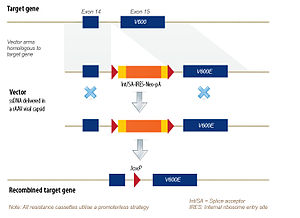
Targeted gene modification
Larry H Bernstein, MD, FCAP, Curator
Leaders in Pharmaceutical Intelligence

Article ID #183: Targeted gene modification. Published on 9/8/2015
WordCloud Image Produced by Adam Tubman
Series E. 2: 7.8
Mario R. Capecchi won a 2007 Nobel Prize for his work on targeted gene modification.
Born in Italy in 1937, scientist Mario R. Capecchi emigrated to the United States after World War II and later became a geneticist and professor. His groundbreaking work on targeted gene modification won him a Nobel Prize in 2007.
The Making of a Scientist II
In 1996, as a Kyoto Prize laureate, I was asked to write an autobiographical sketch of my early upbringing. Through this exercise, shared by all of the laureates, the hope was to uncover potential influences or experiences that may have been key to fostering the creative spirit within us. In my own case, what I saw was that, despite the complete absence of an early nurturing environment, the intrinsic drive to make a difference in our world is not easily quenched and that given an opportunity, early handicaps can be overcome and dreams achieved. This was intended as a message of hope for those who have struggled early in their lives. As I have previously noted, our ability to identify the genetic and environmental factors that contribute to talents such as creativity are too complex for us to currently predict. In the absence of such wisdom our only recourse is to provide all children with the opportunities to pursue their passions and dreams. Our understanding of human development is too meager to allow us to predict the next Beethoven, Modigliani, or Martin Luther King.
The content of the autobiographical sketch was based on my own memories, on conversations with my aunt and uncle, who raised me once I arrived in the United States, and on conversations with my mother. Because of the added exposure resulting from the winning of the Nobel Prize, I have received letters from people who knew me in Italy during those formative early years. In addition members of the press have taken an interest in my story and have sought independent corroboration. An amazing and wonderful surprise is that they have discovered a half-sister of whom I was completely unaware. She is two years younger than I, and was given up for adoption before she was one year old. Most recently I had the opportunity to meet my half-sister. She was a very nice person, as a sister should be. I am grateful for all of these new sources of information and revelation. Where appropriate, I will weave the new information into this retelling of my story.
Autobiographical Sketch
I was born in Verona, Italy on October 6, 1937. Fascism, Nazism, and Communism were raging through the country. My mother, Lucy Ramberg, was a poet; my father, Luciano Capecchi, an officer in the Italian Air Force. This was a time of extremes, turmoil and juxtapositions of opposites. They had a passionate love affair, and my mother wisely chose not to marry him. This took a great deal of courage on her part. It embittered my father.
 |
| Figure 1. A photograph of my mother, Lucy Ramberg, at age 19. |
I have only a few pictures of my mother. She was a beautiful woman with a passion for languages and a flair for the dramatic (see Figure 1). This picture was taken when she was 19. She grew up, with her two brothers, in a villa in Florence, Italy. There were magnificent gardens, a nanny, gardeners, cooks, house cleaners, and private tutors for languages, literature, history, and the sciences. She was fluent in half a dozen languages. Her father, Walter Ramberg, was an archeologist specializing in Greek antiquities, born and trained in Germany. Her mother was a painter born and raised in Oregon, USA. In her late teens, my grandmother, Lucy Dodd, packed up her steamer trunks and sailed with her mother from Oregon to Florence, Italy, where they settled. My grandmother was determined to become a painter. This occurred near the end of the 19th century, a time when young women were not expected to set off on their own with strong ambitions of developing their own careers.
