AHA, ACC Change in Requirement for Surgical Support: Class IIb -> Class III, Level of Evidence A: Supports Nonemergent PCI without Surgical Backup (Change of class IIb, Level of Evidence B).
Larry H Bernstein, MD, FCAP, Author, Curator, Volumes 1,2,3,4,5,6 Co-Editor and Author, Volume Two & Five, Co-Editor and Justin Pearlman, MD, PhD, FACC, Content Consultant to Six-Volume e-SERIES A: Cardiovascular Diseases

Article ID #68: AHA, ACC Change in Requirement for Surgical Support for PCI Performance: Class IIb -> Class III, Level of Evidence A: Support Nonemergent PCI without Surgical Backup (Change of class IIb, Level of evidence B). Published on 7/17/2013
WordCloud Image Produced by Adam Tubman
Voice of content consultant: Justin Pearlman, MD, PhD, FACC
The American Heart Association (AHA) and the American College of Cardiology (ACC) have convened teams of experts to summarize evidence and opinion regarding a wide range of decisions relevant to cardiovascular disease. The system accounts for some of the short comings of “evidence based medicine” by allowing for expert opinion in areas where evidence is not sufficient. The main argument for evidence-based medicine is the existence of surprises, where a plausible decision does not actually appear to work as desired when it is tested. A major problem with adhesion to evidence based medicine is that it can impede adaptation to individual needs (we are all genetically and socially/environmentally unique) and impede innovation. Large studies carry statistical weight but do not necessary consider all relevant factors. Commonly, the AFFIRM trial is interpreted as support that rate control suffices for most atrial fibrillation (AFIB), but half of those randomized to rhythm control were taken off anticoagulation without teaching patients to check their pulse daily for recurrence of AFIB. Thus the endorsed “evidence” may have more to do with the benefits of anticoagulation for both persisting and recurring AFIB and rhythm control may yet prove better than rate control. However, with wide acceptance of a particular conclusion, randomizing to another treatment may be deemed unethical, or may simply not get a large trial due to lack of economic incentive, leaving only the large trial products as the endorsed options. A medication without patent protection, such as bismuth salts for H Pylori infection, lacks financial backing for large trials.
The American Heart Association Evidence-Based Scoring System
Classification of Recommendations
● Class I: Conditions for which there is evidence, general
agreement, or both that a given procedure or treatment is
useful and effective.
● Class II: Conditions for which there is conflicting evidence,
a divergence of opinion, or both about the usefulness/
efficacy of a procedure or treatment.
● Class IIa: Weight of evidence/opinion is in favor of
usefulness/efficacy.
● Class IIb: Usefulness/efficacy is less well established by
evidence/opinion.
● Class III: Conditions for which there is evidence, general
agreement, or both that the procedure/treatment is not useful/
effective and in some cases may be harmful.
Level of Evidence
● Level of Evidence A: Data derived from multiple randomized
clinical trials
● Level of Evidence B: Data derived from a single randomized
trial or nonrandomized studies
● Level of Evidence C: Consensus opinion of experts
Circulation 2006 114: 1761 – 1791.
Assessment of Coronary Artery Disease by Cardiac Computed Tomography
A Scientific Statement From the American Heart Association Committee on Cardiovascular Imaging and Intervention, Council on Cardiovascular Radiology and Intervention, and Committee on Cardiac Imaging, Council on Clinical Cardiology
- Matthew J. Budoff, MD, FAHA;
- Stephan Achenbach, MD;
- Roger S. Blumenthal, MD, FAHA;
- J. Jeffrey Carr, MD, MSCE;
- Jonathan G. Goldin, MD, PhD;
- Philip Greenland, MD, FAHA;
- Alan D. Guerci, MD;
- Joao A.C. Lima, MD, FAHA;
- Daniel J. Rader, MD, FAHA;
- Geoffrey D. Rubin, MD;
- Leslee J. Shaw, PhD;
- Susan E. Wiegers, MD
Action Points
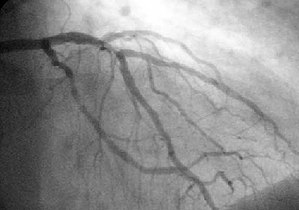
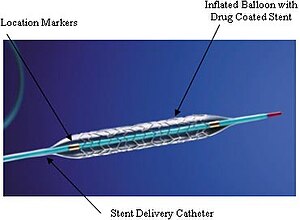
- Patients with indications for nonemergency PCI who presented at hospitals without on-site cardiac surgery, were randomly assigned to undergo PCI at a hospital without on-site cardiac surgery or at a hospital with on-site cardiac surgery.
