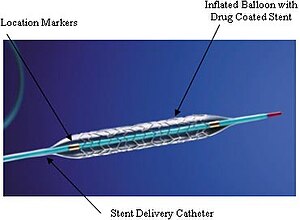
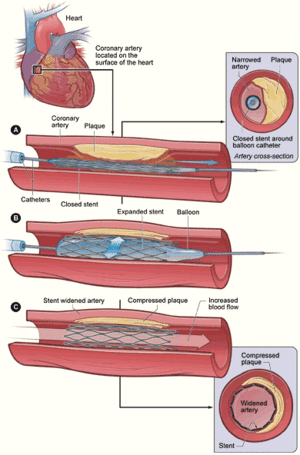
Stent Design and Thrombosis: Bifurcation Intervention, Drug Eluting Stents (DES) and Biodegrable Stents
Curator: Aviva Lev-Ari, PhD, RN
UPDATED 2/8/2014
Reva Completes Drug-Eluting Bioresorbable Stent Trial Enrollment
January 24, 2014 — Reva Medical Inc. has completed enrollment in the clinical
trial of the ReZolve2 drug-eluting
bioresorbable scaffold. A total of 112 patients from three continents have been enrolled in the trial to provide the data needed to apply for CE marking.
The company anticipates filing a CE mark application before the end of 2014. It plans to report an update on trial data at the Paris Course on Revascularization (EuroPCR) in Paris, France, May 2014.
For more information: http://www.teamreva.com
This article has the following SIX Parts:
Part I: Bifurcation Intervention – Stent Design and Thrombosis
Part II: Biodegradable Polymer DES Reduce Stent Thrombosis Rates vs. Durable Polymer DES
Part III: Stent Flexibility versus Stent Concertina Longitudinal Deformation Effect on Outcomes
Part IV: Stent Thrombosis Through the Generations of Stent Design
Part V: Stent Thrombosis in Randomized Trials of Drug-Eluting Stents: Reappraisal of the Synthesis of Evidence!
Part VI. Duration of Dual Antiplatelet Therapy following Zotarolimus-Eluting Stents and A New Strategy for Discontinuation of Dual Antiplatelet Therapy
Conclusions by Larry H Bernstein, MD, FCAP
Part I
Bifurcation Intervention – Stent Design and Thrombosis
The 5 Ts of Bifurcation Intervention: Type, Technique, Two Stents, T-Stenting, Trials
Ron Waksman, MD, FACC; Laurent Bonello, MD
Editorials published in JACC: Cardiovascular Interventions reflect the views of the authors and do not necessarily represent the views of JACC: Cardiovascular Interventions or the American College of Cardiology.
J Am Coll Cardiol Intv. 2008;1(4):366-368. doi:10.1016/j.jcin.2008.06.006
http://interventions.onlinejacc.org/article.aspx?articleid=1110233
Bifurcation, the division of an artery into 2 branches, is a common anatomy feature of the human coronary tree and is recognized as a common site for atherosclerotic plaque buildup due to the differences in coronary flow, turbulence, and shear stress at the site of the bifurcation. The prevalence of bifurcation stenosis in the human coronary tree is reported to be between 15% to 20% of all interventions and is considered complex and challenging for percutaneous intervention.
Numerous techniques and devices have been proposed to address the treatment of bifurcation lesions: balloon angioplasty, metallic stents, drug-eluting stents (DES), newly designed stents with dedicated access to the side branch, and full bifurcated stents. Clearly, the interest in the treatment of bifurcation stenting has increased with the availability to significantly reduce the recurrence rate, but this was associated with the increasing fear of stent thrombosis. Despite this extensive body of work and the latest innovations of 2008, there is not a “one size fits all” solution to the bifurcation puzzle, while the optimal percutaneous coronary intervention technique remains undetermined.
In this issue of JACC: Cardiovascular Interventions, Routledge et al. (1) present 2-year outcome data of 477 patients treated for bifurcation coronary disease with provisional side branch T-stenting using DES, and claim a systematic approach feasible for 90% of the patients, with acceptable efficacy and safety profiles. This editorial is written in response to this provocative study and will cover the 5 Ts of bifurcation stenting: Type of bifurcation, Techniques, Two stents versus one, T-stenting, and Trial design.
Types Of Bifurcation
Part of the complexity in treating bifurcation lesions and applying technique standardization is in regard to the numerous anatomic patterns of bifurcation stenosis and the lack of consistent, reliable methodology. Further, the variations in anatomy, angulations, and location of the disease within the bifurcation have led to the development of numerous classifications of bifurcation lesions, with differentiation between “true” bifurcation (both the main and the branch are diseased) and “false” bifurcation (either the main or the branch is disease) based on angiography. The most popular and intuitive classification is that of Medina et al. (2), which identifies at least 7 types of bifurcation involving the proximal main branch, the distal main branch, and the side branch, with different variations. If we add this to the classification of the various potential angulations between the main and the side branches, the sizes of the parent vessel and the side branch, and the different potential morphologies of the diseased segment (calcification, fibrosis, and so on), we can identify nearly endless anatomic and morphologic configurations of bifurcations types (3).
Technique
2 stents versus 1
Numerous techniques have been proposed for the treatment of bifurcation lesions. The first decision that the operator must make is whether the procedure will involve 1 or 2 stents. The most important information relates to the size of the side branch and the degree of the disease in this branch. Or do we really care about the side branch? Initially, the thought of using 2 stents for all bifurcated lesions was appealing because this approach usually resulted in an optimal angiographic success rate. Among the most popular techniques that employed the use of 2 stents are the culotte, crush, V-stenting, T-stenting, and simultaneous kissing stents (4). However, after numerous reports of high rates of late complications, including an increase in stent thrombosis and restenosis frequency, systematical use of 2 stents did not live up to expectations (5–8). These poor outcomes were observed regardless of the technique used and thus discouraged the liberal use of 2 stents. Therefore, the provisional strategy gained ground: try 1 stent first, and, if the result is not acceptable (dissection, impaired lumen, or flow of the other branch), use a second stent for the side branch. The superiority of such a provisional approach over a 2-stent technique was confirmed by the Nordic Bifurcation study (9). The results of this study had operators favoring the provisional rather than the 2-stent approach. However, many questions still remain regarding this approach: can we predict which bifurcation will require 2, rather than 1 stent? In how many patients is the provisional approach feasible? If a second stent is required, what then is the optimal technique for implantation of the second stent? Is provisional stenting still superior to the 2-stent approach with the new generation of stents available? And lastly, are the latest technique modifications, including pre- and post-kissing, clinically beneficial?
The present study demonstrated that provisional stenting is feasible in 90% of all patients, and those who received a second stent in the side branch, 28%, had similar long-term outcomes as those treated with 1 stent. The outcome of this study is similar to that of the Nordic Bifurcation study, which observed no difference in outcomes at 6 months’ follow-up between 1 and 2 stents (9). Finally, the latest Nordic Bifurcation Stent Technique study, comparing the culotte and crush techniques, reported low rates of angiographic restenosis and major adverse cardiac events for both techniques (10), with similar angiographic and clinical outcomes as the provisional approach with T-stenting reported in the Routledge et al. study (1). This leaves us with the question of whether, in 2008, provisional stenting is still superior to 2 stents when an improved technique is applied and new-generation stents are used?
T-stenting
Use of the provisional T-stenting technique is gaining interest because of its simplicity and subsequent reports of good mid-term outcomes (11–13). As illustrated in the present report by Routledge et al. (1), it is feasible in a large majority of patients and is associated with low rates of recurrent events during long-term follow-up. In the past, the technique was described to resolve dissections of a side branch (8) or as a new technique for the use of 2 stents for the treatment of bifurcation lesions (11). In the present study, the authors used provisional T-stenting as the default technique. From a technical point of view, provisional T-stenting offers several advantages compared with other bifurcation techniques: it is simple to perform in most cases, and it limits the need for a second stent, as illustrated by the low rate of stenting in the side branch in the present study. One technical aspect of the procedure remains in question: is kissing post-procedure mandatory in the provisional T-stenting approach with 1 or 2 stents? Bench testing observed that the final kissing balloon may have several interesting advantages: it opens the stent cells to the side branch, it allows the side branch ostium to be at least partially covered by stent struts, and it prevents the main branch stent from becoming deformed by side branch dilation. Further, in previous studies involving crush stenting, kissing balloon was shown to be critical in preventing restenosis (14). Nevertheless, the clinical impact of a final kissing balloon in provisional T-stenting must be established in future trials. Several limitations should be considered with T-stenting: it is not applicable for all lesions, it is dependent on the bifurcation angle and cannot be applied to angles <40°; the second stent, if needed, may not be able to fully cover the ostium, which will result in switching to a mini-crush technique, and like other techniques, there is a learning curve. Nevertheless, among today’s available options, the provisional T-stenting technique seems to be the simplest and is associated with favorable long-term outcomes.