 |
| Figure 2. A painting done by my grandmother, Lucy Dodd Ramberg, of her three children, left to right, Edward, Lucy, and Walter. It was painted at their villa in Florence, Italy in 1913. |
 |
| Figure 3. A painting by Lucy Dodd Ramberg of my mother, Lucy, and uncle Edward having tea at the villa in Florence, Italy (1913). |
My grandmother became a very gifted painter. Let me share with you a couple of her paintings, which also illustrate the young lives of her children. These paintings are very large, approximately seven feet by five feet. The first painting (Figure 2) is the center panel of a triptych depicting my mother and her two brothers Walter and Edward (both of whom became physicists) surrounded by olive trees at the villa in Florence. The influence of the French impressionist painters is evident. The second painting (Figure 3) is of my mother, age 8, and her younger brother Edward, age 6, having a tea party, again at the villa in Florence. Their father, the German archeologist, was killed as a young man in World War I. My grandmother finished raising her three children on her own by painting, mostly portraits, and by converting the family villa into a finishing school for young women, primarily from the United States.
 |
| Figure 4. A photograph of the chalet where my mother and I lived in Wolfgrübben just north of Bolzano, Italy. In the foreground is my mother, Lucy. |
My mother’s love and passion was poetry. She published in German. She received her university training at the Sorbonne in Paris and was a lecturer at that university in literature and languages. At that time, she joined with a group of poets, known as the Bohemians, who were prominent for their open opposition to Fascism and Nazism. In 1937, my mother moved to the Tyrol, the Italian Alps. Figure 4 shows the chalet north of Bolzano, in Wolfgrübben, with my mother in the foreground. We lived in this chalet until I was 3½ years old. In the spring of 1941, German officers came to our chalet and arrested my mother. This is one of my earliest memories. My mother had taught me to speak both Italian and German, and I was quite aware of what was happening. I sensed that I would not see my mother again for many years, if ever. She was incarcerated as a political prisoner in Germany.
I have believed that her place of incarceration was Dachau. This was based on conversations with my uncle Edward, my mother’s younger brother. During World War II, my uncle lived in the United States. Throughout these war years, he made many attempts to locate where my mother was being held. The most reliable information indicated that the location was near Munich. Dachau is located near Munich and was built to hold political prisoners. My mother survived her captivity, but after the war, despite my prodding, she refused to talk about her war experiences.
Reporters from the Associated Press (AP) have found records that my mother was indeed a prisoner during the war in Germany. In fact, they have found records of German interest in my mother’s political activities preceding 1939. In that year, they had her arrested by the Italian authorities and jailed in Perugia and subsequently released. However, the AP reporters did not find records indicating that my mother was incarcerated in Dachau. Though Germans were noted for their meticulous record keeping, it would be difficult now to evaluate the accuracy of the existing war records, particularly for cases where data is missing. It is clear, however, that exactly where in Germany my mother was held has not yet been determined. Regardless of which prison camp was involved, her experiences were undoubtedly more horrific than mine. She had aged beyond recognition during those five years of internment. Following her release, though she lived until she was 82 years old, she never psychologically recovered from her wartime experiences.
My mother had anticipated her arrest by German authorities. Prior to their arrival, she had sold most of her possessions and gave the proceeds to an Italian peasant family in the Tyrol so that they could take care of me. I lived on their farm for one year. It was a very simple life. They grew their own wheat, harvested it, and took it to the miller to be ground. From the flour they made bread which they took to the baker to be baked. During this time, I spent most of my time with the women of the farm. In the late fall, the grapes were harvested by hand and put into enormous wooden vats. The children, including me, stripped, jumped into the vats and mashed the grapes with our feet. We became squealing masses of purple energy. I still remember the pungent odor and taste of the fresh grapes. Most recently, members of the Dolomiten Press have located this farm and I had the opportunity to visit it. It is still owned by the same family that occupied it when I was there. The old farm house has been taken down and a new one erected. However, the pictures of the old farm house, as well as the surrounding land are remarkably consistent with my memories.
World War II was now fully under way. The American and British forces had landed in Southern Italy and were proceeding northward. Bombings of northern Italian cities were a daily occurrence. As constant reminders of the war, curfews and blackouts were in effect every night; no lights were permitted. In the night we could hear the drone of presumed American and British reconnaissance planes which we nicknamed “Pepe.” One hot afternoon, American planes swooped down from the sky and began machine gunning the peasants in the fields. A senseless exercise. A bullet grazed my leg, fortunately not breaking any bones. I still have the scar, which, many years later my daughter proudly had me display to her third-grade class in Utah.
