Artificial Intelligence and Cardiovascular Disease
Reporter and Curator: Dr. Sudipta Saha, Ph.D.

3.3.18 Artificial Intelligence and Cardiovascular Disease, Volume 2 (Volume Two: Latest in Genomics Methodologies for Therapeutics: Gene Editing, NGS and BioInformatics, Simulations and the Genome Ontology), Part 2: CRISPR for Gene Editing and DNA Repair
Cardiology is a vast field that focuses on a large number of diseases specifically dealing with the heart, the circulatory system, and its functions. As such, similar symptomatologies and diagnostic features may be present in an individual, making it difficult for a doctor to easily isolate the actual heart-related problem. Consequently, the use of artificial intelligence aims to relieve doctors from this hurdle and extend better quality to patients. Results of screening tests such as echocardiograms, MRIs, or CT scans have long been proposed to be analyzed using more advanced techniques in the field of technology. As such, while artificial intelligence is not yet widely-used in clinical practice, it is seen as the future of healthcare.
The continuous development of the technological sector has enabled the industry to merge with medicine in order to create new integrated, reliable, and efficient methods of providing quality health care. One of the ongoing trends in cardiology at present is the proposed utilization of artificial intelligence (AI) in augmenting and extending the effectiveness of the cardiologist. This is because AI or machine-learning would allow for an accurate measure of patient functioning and diagnosis from the beginning up to the end of the therapeutic process. In particular, the use of artificial intelligence in cardiology aims to focus on research and development, clinical practice, and population health. Created to be an all-in-one mechanism in cardiac healthcare, AI technologies incorporate complex algorithms in determining relevant steps needed for a successful diagnosis and treatment. The role of artificial intelligence specifically extends to the identification of novel drug therapies, disease stratification or statistics, continuous remote monitoring and diagnostics, integration of multi-omic data, and extension of physician effectivity and efficiency.
Artificial intelligence – specifically a branch of it called machine learning – is being used in medicine to help with diagnosis. Computers might, for example, be better at interpreting heart scans. Computers can be ‘trained’ to make these predictions. This is done by feeding the computer information from hundreds or thousands of patients, plus instructions (an algorithm) on how to use that information. This information is heart scans, genetic and other test results, and how long each patient survived. These scans are in exquisite detail and the computer may be able to spot differences that are beyond human perception. It can also combine information from many different tests to give as accurate a picture as possible. The computer starts to work out which factors affected the patients’ outlook, so it can make predictions about other patients.
In current medical practice, doctors will use risk scores to make treatment decisions for their cardiac patients. These are based on a series of variables like weight, age and lifestyle. However, they do not always have the desired levels of accuracy. A particular example of the use of artificial examination in cardiology is the experimental study on heart disease patients, published in 2017. The researchers utilized cardiac MRI-based algorithms coupled with a 3D systolic cardiac motion pattern to accurately predict the health outcomes of patients with pulmonary hypertension. The experiment proved to be successful, with the technology being able to pick-up 30,000 points within the heart activity of 250 patients. With the success of the aforementioned study, as well as the promise of other researches on artificial intelligence, cardiology is seemingly moving towards a more technological practice.
One study was conducted in Finland where researchers enrolled 950 patients complaining of chest pain, who underwent the centre’s usual scanning protocol to check for coronary artery disease. Their outcomes were tracked for six years following their initial scans, over the course of which 24 of the patients had heart attacks and 49 died from all causes. The patients first underwent a coronary computed tomography angiography (CCTA) scan, which yielded 58 pieces of data on the presence of coronary plaque, vessel narrowing and calcification. Patients whose scans were suggestive of disease underwent a positron emission tomography (PET) scan which produced 17 variables on blood flow. Ten clinical variables were also obtained from medical records including sex, age, smoking status and diabetes. These 85 variables were then entered into an artificial intelligence (AI) programme called LogitBoost. The AI repeatedly analysed the imaging variables, and was able to learn how the imaging data interacted and identify the patterns which preceded death and heart attack with over 90% accuracy. The predictive performance using the ten clinical variables alone was modest, with an accuracy of 90%. When PET scan data was added, accuracy increased to 92.5%. The predictive performance increased significantly when CCTA scan data was added to clinical and PET data, with accuracy of 95.4%.
Another study findings showed that applying artificial intelligence (AI) to the electrocardiogram (ECG) enables early detection of left ventricular dysfunction and can identify individuals at increased risk for its development in the future. Asymptomatic left ventricular dysfunction (ALVD) is characterised by the presence of a weak heart pump with a risk of overt heart failure. It is present in three to six percent of the general population and is associated with reduced quality of life and longevity. However, it is treatable when found. Currently, there is no inexpensive, noninvasive, painless screening tool for ALVD available for diagnostic use. When tested on an independent set of 52,870 patients, the network model yielded values for the area under the curve, sensitivity, specificity, and accuracy of 0.93, 86.3 percent, 85.7 percent, and 85.7 percent, respectively. Furthermore, in patients without ventricular dysfunction, those with a positive AI screen were at four times the risk of developing future ventricular dysfunction compared with those with a negative screen.
In recent years, the analysis of big data database combined with computer deep learning has gradually played an important role in biomedical technology. For a large number of medical record data analysis, image analysis, single nucleotide polymorphism difference analysis, etc., all relevant research on the development and application of artificial intelligence can be observed extensively. For clinical indication, patients may receive a variety of cardiovascular routine examination and treatments, such as: cardiac ultrasound, multi-path ECG, cardiovascular and peripheral angiography, intravascular ultrasound and optical coherence tomography, electrical physiology, etc. By using artificial intelligence deep learning system, the investigators hope to not only improve the diagnostic rate and also gain more accurately predict the patient’s recovery, improve medical quality in the near future.
The primary issue about using artificial intelligence in cardiology, or in any field of medicine for that matter, is the ethical issues that it brings about. Physicians and healthcare professionals prior to their practice swear to the Hippocratic Oath—a promise to do their best for the welfare and betterment of their patients. Many physicians have argued that the use of artificial intelligence in medicine breaks the Hippocratic Oath since patients are technically left under the care of machines than of doctors. Furthermore, as machines may also malfunction, the safety of patients is also on the line at all times. As such, while medical practitioners see the promise of artificial technology, they are also heavily constricted about its use, safety, and appropriateness in medical practice.
Issues and challenges faced by technological innovations in cardiology are overpowered by current researches aiming to make artificial intelligence easily accessible and available for all. With that in mind, various projects are currently under study. For example, the use of wearable AI technology aims to develop a mechanism by which patients and doctors could easily access and monitor cardiac activity remotely. An ideal instrument for monitoring, wearable AI technology ensures real-time updates, monitoring, and evaluation. Another direction of cardiology in AI technology is the use of technology to record and validate empirical data to further analyze symptomatology, biomarkers, and treatment effectiveness. With AI technology, researchers in cardiology are aiming to simplify and expand the scope of knowledge on the field for better patient care and treatment outcomes.
References:
https://www.news-medical.net/health/Artificial-Intelligence-in-Cardiology.aspx
https://www.bhf.org.uk/informationsupport/heart-matters-magazine/research/artificial-intelligence
https://www.medicaldevice-network.com/news/heart-attack-artificial-intelligence/
https://www.nature.com/articles/s41569-019-0158-5
https://www.ncbi.nlm.nih.gov/pmc/articles/PMC5711980/
http://www.onlinejacc.org/content/71/23/2668
http://www.scielo.br/pdf/ijcs/v30n3/2359-4802-ijcs-30-03-0187.pdf
https://clinicaltrials.gov/ct2/show/NCT03877614
https://www.europeanpharmaceuticalreview.com/news/82870/artificial-intelligence-ai-heart-disease/
https://www.news-medical.net/health/Artificial-Intelligence-in-Cardiology.aspx
https://www.sciencedaily.com/releases/2019/05/190513104505.htm
