Table 1 Comparison of Bifurcation Studies in the DES Era
Bifurcation stenting continues to challenge the interventional cardiologist. Despite the recent literature, including the present manuscript, there is a lack of consensus on an array of important issues, such as: Which branches deserve protection? Should provisional stenting be the default strategy of bifurcation stenting? Should we always pre-dilate the side branch? And if 2 stents are required, which technique would be the best? Is kissing always mandatory for both branches? Are DES more thrombogenic? And finally, how will the special dedicated bifurcated stents be integrated into current practice? With further trials and perhaps the sixth T in bifurcation stenting (Time), the answers to these important questions will be answered.
References
1 Routledge H.C., Morice M.-C., Lefèvre T.; 2-year outcome of patients treated for bifurcation coronary disease with provisional side branch T-stenting using drug-eluting stents. J Am Coll Cardiol Intv. 1 2008:358-365.
2 Medina A., Suárez de Lezo J., Pan M.; A new classification of coronary bifurcation lesions. Rev Esp Cardiol. 59 2006:183
3 Thomas M., Hildick-Smith D., Louvard Y.; Percutaneous coronary intervention for bifurcation disease. A consensus view from the first meeting of the European bifurcation club. Euro Intervention. 2 2006:149-153.
4 Louvard I., Lefevre T., Morice M.C.; Percutaneous coronary intervention for bifurcation coronary disease. Heart. 90 2004:713-722.
5 Iakovou I., Schmidt T., Bonizzoni E.; Incidence, predictors, and outcome of thrombosis after successful implantation of drug-eluting stents. JAMA. 293 2005:2126-2130.
6 Finn A.V., Kolodgie F.D., Harnek J.; Differential response of delayed healing and persistent inflammation at sites of overlapping sirolimus- or paclitaxel-eluting stents. Circulation. 112 2005:270-278.
7 Daemen J., Wenaweser P., Tsuchida K.; Early and late coronary stent thrombosis of sirolimus-eluting and paclitaxel-eluting stents in routine clinical practice: data from a large two-institutional cohort study. Lancet. 369 2007:667-678.
8 Carrie D., Karouny E., Chouairi S., Puel J.; “T” shaped stent placement: a technique for the treatment of dissected bifurcation lesions. Cathet Cardiovasc Diagn. 37 1996:311-313.
9 Steigen T.K., Maeng M., Wiseth R.; Randomized study on simple versus complex stenting of coronary artery bifurcation lesions: the Nordic Bifurcation study. Circulation. 114 2006:1955-1961.
10 Gunnes P, Niemela M, Kervinen K, et al, for the Nordic-Baltic PCI Study Group. Eight months angiographic follow-up in patients randomized to crush or culotte stenting of coronary artery bifurcation lesions. The Nordic Bifurcation Stent Technique study. Paper presented at: ACC 2008 Late Breaking Trials; April 1, 2008; Chicago, IL.
11 Palvakis G., de Man F., Hamer B., Doevendas P., Stella P.R.; Registry of new technique on coronary bifurcation lesions: the Utrech-“T” experience. Euro Intervention. 3 2007:262-268.
12 Pan M., Suárez de Lezo J., Medina A.; Drug-eluting stents for the treatment of bifurcation lesions: a randomized comparison between paclitaxel and sirolimus stents. Am Heart J. 153 2007:15-17.
13 Ormiston J.A., Webster M.W., El Jack S.; Drug-eluting stents for coronary bifurcations: bench testing of provisional side-branch strategies. Catheter Cardiovasc Interv. 67 2006:49-55.
14 Ge L., Airoldi F., Iakovou I.; Clinical and angiographic outcome after implantation of drug-eluting stents in bifurcation lesions with the crush stent technique: importance of final kissing balloon post-dilation. J Am Coll Cardiol. 46 2005:613-620.
15 Hoye A., Iakovou I., Ge L.; Long-term outcomes after stenting of bifurcation lesions with the “crush” technique: predictors of an adverse outcome. J Am Coll Cardiol. 47 2006:1949-1958.
16 Sharma S.K.; Simultaneous kissing drug-eluting stent technique for percutaneous treatment of bifurcation lesions in large-size vessels. Catheter Cardiovasc Interv. 65 2005:10-16.
17 Moussa I., Costa R.A., Leon M.B.; A prospective registry to evaluate sirolimus-eluting stents implanted at coronary bifurcation lesions using the “crush technique”. Am J Cardiol. 97 2006:1317-1321.
18 Yanagi D., Shirai K., Takamiya Y.; Results of provisional stenting with a sirolimus-eluting stent for bifurcation lesion: multicenter study in Japan. J Cardiol. 51 2008:89-94.
19 Di Mario C., Morici N., Godino C.; Predictors of restenosis after treatment of bifurcational lesions with paclitaxel eluting stents: a multicenter prospective registry of 150 consecutive patients. Catheter Cardiovasc Interv. 69 2007:416-424.
20 Tsuchida K., Colombo A., Lefèvre T.; The clinical outcome of percutaneous treatment of bifurcation lesions in multivessel coronary artery disease with the sirolimus-eluting stent: insights from the Arterial Revascularization Therapies Study part II (ARTS II). Eur Heart J. 28 2007:433-442.
SOURCE
J Am Coll Cardiol Intv. 2008;1(4):366-368. doi:10.1016/j.jcin.2008.06.006
http://interventions.onlinejacc.org/article.aspx?articleid=1110233
Bifurcation Stenting
David Hildick-Smith, MD
Consultant Cardiologist and Director of Cardiac Research
Brighton-Sussex University Hospital NHS Trust
Brighton, UK
Slide 1
Bifurcation stenting and its various ramifications in the modern cardiology world. The objectives of this presentation are to talk about some of the difficulties of bifurcation stenting, to summarize the recent study data, and to talk a little bit about dedicated stent systems, as well.
Dedicated Bifurcation Stent Systems – Main types:
• Side branch facilitation
• Side branch stenting incorporated
• Main branch stenting with enhanced access
• True dedicated systems
Slide 32
So we then have the issue of dedicated stent systems. Are they the answer to some of these questions? Are they going to bail us out of these difficult geometric issues of bifurcations? There are a number of dedicated stent systems in development and available at the moment, and they fall into a few different groups. There are systems which simply facilitate side branch access. There are systems which actually incorporate side branch stenting as the primary philosophy. There are those which are essentially a main branch stent with enhanced access. And then there are the truly dedicated systems.
Bifurcation Stenting: Should You Keep it Simple
You Keep it Simple
Facilitation
Increasing success of provisional T
Slide 33
If we look at the facilitation group, there are stent systems available where there’s a wire that is integral to the balloon system, and will perhaps then facilitate getting into the side branch, and may certainly facilitate making sure that you are,
Side Branch Ostial Coverage Stents:
• Scaffold side branch ostium
• Allow subsequent main vessel stenting
The side branch ostial coverage stents are intended to scaffold the side branch and retain main vessel stenting capabilities. There are a couple of stents of this nature on the market at the moment which are undergoing clinical trials to see their general applicability.
Main Vessel Enhanced Access Stents
•Pop-up/expand into side vessel
•Improve subsequent or immediate access to side branch
Slide 35
The next group is the main vessel enhanced access stents, which, either through a pop-up mechanism with mechanical scaffolding of the side branch ostium, or with a proximal stent which is self-expanding, enhance the access to the side branch, so that you have both immediate access and subsequent access. Which is one of the things that people worry about in this situation, which is, what happens if you have to come back to that side branch vessel a few months later? Will you be able to gain access to it? So these tools may have a role there.