For reasons that have never been clear to me, my mother’s money ran out after one year and, at age 4½, I set off on my own. I headed south, sometimes living in the streets, sometimes joining gangs of other homeless children, sometimes living in orphanages, and most of the time being hungry. My recollections of those four years are vivid but not continuous, rather like a series of snapshots. Some of them are brutal beyond description, others more palatable.
There are records in the archives of Ritten, a region of the Southern Alps of Italy, that I left Bozen to go to Reggio Emilia on July 18, 1942. AP reporters exploring this history have suggested that my father came to the farm, picked me up, and that we went together to Reggio Emilia where he was living. I have no memory of his coming to the farm, nor of having travelled with him to Reggio Emilia. I have recently received a letter from a man who remembers me as the youngest member of his street gang operating in Bolzano, which is on the way to Reggio Emilia.
I did end up in Reggio Emilia, which is approximately 160 miles south of Bolzano. I knew that my father lived in Reggio Emilia and I have previously noted that I had lived with him a couple of times from 1942-1946, for a total period of approximately three weeks. The question has been raised why I didn’t live with him for a much longer period. The reason was that he was extremely abusive. Amidst all of the horrors of war, perhaps the most difficult for me to accept as a child was having a father who was brutal to me.
Recently, I have also received a very nice letter from the priest in Reggio Emilia who ran the orphanage in which I was eventually placed. I remember him because he was one of the very few men I encountered in Reggio Emilia who showed compassion for the children and took an interest in me. I am surprised, but pleased, that after all these years he still remembers me among the thousands of children he was responsible for over the years. Further, I believe I was at that orphanage for only several months, the first time in the fall of 1945, after which I ran away, followed by a second period, in the same orphanage, in the spring of 1946. But his memory is genuine, for he recounts incidents consistent with my memories that could only have been known through our common experience.
In the spring of 1945, Munich was liberated by the American troops. My mother had survived her captivity and set out to find me. In October 1946, she succeeded. As an example of her flair for the dramatic, she found me on my ninth birthday, and I am sure that this was by design. I did not recognize her. In five years she had aged a lifetime. I was in a hospital when she found me. All of the children in this hospital were there for the same reasons: malnutrition, typhoid, or both. The prospects for most of those children ever leaving that hospital were slim because they had no nourishing food. Our daily diet consisted of a bowl of chicory coffee and a small crust of old bread. I had been in that hospital in Reggio Emilia for what seemed like a year. Scores of beds lined the rooms and corridors of the hospital, one bed touching the next. There were no sheets or blankets. It was easier to clean without them. Our symptoms were monotonously the same. In the morning we awoke fairly lucid. The nurse, Sister Maria, would take our temperature. She promised me that if I could go through one day without a high fever, I could leave the hospital. She knew that without any clothes I was not likely to run away. By late morning, the high, burning fever would return and we would pass into oblivion. Consistent with the diagnosis of typhoid, many years later I received a typhoid/paratyphoid shot, went into shock, and passed out.
 |
| Figure 5. A photograph of my uncle Edward Ramberg working in his laboratory at RCA Princeton, New Jersey. |
The same day that my mother arrived at the hospital, she bought me a full set of new clothes, a Tyrolean outfit complete with a small cap with a feather in it. I still have the hat. We went to Rome to process papers, where I had my first bath in six years, and then on to Naples. My mother’s younger brother, Edward, had sent her money to buy two boat tickets to America. I was expecting to see roads paved with gold in America. As it turned out, I found much more: opportunities.
On arriving in America, my mother and I lived with my uncle and aunt, Edward and Sarah Ramberg. Edward, my mother’s younger brother was a brilliant physicist. He was a Ph.D. student in quantum mechanics with Arnold Sommerfeld and translated one of Sommerfeld’s major texts into English. Among Edward’s many contributions was his discovery of how to focus electrons, knowledge which he used in helping to build the first electron microscope at RCA. Edward’s books on electron optics have been published in many languages. During my visit to Japan to celebrate the Kyoto Prize, several Japanese physicists approached me to express how grateful they were for my uncle’s texts from which they learned electron optics. Another achievement, of which he was less proud was being a principal contributor to the development of both black and white and color television. While I grew up in his home, television was not allowed. Figure 5 shows a photograph of my uncle working in his laboratory.