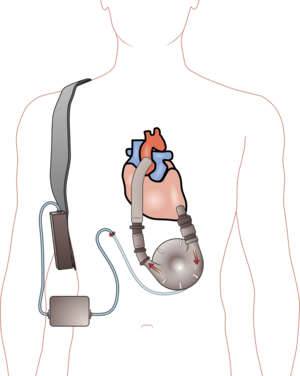
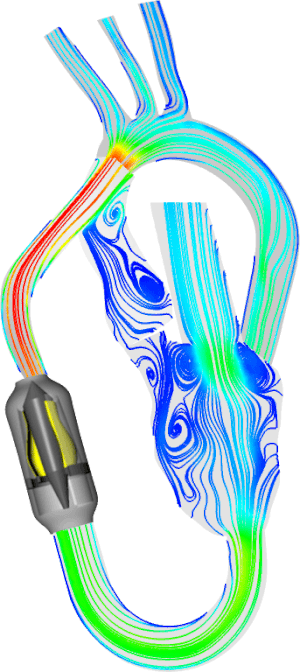
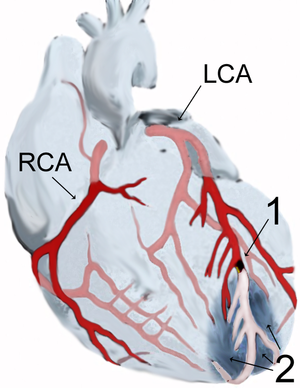


 The MIT-BIH Polysomnographic Database is a collection of recordings of multiple physiologic signals during sleep. Subjects were monitored in Boston’s Beth Israel Hospital Sleep Laboratory for evaluation of chronic obstructive sleep apnea syndrome, and to test the effects of constant positive airway pressure (CPAP), a standard therapeutic intervention that usually prevents or substantially reduces airway obstruction in these subjects. The database contains over 80 hours’ worth of four-, six-, and seven-channel polysomnographic recordings, each with an ECG signal annotated beat-by-beat, and EEG and respiration signals annotated with respect to sleep stages and apnea. For further information, see
The MIT-BIH Polysomnographic Database is a collection of recordings of multiple physiologic signals during sleep. Subjects were monitored in Boston’s Beth Israel Hospital Sleep Laboratory for evaluation of chronic obstructive sleep apnea syndrome, and to test the effects of constant positive airway pressure (CPAP), a standard therapeutic intervention that usually prevents or substantially reduces airway obstruction in these subjects. The database contains over 80 hours’ worth of four-, six-, and seven-channel polysomnographic recordings, each with an ECG signal annotated beat-by-beat, and EEG and respiration signals annotated with respect to sleep stages and apnea. For further information, see