True Dedicated Bifurcation Stent
Stenting of both branches
Slide 36
The fourth group is the true dedicated bifurcation stent. These are clearly the most useful, but of course, mechanically and from an engineering point of view, the most difficult to create and make work. They will certainly have a potential role in bifurcation stenting, but there’s a little way to go before they could be used in a wide manner.
Slide 37
The dedicated systems, while most are quite ingenious, unfortunately most will not survive in their current format. But the true dedicated bifurcation stent will certainly have a role in the left main. And, as we come back increasingly from these bifurcations to the left main and get a mandate to be able to treat that, this is an area where there will be a significant place for dedicated bifurcation stent systems.
Conclusions
• Bifurcations remain troublesome
• Provisional T stenting is the gold standard
• Subsets of bifurcations require complex strategies
• Large side branches
• Longer ostial disease
• Current complex strategies fail us
• Crush fails more than culotte
• Dedicated devices will have a role
• Large bifurcations in main coronary tree
• Left main
Slide 38
In conclusion, bifurcation stenting is still a troublesome area. Provisional T stenting is the gold standard approach across the board, but we mustn’t forget that there may well be, and I believe there are, subsets of bifurcations which do require a complex strategy. These are the ones with large side branches and significant length of disease at the ostium of that side branch. The current complex strategies do fail from a mechanical point of view, and in that respect crush fails more than culotte. Although it’s a difficult time for dedicated devices at the moment, I think they will have a role, particularly in large bifurcations in the main coronary tree and, most particularly of all, in the left main stem.
SOURCE
http://www.theheart.org/documents/satellite_programs/intervsurgery/913801/BifurcationStenting_REVISED_FINAL.pdf
Part II
Biodegradable Polymer DES Reduce Stent Thrombosis Rates vs. Durable Polymer DES
March 27, 2012 — Biodegradable polymer drug-eluting stents (DES) provide better long-term safety and efficacy than durable polymer DES, according to findings from an analysis of three major clinical trials —
- ISAR-TEST 3,
- ISAR-TEST 4 and
- LEADERS.
The data were presented at at the American College of Cardiology’s 61st Annual Scientific Session. The findings provide the first combined long-term data on the comparison between biodegradable polymer DES and durable polymer DES. Designed to improve long-term clinical outcomes while also shortening healing time, biodegradable polymer DES are a new generation of DES that have undergone little research and thus have yet to substantiate its claims. The three analyzed studies showed that after four years, use of biodegradable polymer DES resulted in
- lower rates of target lesion revascularization,
- definite stent thrombosis and
- cardiac death and
- heart attack than durable polymer DES.
“Because it is often difficult to design individual trials to test for differences in rarely occurring adverse events [like stent clotting], we pooled the data from the three largest trials to see if any differences between the two stent types could be seen,” said co-lead investigator Robert A. Byrne, M.B., B.Ch., Ph.D., a cardiologist at Deutsches Herzzentrum in Munich, Germany. “In addition, by including surveillance out to four years, this helped us better capture the differences between the two stents, as benefit was expected to first emerge with long-term follow-up.”
Among all three analyzed trials, 2,358 patients were randomly assigned to angioplasty with a biodegradable polymer DES (sirolimus-eluting = 1,501; biolimus-eluting = 857), while 1,704 patients were treated with a durable polymer SES (all sirolimus-eluting).
At the four-year follow-up point, the researchers found that the risk of target lesion revascularization (the study’s primary efficacy endpoint) was significantly lower among those patients treated with a biodegradable polymer DES than for those treated with a durable polymer DES (hazard ratio [HR] 0.82, 95 percent confidence interval [CI] 0.68-0.98, P=0.029). In addition, the risk of having a blood clot, called stent thrombosis (the study’s primary safety endpoint), was also significantly lower for those patients treated with a biodegradable polymer DES compared to those treated with a durable polymer DES (HR 0.56, 95 percent CI 0.35-0.90, P=0.015). This was driven by a lower risk of very late stent thrombosis (clots occurring more than one year after angioplasty) for the biodegradable polymer group (HR 0.22, 95 percent CI 0.08-0.61, P=0.004).
Furthermore, the incidence of heart attack late after stenting was lower for patients treated with biodegradable polymer versus durable polymer stents (HR 0.59, 95 percent CI 0.73-0.95, P=0.031).
While the arrival of DES has allowed interventionalists to provide treatment for more complex patients, concerns have arisen about the stents’ long-term safety, particularly concerning stent thrombosis. As a result, the polymer coating on the first-generation stents was targeted as an area for improvement. Specifically, the durable polymer remains in the coronary artery wall beyond the time when its useful function is served. This may cause delayed healing and a hypersensitivity reaction, leading to inflammation and stent thrombosis.
As a potential solution to these problems, new-generation stents with a bioabsorbable polymer were created. This polymer, which fully degrades and leaves a bare-metal stent in place, has been suggested to shorten healing time and cause less inflammation and subsequent stent thrombosis.
“These findings show that biodegradable polymer DES can provide better long-term safety and efficacy,” said Byrne. “This advantage, coupled with a shortened healing time compared with durable polymer DES, means that biodegradable polymer stents look to become an important tool for the interventional cardiologist in everyday practice.”
The current analysis was industry independent, supported in part by a grant from the Swiss National Science Foundation, and conducted at the ISAR Research Center in Munich, Germany, and the Clinical Trials Unit in Bern, Switzerland.
This study was simultaneously published in the European Heart Journal and was released online at the time of presentation.
The results offer a promising outlook for Boston Scientific’s Synergy DES, now in development. It uses the same platform stent as the Ion and Promus, but instead of a duable polymer it uses abluminal biodegradable polymer containing everolimus. The company presented its first-in-man study at TCT 2011 and hopes to begin its EVOLVE II U.S. Food and Drug Administration (FDA) investigational decive exemption trial later this year.
For more information: www.acc.org
Biosensensors BioMatrix Flex was among the stents included in this study. It uses an abluminal, biodegradable polymer as a carrier for its BA9 drug.
http://www.dicardiology.com/article/biodegradable-polymer-des-reduce-stent-thrombosis-rates
First Patient Enrolled in Dissolving Drug-Polymer Coronary Stent Trial
February 21, 2011 – The first patient has been enrolled the DESSOLVE II study to support CE mark for a coronary stent that uses a bioresorbable drug polymer. The MiStent drug-eluting coronary stent system (MiStent DES), by Micell Technologies.
The trial involves treatment of patients with de novo lesions in the native coronary arteries. Stefan Verheye, M.D., Ph.D. at Middelheim Hospital, Antwerp, Belgium enrolled the first patient in the study.
The MiStent DES employs supercritical fluid technology, which applies a precisely controlled absorbable polymer – active drug (sirolimus) matrix onto a cobalt-chromium stent. The polymer dissolves and releases the drug into the surrounding tissue in a controlled manner, designed to optimize dosing of the drug throughout the affected artery. In preclinical trials, the drug completely elutes and the polymer is eliminated from the stent within 45 to 60 days in-vivo, resulting in a bare-metal stent.
DESSOLVE II is a prospective, controlled, 2:1 unbalanced randomized, multicenter study of approximately 270 patients. Patients will be enrolled at 26 clinical sites in Europe, New Zealand and Australia. Candidates for the trial are patients with documented stable or unstable angina pectoris or ischemia. The primary endpoint is superiority of MiStent DES in minimizing in-stent late lumen loss at nine months, compared to Medtronic’s Endeavor DES, as measured with angiography in treated de-novo lesions ranging in diameter from 2.5 to 3.5 mm and amenable to treatment with a maximum 23 mm long stent.
Along with secondary clinical endpoints such as major adverse cardiac events and revascularization rates, the extent of stent coverage and re-endothelialization, via optical coherence tomography (OCT), and endothelial function (vasomotor response) will be evaluated in a subgroup of patients at nine months.