My aunt and uncle were Quakers and they did not support violence as solutions to political problems anywhere in the world. During World War II, my uncle did alternative service rather than bear arms. He worked in a mental institution in New Hampshire, cleared swamps in the south, and was a guinea pig for the development of vaccines against tropical diseases. After the war he settled in a commune in Pennsylvania, called Bryn Gweled, which he helped found. People of all races and religious affiliations were welcomed in this community. It was a marvelous place for children: it contained thick woods for exploration and had communal activities of all kinds – painting, dance, theater, sports, electronics, and many sessions devoted to the discussion of the major religious philosophies of the world. Every week there were communal work parties, putting in roads, phone lines, and electrical lines, building a community center and so on.
The contrast between living primarily alone in the streets of Italy and living in an intensely cooperative and supportive community in Pennsylvania was enormous. Time was needed for healing and for erasing the images of war from my mind. I remember that for many years after coming to the United States I would go to sleep tossing and turning with such force that by morning the sheets were torn and the bed frame broken. This activity disturbed my aunt and uncle to the extent that Sarah would take me from one child psychologist or psychiatrist, to another. These professionals were not very helpful, but the support of the community was. The nightly activity eventually subsided. There may be lessons to be learned from such experiences for the treatment of the children from Darfur, the Congo, and now Kenya.
Sarah and Edward took on the challenge of converting me into a productive human being. This, I am sure, was a very formidable task. I had received little or no formal education or training for living in a social environment. Quakers do not believe in frills, but rather in a life of service. My aunt and uncle taught me by example. I was given few material goods, but every opportunity to develop my mind and soul. What I made of myself would be entirely up to me. The day after I arrived in America, I went to school. I started in the third grade in the Southampton public school system. Sarah also took on the task of teaching me to read, starting from the very beginning.
The first task was to learn English. I had a marvelous third grade teacher. She was patient and encouraging. The class was studying Holland, so I started participation in class functions by painting a huge mural on butcher block paper with tulips, windmills, children ice skating, children in Dutch costumes, and ships. It was a collage of activities and colors. This did not require verbal communication.
I was a good, but not serious, student in grade school and high school. Academics came easily to me. I attended an outstanding high school, George School, a Quaker school north of Philadelphia. The teachers were superb, challenging, enthusiastic, competent, and caring. They enjoyed teaching. The campus was also magnificent, particularly in the spring when the cherry and dogwood trees were bursting with blossoms. An emphasis on Quaker beliefs permeated all of the academic and sports programs. A favorite period for many, including me, was Quaker meeting, a time set aside for silent meditation, and taking stock of where we were going. My wife and I sent our daughter to George School for her own last two years in high school so that she might also benefit from the personal virtues it promotes, and we think she has.
Sports were very important to me at George School, and physical activity has remained an important activity for me to this day. I played varsity football, soccer, and baseball, and wrestled. I was particularly proficient at wrestling. I enjoyed the drama of a single opponent, as well as the physical and psychological challenges of the sport. After George School, I went to Antioch, a small liberal arts college in Ohio.
At Antioch College I became a serious student, converting to academics all of the energy I had previously devoted to sports. Coming from George School, I carried the charge of making this a better, more equitable world for all people. Most of the problems appeared to be political, so I started out at Antioch majoring in political science. However, I soon became disillusioned with political science since there appeared to be little science to this discipline, so I switched to the physical sciences – physics and chemistry. I found great pleasure in the simplicity and elegance of mathematics and classical physics. I took almost every mathematics, physics, and chemistry course offered at Antioch, including Boolean algebra and topology, electrodynamics, and physical chemistry.
