescu VE, et al. Cancer genome landscapes. Science 2013;339:1546–58.
10. Heitzer E, Auer M, Gasch C, et al. Complex tumor genomes inferred from single circulating tumor cells by array-CGH and next-generation sequencing. Cancer Res 2013;73:2965–75.
11. Healy B. BRCA genes — Bookmaking, fortunetelling, and medical care. N Engl J Med 1997;336:1448–9.
Like this:
Like Loading...
Read Full Post »



















![Anti-A[beta] antibody 3D6 reduces amyloid pathology but aggravates neuronal dysfunction.](https://i0.wp.com/www.nature.com/neuro/journal/vaop/ncurrent/carousel/nn.4163-F1.jpg?resize=405%2C200)
![Worsening of neuronal dysfunction by anti-A[beta] antibodies can occur independently of the effects on A[beta] pathology.](https://i0.wp.com/www.nature.com/neuro/journal/vaop/ncurrent/carousel/nn.4163-F2.jpg?resize=389%2C200)