“Drug-eluting stents have significantly improved and expanded our ability to treat coronary atherosclerotic lesions compared to bare-metal stents,” said William Wijns, M.D., Cardiovascular Center, Aalst, Belgium, and principal investigator of the study. “However, cardiologists are still looking for options to improve safety and outcomes. The MiStent DES may address some of these issues directly. Based on recent GLP animal data, the polymer and drug are gone from the stent within 45 to 60 days. This may reduce the risk of late-stent thrombosis related to long-term exposure to DES nonerodible polymers. Given the relatively short residence time of polymer on the stent, MiStent DES may allow for a shorter duration of dual antiplatelet therapy and be a safer choice for noncompliant patients. These performance-enhancing properties are what interventional cardiologists are looking for in a new drug-eluting stent.”
For more information: www.micell.com
http://www.dicardiology.com/article/first-patient-enrolled-dissolving-drug-polymer-coronary-stent-trial
Part III
Stent Flexibility versus Stent Concertina Effect
Stent flexibility versus concertina effect: mechanism of an unpleasant trade-off in stent design and its implications for stent selection in the cath-lab.
Foin N, Di Mario C, Francis DP, Davies JE.
Abstract
The “concertina effect”, longitudinal deformation of the proximal segments of a deployed stent when force is applied from a guide catheter or other equipment, is a recently recognised problem which seems to particularly affect more recent stent designs. Until now, flexibility and deliverability have been paramount aims in stent design. Developments have focused on optimizing these features which are commonly evaluated by clinicians and demanded by regulatory bodies. Contemporary stent designs now provide high flexibility by reducing the number of connecting links between stent segments and by allowing the connecting links to easily change their length. These design evolutions may, however, simultaneously reduce longitudinal strength and have the unintended effect of inducing some risk of longitudinal compression of the stent (the “concertina effect”) during difficult clinical cases. Progress in stent design and elimination of restenosis by drug coating has improved PCI outcome and enabled new applications. Here we discuss design trade-offs that shaped evolution and improvement in stent design, from early bare metal designs to the latest generation of drug eluting stent (DES) platforms. Longitudinal strength was not recognised as a critical parameter by clinicians or regulators until recently. Measurements, only now becoming publically available, seem to confirm vulnerability of some modern designs to longitudinal deformation. Clinicians could be more guarded in their assumption that changes in technology are beneficial in all clinical situations. Sometimes a silent trade-off may have taken place, adopting choices that are favourable for the vast majority of patients but exposing a few patients to unintended hazard.
Int J Cardiol. 2013 Apr 15;164(3):259-61. doi: 10.1016/j.ijcard.2012.09.143. Epub 2012 Oct 22.
http://www.ncbi.nlm.nih.gov/pubmed/23084111
Stent “Concertina:” Stent Design Does Matter
On-Hing Kwok, MBBS
From the Cardiac Catheterization & Intervention Center, Hong Kong Sanatorium & Hospital, Happy Valley, Hong Kong.
ABSTRACT: The development of modern coronary stent platforms has transformed the landscape of interventional cardiology. Contemporary coronary stents are much more deliverable than older-generation stents. However, longitudinal deformation has emerged as a “new” complication in modern coronary stent platforms. Although most reported cases of longitudinal stent deformation involve mechanical or technical mishaps, it appears that it is more frequently associated with a particular stent design: the “offset peak-to-peak” stent design. This review summarizes the latest data around stent performance. Within this context, two clinical cases where longitudinal deformation was observed in the absence of any mechanical mishaps are also presented. Collectively, this evidence suggests that stent design may be a major determinant of stent performance.
SOURCE
Journal Cardiology, Volume 25 – Issue 6 – June 2013
Key words: longitudinal deformation, stent design, stent concertina, drug-eluting stent
Over the past decades, stent design and material has undergone significant evolution. The introduction of the drug-eluting stent (DES) has also made “drug delivery” another major determinant in modern stent design.1
Coronary stent design. The majority of early coronary stents were made of stainless steel. These designs were associated with variable basic manufacture, cell geometry, and strut thickness.2 Use of alloys such as cobalt chromium and platinum chromium has enabled stents to have thinner struts, while maintaining strength and radioopacity.3 Thin-strut stents improve deliverability and conformability. However, there is limited evidence suggesting that thinner struts may result in less vessel wall damage and hence less risk of restenosis.4-6 Although thin-strut DESs have never been shown to have lower restenosis rates than thick-strut DESs, the trend of thinner strut platforms has triggered innovative designs to maintain stent radial strength. The development of longer, thinner, more flexible, and easier-to-deliver stent platforms made percutaneous coronary intervention (PCI) possible even in the most tortuous anatomy and calcified vessels.7 However, longitudinal stent strength may be compromised with these modern designs.3 Stent design requires careful consideration of several performance characteristics, including crimped and expanded stent flexibility, shortening upon expansion, trackability, scaffolding, radioopacity, longitudinal strength, radial strength, and recoil.8
Stent longitudinal flexibility and deliverability prior to deployment, and vessel conformability after deployment, are widely dependent on the number, orientation, shape, thickness, and material of the crests and links.9 These parameters also determine the longitudinal strength of the stent, defined as maintenance of intact stent architecture upon exposure to compressing or elongating forces.9 Alteration of any one feature of a stent platform will undoubtedly impact other aspects of stent performance and may result in clinical complications. For instance, thinner struts improve deliverability, but reduce radio-opacity of the cobalt chromium stents. In addition, reduction of the number of fixed links between cells or alteration of their geometry may enhance flexibility and conformability, but as a consequence may compromise longitudinal strength.7
Although stent flexibility may be influenced by a variety of factors, it has been shown that stent longitudinal integrity, defined by the number of links between hoops, correlates with stent stiffness. In addition, the alignment of the links with the long axis of the stent may also be an important factor for longitudinal integrity.9
Architectural design differences are major factors affecting resistance against longitudinal compression. The peak-to-peak or peak-to-valley strut architectures of platforms result in variation between the longitudinal stiffness and strength of stents. It is highly likely that the occurrence of longitudinal deformation is dependent on a particular stent design.10
Longitudinal stent deformation. Until recently, the longitudinal strength of coronary stents has never been considered a standard parameter of stent performance. However, recent evidence identified longitudinal compression, or postdeployment stent shortening, as a newly observed complication. Longitudinal stent deformation is defined as the distortion or shortening of a stent in the longitudinal axis following successful stent deployment.3 This phenomenon describes the effect of a longitudinal compression force on the stent rings, causing them to nest or concertinate.8
PCI procedures involve multiple and complex techniques that may increase the risk for longitudinal stent compression. These include the use of extra-support guide catheters, aggressive guide catheter manipulation (deep-seat), mother and child catheter systems, multiple balloon postdilations, bifurcation stent techniques, and adjunctive devices such as intravascular ultrasound (IVUS), distal protection devices, etc.7 In a clinical setting, longitudinal compression may occur in various situations (Table 1),8 and it may simply represent an angiographic detection of an exceptional PCI complication. Protrusion of struts into the lumen and extensive malapposition of struts due to longitudinal deformation may result in disruption of flow and increasing the risk of stent thrombosis. Moreover, longitudinal deformation of a DES may result in uneven drug delivery and increase the risk for in-stent restenosis (ISR).9
Clinical reports of longitudinal deformation. Hanratty and Walsh recently described 3 cases where longitudinal compression of a previously deployed stent resulted in stent deformation. Two cases were detected angiographically, while 1 was detected on adjunctive imaging. The complication was first reported with the Promus Element (Boston Scientific) platform. However, Hanratty and Walsh noted that this phenomenon has since been observed with all modern DES platforms. They concluded that such deformation could potentially result in a suboptimal technical result for the medium- to long-term and increase the risk for stent thrombosis and ISR if left undetected.7
References
1. Htay T, Liu MW. Drug-eluting stent: a review and update. Vasc Health Risk Manag. 2005;1(4):263-276.
2. Colombo A, Stankovic G, Moses JW. Selection of coronary stents. J Am Coll Cardiol. 2002;40(6):1021-1033.
3. Williams PD, Mamas MM, Morgan K, et al. Longitudinal stent deformation — a retrospective analysis of frequency and mechanisms. EuroIntervention. 2012;8(2):267-274. Epub AOP 2011.
4. Pache J, Kastrati A, Mehilli J, et al. Intracoronary stenting and angiographic results: strut thickness effect on restenosis outcome (ISAR-STEREO-2) trial. J Am Coll Cardiol. 2003;41(8):1283-1288.