Although I found physics and mathematics intellectually satisfying, it was becoming apparent that what I was learning came from the past. The newest physics that was taught at Antioch was quantum mechanics, a revolution that had occurred in the 1920’s and earlier. Also, many frontiers of experimental physics, particularly experimental particle physics, were requiring the use of larger and larger accelerators, which involved bigger and bigger teams of scientists and support groups to execute the experiments. I was looking for a science in which the individual investigator had a more intimate, hands-on involvement with the experiments. Fortunately, Antioch had an outstanding work-study program; one quarter we studied on campus, the next was spent working on jobs related to our fields of interest. The jobs, in my case laboratory jobs, were maintained all over the country, and every three months we packed up our bags and set off for a new city and a new work experience. So one quarter off I went to Boston and the Massachusetts Institute of Technology (MIT).
There I encountered molecular biology as the field was being born (late 1950’s). This was a new breed of science and scientist. Everything was new. There were no limitations. Enthusiasm permeated this field. Devotees from physics, chemistry, genetics, and biology joined its ranks. The common premises were that the most complex biological phenomena could, with persistence, be understood in molecular terms and that biological phenomena observed in simple organisms, such as viruses and bacteria, were mirrored in more complex ones. Implicit corollaries to this premise were that whatever was learned in one organism was likely to be directly relevant to others and that similar approaches could be used to study biological phenomena in many organisms. Genetics, along with molecular biology, became the principal means for dissecting complex biological phenomena into workable subunits. Soon all organisms came under the scrutiny of these approaches.
I became a product of the molecular biology revolution. The next generation. As an Antioch college undergraduate, I worked several quarters in Alex Rich’s laboratory at MIT. He was an x-ray crystallographer, with very broad interests in molecular biology. While at MIT I was also fortunate to be influenced bySalvador Luria, Cyrus Leventhal and Boris Magasanik, through courses, seminars, and personal discussions. At that time Sheldon Penman and Jim Darnell were also working in Alex Rich’s laboratory. When placed in the same room, these two were particularly boisterous, providing comic relief to the fast moving era.
After Antioch, I set off for what I perceived as the “Mecca” of molecular biology, Harvard University. I had interviewed with Professor James D. Watson, of “Watson and Crick” fame, and asked him where should I do my graduate studies. His reply was curt and to the point: “Here. You would be fucking crazy to go anywhere else.” The simplicity of the message was very persuasive.
 |
| Figure 6. A photograph of James D. Watson. |
James D. Watson had a profound influence on my career (see Figure 6). He was my mentor. He did not teach me how to do molecular biology; because of my Antioch job experiences, I had already become a proficient experimenter. Jim instead taught me the process of science – how to extract the questions in a field that are critical to it and at the same time approachable through current technology. As an individual, he personified molecular biology, and, as his students, we were its eager practitioners. His bravado encouraged self-confidence in those around him. His stark honesty made our quest for truth uncompromising. His sense of justice encouraged compassion. He taught us not to bother with small questions, for such pursuits were likely to produce small answers. At a critical time, when I was contemplating leaving Harvard as a faculty member and going to Utah, he, being familiar with my self-sufficiency, counseled me that I could do good science anywhere. The move turned out to be a good decision. In Utah I had the luxury to pursue long-term projects that were not readily possible at Harvard, which, in too many cases had become a bastion of short-term gratification.
Doing science in Jim’s laboratory was exhilarating. As a graduate student, I was provided with what appeared to be limitless resources. I could not be kept out of the laboratory. Ninety-hour weeks were common. The lab was filled with talented students, each working on his or her own set of projects. Represented was a mixture of genetics, molecular biology, and biochemistry. We were cracking the genetic code, determining how proteins were synthesized, and isolating and characterizing the enzymatic machinery required for transcription. At this time, Walter Gilbert was also working in Jim’s laboratory. He was then a member of the physics department, but had also been bitten by the molecular biology bug. Jim and Wally complemented each other brilliantly, because they approached science from very different perspectives. Jim was intuitive, biological; Wally quantitative, with a physicist’s perspective. They were both very competitive. As students, we received the benefit of both, but also their scrutiny. They were merciless, but fair. You had to have a tough hide, but you learned rigor, both with respect to your science and your presentations. Once you made it through Jim’s laboratory, the rest of the world seemed a piece of cake. It was excellent training. Despite the toughness, which at times was hard, Jim was extremely supportive. He also made sure that you, the student, received full credit for your work. Despite the fact that Jim was responsible for all of the resources needed to run his laboratory, if you did all of the work for a given paper, then you were the sole author on that paper. Among all of the laboratory heads in the world, I believe that Jim Watson was among very few in implementing this policy.