- The rates of death, myocardial infarction, repeat revascularization, and stroke did not differ significantly between the groups.
- Community hospitals without surgical services can safely perform percutaneous coronary intervention (PCI) in low-risk patients — and not refuse higher-risk patients either, the MASS COMM trial found.
Summary
- The co-primary endpoint of major adverse cardiac events (MACE) at 30 days occurred at a rate of 9.5% in the 10 hospitals without surgical backup versus 9.4% in the seven hospitals with onsite surgery (P<0.001 for noninferiority), Alice K. Jacobs, MD, of Boston University School of Medicine, and colleagues found.
- The other co-primary endpoint of MACE at 12 months was also significant, occurring in 17.3% of patients in hospitals without backup versus 17.8% in centers with surgical services (P<0.001 for non-inferiority), they reported in the study published online by the New England Journal of Medicine. The findings were also reported at the American College of Cardiology meeting.
Study Characteristics and Results
Primary Endpoints
- death
- myocardial infarction
- repeat revascularization
- stroke
Rate of stent thrombosis at 30 days
CPORT – E trial
MASS COMM study
To further the evidence, Jacobs and colleagues in 2006 had designed and carried out the Randomized Trial to Compare Percutaneous Coronary Intervention between Massachusetts Hospitals with Cardiac Surgery On-Site and Community Hospitals without Cardiac Surgery On-Site (MASS COMM) in collaboration with the Massachusetts Department of Public Health who collaborated to obtain “evidence on which to base regulatory policy decisions about performing non-emergent PCI in hospitals without on-site cardiac surgery.”
- Hospitals without backup surgery were required to perform at least 300 diagnostic catheterizations per year, and operators were mandated to have performed a minimum of 75 PCI procedures per year.
- The researchers randomized 3,691 patients to each arm in a 3:1 ratio (without/with backup). The median follow-up was about 1 year.
- The median age of patients was 64, one-third were women, and 92% were white. Both groups had similar median ejection fractions at baseline (55%).
- The mean number of vessels treated was 1.17 and most patients (84%) had one vessel treated. The mean number of lesions treated was 1.45 and most patients (67%) had one lesion treated.
The indications for PCI were:
- all-cause death
- repeat revascularization
- stroke
- definite or probable stent thrombosis
- major vascular complications
- the relative risks at 30 days and 12 months “were consistent with those of the primary results” (RR 1.02 and 0.98, respectively).
However, they cautioned that new sites perhaps should be monitored as they gain experience.
- rates of procedural success,
- proportion with complete revascularization, or
- the proportion of guideline-indicated appropriate lesions for PCI.
- loss of data for 13% of patients, the
- exclusion of some patients for certain clinical and anatomical features, and
- not having the power to detect non-inferiority in the separate components of the primary endpoint, researchers wrote.
Cardio Notes: Score Predicts PCI Readmission
Published: Jul 15, 2013

A simple calculation of patient variables before PCI may help stem the tide of readmission within the first month. Also this week, two blood pressure drugs that benefit diabetics and imaging cardiac sympathetic innervation.
Pre-PCI Factors Predict Return Trip
A new 30-day readmission risk prediction model for patients undergoing percutaneous coronary intervention (PCI) showed it’s possible to predict risk using only variables known before PCI, according to a study published online in Circulation: Cardiovascular Quality and Outcomes.
After multivariable adjustment, the 10 pre-PCI variables that predicted 30-day readmission were older age (mean age 68 in this study), female sex, insurance type (Medicare, state, or unknown), GFR category (less than 30 and 30-60 mL/min per 1.73m2), current or history of heart failure, chronic lung disease, peripheral vascular disease, cardiogenic shock at presentation, admit source (acute and non-acute care facility or emergency department), and previous coronary artery bypass graft surgery.
Additional significant variables post-discharge that predicted 30-day readmission were beta-blocker prescribed at discharge, post-PCI vascular or bleeding complications, discharge location, African American race, diabetes status and modality of treatment, any drug-eluting stent during the index procedure, and extended length of stay.
A risk score calculator using the pre-PCI variables will be available online soon, according to Robert W. Yeh, MD, MSc, of Massachusetts General Hospital in Boston, and colleagues.
- Ischemic Preconditioning Reduces Myocardial Necrosis in Elective PCI – TCTMD (pharmaceuticalintelligence.com)
- Too much blood: Researchers fear the ‘gift of life’ may sometimes endanger it (o.canada.com)
- Representatives of The Valley Hospital Heart and Vascular Institute Recognized by the American Heart Association (paramuspost.com)