5. Moreno R, Jimenez-Valero S, Sanchez-Recalde A. Periprocedural (30-day) risk of myocardial infarction after drug-eluting coronary stent implantation: a meta-analysis comparing cobalt-chromium and stainless steel drug-eluting coronary stents. EuroIntervention. 2011;6(8):1003-1010.
6. Kastrati A, Mehilli J, Dirschinger J, et al. Strut thickness effect on restenosis outcome (ISAR-STEREO) trial. Circulation. 2001;103(23):2816-2821.
7. Hanratty CG, Walsh SJ. Longitudinal compression: a “new” complication with model coronary stent platforms — a time to think beyond deliverability. EuroIntervention. 2011;7(7):872-877. Epub AOP 2011.
8. Prabhu S, Schikorr T, Mahmoud T, Jacobs J, Potgieter A, Simonton C. Engineering assessment of the longitudinal compression behavior of contemporary coronary stents. EuroIntervention. 2012;8(2):275-281.
9. Ormiston JA, Webber B, Webster MWI. Stent longitudinal integrity — bench insights into a clinical problem. JACC Cardiovasc Interv. 2011;4(12):1310-1317.
10. Mortier P, De Beule M. Stent design back in the picture: an engineering perspective on longitudinal stent compression. EuroIntervention. 2011;7(7):773-776.
11. Stone GW, Teirstein PS, Meredith IT, et al; PLATINUM Trial Investigators. A prospective randomised evaluation of a novel everolimus-eluting coronary stent: the PLATINUM trial. J Am Coll Cardiol. 2011;57(16):1700-1708.
12. Pitney M, Pitney K, Jepson N, et al. Major stent deformation/pseudofracture of 7 Crown Endeavor/Micro Driver stent platform: incidence and causative factors. EuroIntervention. 2011;7(2):256-262.
13. Finet G, Rioufol G. Coronary stent longitudinal deformation by compression: is this a new global stent failure, a specific failure of a particular stent design, or simply an angiographic detection of an exceptional complication. Eurointervention. 2012;8(2):177-181. Epub AOP 2011.
Disclosure: The author has completed and returned the ICMJE Form for Disclosure of Potential Conflicts of Interest. The author reports no conflicts of interest regarding the content herein.
Manuscript submitted September 12, 2012, provisional acceptance given October 31, 2012, final version accepted January 14, 2013.
Address for correspondence: On-Hing Kwok, MBBS, FRCP, FACC, FSCAI, Cardiology Center, 6/F Li Shu Fan Building, Hong Kong Sanatorium & Hospital, 2 Village Road, Happy Valley, Hong Kong. Email: vohkwok@hksh.com
SOURCE
J INVASIVE CARDIOL 2013;25(6):E114-E119
Part IV
Stent Thrombosis Through the Generations of Stent Design
A recent retrospective analysis provided further valuable information on the frequency and mechanisms of longitudinal stent deformation. The study involved 4455 interventional cases performed during a 4-year period. Stent deformation occurred in a total of 9 cases (0.2%) and affected 0.097% of stents deployed. In 6 cases, the Promus Element stent was involved, and there was 1 case each involving Endeavor (Medtronic), Biomatrix (Biosensors Interventional Technologies), and Taxus Liberté (Boston Scientific) stents. Stent deformation varied from 0% in several stent types to 0.86% in the case of Promus Element.3 It was virtually unseen in the Cypher and Xience (Abbott Vascular) platforms. Longitudinal stent deformation is probably not a “class effect,” but highly dependent on a particular stent design.
http://www.invasivecardiology.com/articles/stent-“concertina”-stent-design-does-matter
Author(s):
Lawrence Rajan, MD and David J. Moliterno, MD
From the Gill Heart Institute and Division of Cardiovascular Medicine, University of Kentucky, Lexington, Kentucky.
Stent thrombosis (ST), while infrequent, remains a dreaded complication of percutaneous coronary revascularization because of the associated rates of
- major myocardial infarction (60%-70%) and
- early mortality (20%-25%).1
As coronary stents became more widely used in clinical practice during the late 1990s to treat acute vessel closure and to reduce restenosis, the emergence of ST redirected the efforts of the cardiology community to mitigate or eliminate this potentially catastrophic event. Advances in
- stent design and strut thinness,
- the advent of drug-eluting stent (DES) options, and
- more potent antithrombotic therapy
have been substantial influences on ST.
- DESs have been associated with higher ST rates as compared to their bare-metal counterparts, particularly when utilized among high-risk groups and high-risk lesions.
More recently, early meta-analyses of smaller studies have suggested
- reduced ST rates with newer-generation DESs versus prior versions.2 Similarly, observations from a randomized trial suggested
- lower ST rates with the newer-generation everolimus-eluting stent (<1%) compared to rates for the older-generation paclitaxel-eluting stent (3%).3
So while this uncommon but catastrophic complication persists in contemporary practice, its low frequency has made it difficult to study, particularly in the real-world setting.
In the current issue of the Journal of Invasive Cardiology, Dores et al have analyzed the outcome data from a large-volume, single-center prospective registry evaluating the incidence of definite ST.4 The study consisted of 3806 patients who underwent percutaneous coronary intervention between January 2003 and December 2010. In the registry, a total of 2388 patients (62.7%) were treated with first-generation DESs (sirolimus-eluting and paclitaxel-eluting stents), while 1418 patients (37.3%) were treated with second-generation DESs (everolimus-eluting and zotarolimus-eluting stents). The overall occurrence of Academic Research Consortium (ARC)-defined definite ST at 12 months was 1.2% (46 events). After correction for baseline differences between study groups and other variables deemed to influence the occurrence of ST, Dores et al concluded that the
- use of first-generation DESs was associated with a 2.4-fold increase in the risk of definite ST. Among the cases receiving a first-generation DES,
- the risk of ST was higher for paclitaxel-eluting versus sirolimus-eluting stents.
The observations from Dores et al are consistent with prior reports, in that the rates of definite ST are low and decreasing in recent years. As can be seen in Dores’s Figure 3 considering annual frequency of definite ST, the numerically highest years were 2003 and 2004, and over the most recent years, rates have averaged closer to 1%. Questions will remain in the field of ST, some of which will require large-scale registry data to help consider their relevance and possible answers.
The underlying challenge remains how to afford to study such low-frequency events with multifactorial and variable etiologies. Beyond the events during the interventional procedure and device utilized (ie, type of DES), many other factors that affect the rate of ST (eg, patient genotype and phenotype) are still being unraveled. Considerable research has gone into finding predictive subsets for those at increased risk for ST.5 Among identified factors are the timing and acuity of presentation. Patients presenting with an ACS are known to be more vulnerable to early ST than patients with chronic stable disease. The initial plaque rupture of ACS triggers a prothrombotic avalanche of events, from platelet activation to local thrombus formation and occlusion, spasm, and distal embolization of microcirculatory debris.6 It is interesting to note in the Dores et al. registry that patients receiving second-generation DESs more often presented with an ACS, making their observations reassuring that ST rates can be kept low with evolving care strategies.
In an analysis of the ACUITY trial, which particularly enrolled patients with ACS,7 early ST occurred with similar frequency after anticoagulation with either heparin plus glycoprotein IIb/IIIa inhibitors or bivalirudin (with or without IIb/IIIa inhibitors), and not surprisingly was predicted by diffuse atherosclerosis, suboptimal angiographic results, and inadequate pharmacotherapy. Such patients also had a higher incidence of renal insufficiency and insulin-dependent diabetes mellitus. The ACUITY subanalysis found that the rate of ST within 30 days was 1.4%, significantly higher than the 0.3%-0.5% ST rates reported among patients with stable coronary artery disease.
Among the most critical factors in mitigating the risk of ST are adequate and consistent dual-antiplatelet therapy (DAPT). A remarkable interpatient variability in the antiplatelet response to clopidogrel has been well documented. The frequency of
- clopidogrel hyporesponsiveness has been reported among as many as 30% of patients undergoing PCI, yet the clinical relevance of antiplatelet responsivity is modest,8 again since the factors related to ST are many.