The summer before I started graduate school, Marshall Nirenberg had announced that polyU directs the synthesis of polyphenylalanine in a cell free protein synthesizing extract. That paper was a bombshell! I decided I would generate a cell-free extract capable of synthesizing real, functional proteins. Jim’s laboratory had started working on the RNA bacteriophage, R17. Its genome also served as messenger RNA to direct the synthesis of its viral proteins. That would be my message. The cell-free protein synthesizing extract worked beautifully. Authentic viral coat protein and replicase were shown to be synthesized in the extract1. Further, the coat protein was functional, it bound to a specific sequence of the R17 genome, thereby modulating the synthesis of the replicase. To this day, the high affinity of the viral coat protein for this RNA sequence is exploited as a general reporter system to track RNA trafficking within living cells and neuronal axons. In collaboration with Gary Gussin, also a graduate student in Jim’s laboratory, this system was used to determine the molecular mechanism of genetic suppression of nonsense mutations2. In collaboration with Jerry Adams, another graduate student in Jim’s laboratory, the system was also used to determine that initiation of the synthesis of all proteins in bacteria proceeded through the use of formyl-methionine-tRNA3,4. A similar mechanism is involved in the initiation of protein synthesis in all eukaryotic organisms. Finally, I used the same in vitro system to show that termination of protein synthesis unexpectedly utilized protein factors, rather than tRNA, to accomplish this end5,6. Jim Watson would later offer the very complimentary comment “that Capecchi accomplished more as a graduate student than most scientists accomplish in a lifetime.” It was, indeed, a productive time, but it wasn’t work; it was sheer joy.
While a graduate student in Jim’s laboratory, I was invited to become a junior fellow of the Society of Fellows at Harvard. Being a junior fellow was very special. The society’s membership, junior and senior fellows, represented a broad spectrum of disciplines; all the members were talented, and most of them were much more verbal than I. Social discourse centered around meals, prepared by an exquisite French chef and ending with fine brandy and Cuban cigars. Frequent guests at these dinners were the likes of Leonard Bernstein. Surreal maybe, but also very special.
 |
| Figure 7. A photograph of Karl G. Lark. |
From Jim’s laboratory, I joined the faculty in the Department of Biochemistry at Harvard Medical School, across the river in Boston. During my four years at Harvard Medical School I quickly rose through the ranks, but then, I unexpectedly decided to go to Utah. I was looking for something different. There were excellent scientists in the department I was in at Harvard Medical School, but the department was not built with synergy in mind. Each research group was an island onto itself. At that time, they were also unwilling to hire additional young faculty and thereby provide the department with a more youthful, energetic character. At the University of Utah, I would be joining a newly formed department that was being assembled by a very talented scientist and administrator, Karl G. Lark (Figure 7). He had excellent taste in scientists and a vision of assembling a faculty that would enjoy working together and striving together for excellence. I could be a participant in the growth of that department and help shape its character. Furthermore, the University’s administration, led then by President David P. Gardner, was in synchrony with this vision and a strong supporter. Gordon had already attracted Baldomero (Toto) Olivera, Martin Rechsteiner, Sandy Parkinson, and Larry Okun to Utah. After I arrived at Utah, we were able to bring to Utah such outstanding scientists as Ray Gesteland, John Roth, and Mary Beckerle. Utah also provided wide open space, an entirely new canvas upon which to create a new career (see Figures 8). These are views from one of the homes in Utah which I have shared with my wife, Laurie Fraser, and daughter, Misha. The air is clean, and I can look for long distances. The elements of nature are all around us. What a place to begin a new life!
 |
| Figure 8. Views from one of our homes in Utah and a photograph of my wife, Laurie Fraser, and daughter, Misha, just after she was born. Misha is now graduating from the University of California, Santa Cruz as an arts major. |
From Les Prix Nobel. The Nobel Prizes 2007, Editor Karl Grandin, [Nobel Foundation], Stockholm, 2008
This autobiography/biography was written at the time of the award and later published in the book series Les Prix Nobel/ Nobel Lectures/The Nobel Prizes. The information is sometimes updated with an addendum submitted by the Laureate.
Copyright © The Nobel Foundation 2007
Dr. Capecchi is a member of the National Academy of Sciences (1991) and the European Academy of Sciences (2002). He has won numerous awards, including the Bristol-Myers Squibb Award for Distinguished Achievement in Neuroscience Research (1992), the Gairdner Foundation International Award for Achievements in Medical Sciences (1993), the General Motors Corporation’s Alfred P. Sloan Jr. Prize for Outstanding Basic Science Contributions to Cancer Research (1994), the German Molecular Bioanalytics Prize, (1996), the Kyoto Prize in Basic Sciences (1996), the Franklin Medal for Advancing Our Knowledge of the Physical Sciences (1997), the Feodor Lynen Lectureship (1998), the Rosenblatt Prize for Excellence (1998), the Baxter Award for Distinguished Research in the Biomedical Sciences (1998), the Helen Lowe Bamberger Colby and John E. Bamberger Presidential Endowed Chair in the University of Utah Health Sciences Center (1999), lectureship in the Life Sciences for the Collège de France (2000), the Horace Mann Distinguished Alumni Award, Antioch College (2000), the Italian Premio Phoenix-Anni Verdi for Genetics Research Award (2000), the Spanish Jiménez-Diáz Prize (2001), the Pioneers of Progress Award (2001), the Albert Lasker Award for Basic Medical Research (2001), the National Medal of Science (2001), the John Scott Medal Award (2002), the Massry Prize (2002), the Pezcoller Foundation-AACR International Award for Cancer Research (2003), the Wolf Prize in Medicine (2002/03), the March of Dimes Prize in Developmental Biology (2005),and the Nobel Prize in Physiology and Medicine (2007) with Oliver Smithies and Martin Evans.
Research interests include: the molecular genetic analysis of early mouse development, neural development in mammals, production of murine models of human genetic diseases, gene therapy, homologous recombination and programmed genomic rearrangements in the mouse.
- The Capecchi Lab
- The Nobel Prize in Physiology or Medicine 2007
- The 2001 Lasker Award
- iBiology talk: The Birth of Gene Targeting
http://www.hhmi.org/news/making-scientist
Mario Capecchi received a Kyoto Prize from the Inamori Foundation in 1996. The lecture he delivered when he accepted the prize in Japan in November 1996 tells the story of his remarkable life. The text of the lecture has been edited for length.
Radoslav Bozov commented on Targeted gene modification
Targeted gene modification Larry H Bernstein, MD, FCAP, Curator Leaders in Pharmaceutical Intelligence Series E. 2: …
Larry, same thing, data redundancy of data mining issues, of what data is in reality of physics beyond nano space in time! Working on something that does not exits in space and time, but computable mass of ‘designed’ energy formulated systems: Hox gene does not exist: It is a piece of time we percive through some kind of imagination +1,
The data generated through m/z methods is space-time unaccurate! Guess what is double R doing here instead of double Y, wonder why miRNA are obejctive to polymer degradation process ??!! what are we really seeing is not what is really in there!
Score Expect Method Identities Positives Gaps
19.2 bits(38) 0.076 Composition-based stats. 8/17(47%) 10/17(58%) 0/17(0%)
Query 511 LTEDRRAFAARMAEIGE 527
LT DRR AR+ + E
Sbjct 38 LTRDRRYEVARLLNLTE 54
Breaking news about genomic engineering, T2DM and cancer treatments
Larry H. Bernstein, MD, FCAP, Curator