- Loss-of-function alleles have been identified for clopidogrel metabolism, and these have been associated with an increased risk of adverse cardiovascular events, including ST.
- Among patients with ACS, the need for more rapid and potent pharmacological suppression of platelet reactivity in the prevention of early ST is highlighted in clinical trials testing newer antiplatelet therapies.
In a landmark trial,
- prasugrel, a more potent, consistent, and faster-acting third-generation thienopyridine has shown a significant reduction in overall ST rates compared to clopidogrel (1.1% vs 2.4%).9 Similarly,
- ticagrelor, an oral, reversible, direct-acting inhibitor of the ADP receptor P2Y12 that has a more rapid onset and greater potency of platelet inhibition than clopidogrel was recently studied in a large clinical trial.
- In the Platelet Inhibition and Patient Outcomes (PLATO) study, there was a significant reduction in ST in the ticagrelor group vs the clopidogrel group, with definite ST rates of 1.3% and 1.9%, respectively.10
It is becoming clear that there has been a generational improvement in DESs that has reduced the risk of ST. This has been paralleled by advances in DAPT regimens and interventional techniques that have collectively reduced the risk of ST. While the field will continue to search for answers to the
- optimum duration of DAPT, and whether this is dependent on
- stent type and
- acuity of patient presentation,
DES polymers, design characteristics, and the antiproliferative drugs will also continue to evolve. Understanding incremental improvements in techniques, devices, and drugs will remain quite challenging as the rate of ST slowly moves closer to zero.
References
1. Cutlip DE, Baim DS, Ho KK, et al. Stent thrombosis in the modern era: a pooled analysis of multicenter coronary stent clinical trials. Circulation. 2001;103(15):1967-1971.
2. Palmerini T, Biondi-Zoccai G, Della Riva D, et al. Stent thrombosis with drug-eluting and bare-metal stents: evidence from a comprehensive network meta-analysis. Lancet. 2012;379(9824):1393-1402.
3. Kedhi E, Joesoef KS, McFadden E, et al. Second-generation everolimus-eluting and paclitaxel-eluting stents in real-life practice (COMPARE): a randomised trial. Lancet. 2010;375(9710):201-209.
4. Dores H, Raposo L, Teles RC, et al. Stent thrombosis with second versus first generation drug eluting stents in real world coronary percutaneous intervention. J Invasive Cardiol. 2013;25(7):330-336.
5. Holmes DR Jr, Kereiakes DJ, Garg S, et al. Stent thrombosis. J Am Coll Cardiol. 2010;56(17):1357-1365.
6. Finn AV, Nakano M, Narula J, Kolodgie FD, Virmani R. Concept of vulnerable/unstable plaque. Arterioscler Thromb Vasc Biol. 2010;30(7):1282-1292.
7. Aoki J, Lansky AJ, Mehran R, et al. Early stent thrombosis in patients with acute coronary syndromes treated with drug-eluting and bare metal stents: the Acute Catheterization and Urgent Intervention Triage Strategy trial. Circulation. 2009;119(5):687-698.
8. Holmes DR Jr, Dehmer GJ, Kaul S, Leifer D, O’Gara PT, Stein CM. ACCF/AHA clopidogrel clinical alert: approaches to the FDA “boxed warning.” A report of the American College of Cardiology Foundation Task Force on Clinical Expert Consensus Documents and the American Heart Association. J Am Coll Cardiol. 2010;56(4):321-341.
9. Wiviott SD, Braunwald E, McCabe CH, et al; TRITON-TIMI 38 Investigators. Intensive oral antiplatelet therapy for reduction of ischaemic events including stent thrombosis in patients with acute coronary syndromes treated with percutaneous coronary intervention and stenting in the TRITON-TIMI 38 trial: a subanalysis of a randomised trial. Lancet. 2008;371(9621):1353-1363.
10. Wallentin L, Becker RC, Budaj A, et al; the PLATO Investigators. Ticagrelor versus clopidogrel in patients with acute coronary syndrome. N Engl J Med. 2009;361(11):1045-1057.
Disclosure: The authors have completed and returned the ICMJE Form for Disclosure of Potential Conflicts of Interest. The authors report no conflicts of interest regarding the content herein.
Address for correspondence: David J. Moliterno, MD, Department of Internal Medicine, The University of Kentucky, 900 S. Limestone Avenue, 329 Wethington Building, Lexington, KY 40536-0200. Email: moliterno@uky.edu
Journal of invasive Cardiology, Volume 25 – Issue 7 – July 2013
http://www.invasivecardiology.com/articles/stent-thrombosis-through-generations
Stent Thrombosis With Second- Versus First-Generation Drug-Eluting Stents in Real-World Percutaneous Coronary Intervention: Analysis of 3806 Consecutive Procedures From a Large-Volume Single-Center Prospective Registry
Stent thrombosis (ST) is a serious and often fatal event limiting the efficacy of percutaneous coronary intervention (PCI). The pathophysiology of ST is multifactorial, and underlying causes including stent-, procedure-, lesion-, and patient-related factors seem to play different roles at different time points after the index procedure.1,2 When compared to first-generation (1stGEN) drug-eluting stents (DESs), newer DESs have been associated with a lower rate of ST in several randomized clinical trials, subsequent meta-analyses, and also in some registries, such as the recently published Swedish Coronary Angiography and Angioplasty Registry (SCAAR).3-7 New, second-generation (2ndGEN) DESs have been developed with improved design and materials, both of which may contribute to overcome some of the limitations of the older DESs. Decreased strut thickness — resulting in higher flexibility, conformability, and deliverability — and optimized polymer biocompatibility and drug delivery kinetics have been shown to contribute to a low late-loss rate and to a lower thrombotic risk.1 Despite the evidence pointing in this direction, most of the data comes from post hoc analysis and meta-analysis, mainly because studies defining ST as a primary endpoint are scarce.
We aimed to assess whether or not the systematic use of a 2ndGEN DES, relative to the 1stGEN DES, translates into a higher safety rate in a real-world population where DES implantation was indicated. For that purpose, we conducted an analysis of a single-center prospective registry, evaluating the incidence of definite ST, as defined by the Academic Research Consortium (ARC), at 12 months of follow-up as the primary outcome measure.
Author(s):
Helder Dores, MD, Luís Raposo, MD, Rui Campante Teles, MD, Carina Machado, MD, Sílvio Leal, MD, Pedro Araújo Gonçalves, MD, Henrique Mesquita Gabriel, MD, Manuel Sousa Almeida, MD, Miguel Mendes, MD
Abstract
Background and Aims. When compared to their first-generation (1stGEN) counterparts, second-generation (2ndGEN) drug-eluting stents (DESs) have been associated with better clinical outcomes in randomized clinical trials, namely by reducing the rates of stent thrombosis (ST). Our goal was to investigate whether or not the broad use of newer devices would translate into higher safety in a real-world population. For that purpose, we compared the occurrence of definite ST at 12 months between two patient subsets from a large-volume single-center registry, according to the type of DES used. Total mortality was a secondary endpoint.
Methods and Results. Between January 2003 and December 2010, a total of 3806 patients were submitted to percutaneous coronary intervention (PCI) with only 1stGEN or 2ndGEN DES: 2388 patients (62.7%) were treated with 1stGEN DES only (sirolimus-eluting stent [SES] = 1295 [34.0%]; paclitaxel-eluting stent [PES] = 943 [24.8%]; both stent types were used in 150 patients) and 1418 patients (37.3%) were treated with 2ndGEN DESs only. The total incidence of definite ST (as defined by the Academic Research Consortium) at 12 months was 1.2% (n = 46). After correction for baseline differences between study groups and other variables deemed to influence the occurrence of ST, the use of 1stGEN DES was associated with a significant 2.4-fold increase in the risk of definite ST (95% confidence interval [CI], 1.05-5.42; P=.039) at 12 months; adjusted risk was higher with PES (hazard ratio [HR], 3.6; 95% CI, 1.48-8.70; P=.005) than with SES (HR, 2.3; 95% CI, 0.92-5.65; P=.074). Total mortality (3.7% vs 3.5%) did not differ significantly between groups (adjusted HR, 1.2; 95% CI, 0.81-1.84, P=.348).
Conclusions. Our data suggest that in the real-world setting of contemporary PCI, the unrestricted use of newer 2ndGEN DESs translates into an improvement in PCI safety (relative to 1stGEN DESs), with a significantly lower risk of definite ST at 12 months.
Journal of Invasive Cardiology Volume 25 – Issue 7 – July 2013
J INVASIVE CARDIOL 2013;25(7):330-336
Key words: stent thrombosis, drug-eluting stent
http://www.invasivecardiology.com/articles/stent-thrombosis-second-versus-first-generation-drug-eluting-stents-real-world-percutaneous
Part V
Stent Thrombosis in Randomized Trials of Drug-Eluting Stents:
Reappraisal of the Synthesis of Evidence!
Stent Thrombosis in Randomized Clinical Trials of Drug-Eluting Stents
Laura Mauri, M.D., Wen-hua Hsieh, Ph.D., Joseph M. Massaro, Ph.D., Kalon K.L. Ho, M.D., Ralph D’Agostino, Ph.D., and Donald E. Cutlip, M.D.
N Engl J Med 2007; 356:1020-1029February 12, 2007DOI: 10.1056/NEJMoa067731
http://www.nejm.org/doi/full/10.1056/NEJMoa067731?goback=%2Egde_675087_member_263490750
EDITORIAL on bare-metal stents (BMS) vs sirolimus-eluting stents (SES)
With full interest, we read the article “Stent thrombosis in randomized clinical trials (RCT) of drug-eluting stents (DES)” by Mauri L et al, previously published in the New England Journal of Medicine in 2007 [1]. The authors concluded that “The incidence of stent thrombosis (ST) did not differ significantly between patients with DES and those with bare-metal stents (BMS) in RCT, although the power to detect small differences in rates was limited” [1].
I have the following concerns. First and foremost, ST in the BMS groups occurred more frequently among patients who underwent intervening target lesion revascularization (TLR) versus those who did not [1]. And since brachytherapy was the standard of care for treatment of restenosis at that time, it was used more frequently in patients with restenosis following BMS (9 out of 11 patients with BMS who underwent intervening TLR and subsequently developed definite/probable ST), in whom restenosis occurred more frequently and more diffusely, compared with DES [1]. In an observational study, brachytherapy was associated with a high risk of late (thrombotic) total occlusion of the index vessel at 6-month angiographic follow-up [2]. In that study, the mean time from brachytherapy to late total occlusion was 5.4 ± 3.2 months [2]. Therefore, brachytherapy may constitute selection bias for devices with higher rates of restenosis, by increasing the risk of late ST following intervening procedures for these devices. This might explain the much higher rate of late (beyond 30 days to 1 year) definite/probable ST following BMS compared with sirolimus-eluting stents (SES) (1% versus 0.1%, respectively), which was obviously responsible for the higher overall rate of definite/probable ST following BMS compared with SES at 4-year follow-up (1.7% versus 1.5%, respectively, p=0.7) [1]. It is worth mentioning that
- BMS was associated with a lower rate of very late (beyond 1 year) definite/probable ST compared with SES (0.4% versus 0.9%, respectively) [1]. Second,
- the study included 4 RCT of SES published from 2002 to 2004, and 4 RCT of paclitaxel-eluting stents (PES) published from 2003 to 2005, all of which were published before the Academic Research Consortium (ARC) report that put forward the current standard definitions of ST [3].
Thus, the ARC definitions were applied to all of these trials retrospectively, and therefore, might have missed some of the ST events.
- Third, the study enrolled 878 patients with SES versus 870 treated with the corresponding BMS, 1400 patients with PES versus 1397 treated with the corresponding BMS; thus, it was clearly underpowered for detection of a difference in rare-by-nature events such as ST. Forth, the
- RCT included in the study were the earliest RCT of SES and PES; hence, they enrolled relatively low-risk patient, lesion, and clinical subsets, that do not reflect real-world practice.
- Finally, the individual databases of RCT of PES were managed by Boston Scientific, which might introduce another source of bias!
References
1. Mauri L, Hsieh WH, Massaro JM, et al. Stent thrombosis in randomized clinical trials of drug-eluting stents. N Engl J Med 2007;356:1020-9.
2. Waksman R, Bhargava B, Mintz GS, et al. Late total occlusion after intracoronary brachytherapy for patients with in-stent restenosis. J Am Coll Cardiol. 2000;36:65-8.
3. Cutlip DE, Windecker S, Mehran R, et al. Clinical end points in coronary stent trials: a case for standardized definitions. Circulation 2007;115:2344-51.
Part VI
Duration of Dual Antiplatelet Therapy following Zotarolimus-Eluting Stents and A New Strategy for Discontinuation of Dual Antiplatelet Therapy
Dr. Pearlman: Drug eluting stents decrease in stent stenosis from endothelial exuberant growth at the cost of increased propensity to thrombosis, offset by prolonged use of dual anti platelet medication. The net effect depends on compliance which if good results in net decrease. The risk has increased due to drug eluting stent prevalence, but that is offset by management with dual anti platelet agents, so the net incidence is reduced. There have been a number of presentations based on angioscopy showing thrombus inside bare metal and drug eluting stents that supported the general concensus also supported by TIMI trials that stent thrombosis is promoted by metal stents until they endothelialize, and that drug-eluting stents impede the endothelialization “too well” prolonging that issue, so minimal dual platelet agent duration in practice is 3 months for BMS, 6-12 months for DES, but benefit fades to 2% at 1 year, 1% at 2 years at which point risk-benefit is unconvincing and many stop plavix, while some insist it is a lifetime medication.
With full interest, we read the article “Dual antiplatelet therapy duration and clinical outcomes following treatment with zotarolimus-eluting stents (ZES)” by Kandzari DE, et al [1]. The authors concluded that “Among patients treated with ZES, late-term events of death, myocardial infarction (MI), stroke, and stent thrombosis (ST) do not significantly differ between patients taking 6 months dual antiplatelet therapy (DAPT) compared with continuation beyond 1 year” [1].
I have the following concerns. First, although the authors claimed that their study was based on a pooled analysis of patients who received ZES in 5 ‘clinical trials’; actually, 2 out of 5 were not ‘trials’. One was a registry of direct stenting with ZES [2], and the other was a study of pharmacokinetics of ABT 578 in a subset of the ENDEAVOR II trial, that was not published in a medical journal [3]! Second, patients were classified by “DAPT adherence according to the most recent report of compliance with aspirin and thienopyridine”. Evidence supports that premature discontinuation clopidogrel is the most powerful independent predictor of late ST [4].
There is no evidence, however, that stopping aspirin predisposes to ST following drug-eluting stent implantation. Third, follow-up of DAPT adherence was done at 30 days, 6 months, then annually for 3 years. Reporting DAPT adherence based on “the last reported follow-up interval of compliance with both aspirin and clopidogrel” does not reflect the actual duration of clopidogrel received in any of the comparison groups. Forth, in the second comparison of “6 months on/24 months off” (on DAPT at 6 but not at 24 months) versus “≥24 months” (on DAPT at 6 and 24 months)”, the first group included, by definition, patients who were also on DAPT at 12 months (but not at 24 months). Thus, it cannot be taken to reflect a comparison between 6-month DAPT and 24-month DAPT! Fifth, the ENDEAVOR II and ENDEAVOR III trials were published in 2006, before the publication of ARC report [5,6]. Therefore, the ARC definitions of ST were applied retrospectively in many patients, which might explain the absence of ‘probable’ ST in all comparison groups, in all time points. Sixth, major bleeding was defined exclusively as “any hemorrhagic event that required blood product transfusion”. This might explain why such rates were 0% in all groups, in all time points. Finally, the study involved low-risk patient and lesion subsets, and was statistically underpowered for rare events such as ST, cardiac death, or MI.
References
1. Kandzari DE, Barker CS, Leon MB, et al. Dual antiplatelet therapy duration and clinical outcomes following treatment with zotarolimus-eluting stents. JACC Cardiovasc Interv 2011;4:1119-28.
2. Schultheiss HP, Grube E, Kuck KH, et al. Endeavor II Continued Access Investigators. Safety of direct stenting with the Endeavor stent: results of the Endeavor II continued access registry. EuroIntervention 2007;3:76–81.
3. Pharmacokinetics of ABT-578 in patients from Endeavor stent: results from a subset of a double-blind, randomized, multicenter (ENDEAVOR-II) trial. In: The ENDEAVOR II Study 30-Day Pharmacokinetic Report. Abbot Park, IL: Abbott Laboratories, 2004.
4. Iakovou I, Schmidt T, Bonizzoni E, et al. Incidence, predictors, and outcome of thrombosis after successful implantation of drug-eluting stents. JAMA 2005;293:2126-30.
5. Fajadet J, Wijns W, Laarman GJ, et al. ENDEAVOR II Investigators. Randomized, double-blind, multicenter study of the Endeavor zotarolimus-eluting phosphorylcholine-encapsulated stent for treatment of native coronary artery lesions: clinical and angiographic results of the ENDEAVOR II trial. Circulation 2006;114:798–806.
6. Kandzari DE, Leon MB, Popma JJ, et al. ENDEAVOR III Investigators. Comparison of zotarolimus-eluting and sirolimus-eluting stents in patients with native coronary artery disease: a randomized controlled trial. J Am Coll Cardiol 2006;48:2440–7.
SOURCE
interventions.onlinejacc.org <http://interventions.onlinejacc.org> interventions.onlinejacc.org <http://interventions.onlinejacc.org>
A New Strategy for Discontinuation of Dual Antiplatelet Therapy
With interest, we read the article “A New Strategy for Discontinuation of Dual Antiplatelet Therapy: REal Safety and Efficacy of 3-month dual antiplatelet Therapy following Endeavor zotarolimus-eluting stent implantation (RESET) Trial” by Kim B-K, et al [1]. The authors concluded that Endeavor zotarolimus-eluting stent (E-ZES) with 3-month dual antiplatelet therapy (DAPT) was noninferior to other drug-eluting stents (DES) with 12-month DAPT (standard therapy) with respect to the occurrence of the primary endpoint (a composite of cardiovascular death, myocardial infarction (MI), stent thrombosis (ST), target vessel revascularization (TVR), or bleeding at 1 year) [1].
I have the following concerns. First, the study design was defective since the comparator group should have been composed of patients who received the same stent (E-ZES) and took DAPT for 12 months. Moreover, the comparator group was not homogeneous, since it was composed of patients who received sirolimus-eluting stents (SES, Cypher, 28.5%), everolimus-eluting stents (EES, Xience, 30%), and ZES with a biocompatible polymer (R-ZES, Resolute, 41.5%). This would further complicate the comparison since it dilutes the results of the comparator group by mixing first- (Cypher) with second-generation (Xience and Resolute) DES. Further confusion was added with the unjustified stratified randomization of the comparator group: patients with Diabetes mellitus (DM) and those with acute coronary syndrome (ACS) were assigned to R-ZES; those with short lesions to SES; those with long lesions to EES. Second, whereas the trial compared two regimens (short versus long) of DAPT following DES, the primary endpoint adopted by the authors included ischemia-driven TVR; an event completely unrelated to the safety or efficacy of a DAPT regimen. Third, the authors could not explain why the event rates were very low (cardiovascular death 0.2%, MI 0.2%, ARC definite/probable ST 0.2%) compared with previous reports of the E-ZES at a similar time point: ENDEAVOR II trial (total death 1.2%, MI 2.7%, ST 0.5% at 9 months); ENDEAVOR IV trial (cardiac death 0.5%, MI 1.6%, ARC definite/probable ST 0.9% at 12 months) [2,3]. Forth, unexpectedly, both TVR and ST rates in patients with DM who received E-ZES were lower than the rates for the whole E-ZES group! And in the ACS subgroup, patients who received the standard therapy (R-ZES) had rates of cardiovascular death 0%, MI 0%, and ST 0%, at 12 months! And surprisingly, in the subset of short lesions, despite the shorter duration of DAPT, bleeding rates were higher with E-ZES + 3-month DAPT versus standard therapy (0.6% versus 0%)! Fifth, based on the current low 12-month rates of primary composite endpoint (4.7%) compared with the figure used for statistical power calculation (10-11%), the trial was underpowered for the primary endpoint. Additionally, the non-inferiority margin of 4% was very wide for the 12-month rates of primary endpoint (4.7%). Finally, enrollment of 2117 patients in 26 centers over 20 months speaks of a low enrollment rate of 4.1 patients/center/month, that reflects an overt selection bias.
References
1. Kim BK, Hong MK, Shin DH, et al. A new strategy for discontinuation of dual antiplatelet therapy: the RESET Trial (REal Safety and Efficacy of 3-month dual antiplatelet Therapy following Endeavor zotarolimus-eluting stent implantation). J Am Coll Cardiol 2012;60:1340-8.
2. Fajadet J, Wijns W, Laarman GJ, et al. Randomized, double-blind, multicenter study of the Endeavor zotarolimus-eluting phosphorylcholine-encapsulated stent for treatment of native coronary artery lesions: clinical and angiographic results of the ENDEAVOR II trial. Circulation 2006;114:798-806.
3. Leon MB, Mauri L, Popma JJ, et al. A randomized comparison of the ENDEAVOR zotarolimus-eluting stent versus the TAXUS paclitaxel-eluting stent in de novo native coronary lesions 12-month outcomes from the ENDEAVOR IV trial.
SOURCE
J Am Coll Cardiol 2010;55:543-54.
content.onlinejacc.org content.onlinejacc.org
http://digitalreprints.elsevier.com/i/85787/6
Conclusions
by Larry H Bernstein, MD, FCAP
This has been a six part discussion on the progress of stent design, and the decreasing problem of stent thrombosis, which evades elimination with a tradeoff in greater utility and somewhat greater risk. However, the risk of thrombotic events has become low enough that accurate comparisons of stent technologies, method of placement, and antithrombotic techniques to avoid thrombotic complications is burdened by statistical power limitations. In addition to the issue of sample size, there is an issue of patient characteristics that probably confer increased risk.
In the first part we found that stent placement is done in 15-20% of cases at a bifurcation site, where it is most favorable for plaque buildup from turbulent flow and shear stress. Recall that Routledge et al. (1) presented 2-year outcome data of 477 patients treated for bifurcation coronary disease with provisional side branch T-stenting using drug-eluting stents (DES), and they concluded that a systematic approach is feasible for 90% of the patients, with acceptable efficacy and safety profiles. There are several inherent problems that encumbered any analysis. These were: numerous anatomic configurations of bifurcation types, with the concern for late complications, restenosis, and its frequency, leading to the dilemma of placing two stents versus one stent, and then another as a side branch, if needed. The study (1) did indicate that provisional stenting is feasible in 90% of all patients, and those who received a second stent in the side branch, 28%, had similar long-term outcomes as those treated with 1 stent. The outcome of this study is similar to that of the Nordic Bifurcation study, which observed no difference in outcomes at 6 months’ follow-up between 1 and 2 stents (9). As for technique, the latest Nordic Bifurcation Stent Technique study, comparing the culotte and crush techniques, reported low rates of angiographic restenosis and major adverse cardiac events for both techniques (10). However, kissing balloon was shown to be critical in preventing restenosis. Provisional T-stenting offers several advantages compared with other bifurcation techniques. It seems to be the simplest and is associated with favorable long-term outcomes. It has also been shown that side branches and osteal disease are most problematic and that dedicated devices will have a role in left main disease.
The next issue for consideration is the use of biodegradable drug-eluting stents versus durable polymer DES. Biodegradable polymer DES resulted in lower rates than durable polymer DES of
- target lesion revascularization (hazard ratio [HR] 0.82, 95 percent confidence interval [CI] 0.68-0.98, P=0.029).
- definite stent thrombosis (the study’s primary safety endpoint), (HR 0.56, 95 percent CI 0.35-0.90, P=0.015).
- very late stent thrombosis (clots occurring more than one year after angioplasty) for the biodegradable polymer group (HR 0.22, 95 percent CI 0.08-0.61, P=0.004).
- cardiac death and heart attack (HR 0.59, 95 percent CI 0.73-0.95, P=0.031).
The third topic for consideration is the tradeoff between stent flexibility versus the concertina effect. Longitudinal strength was not recognized as a critical parameter by clinicians or regulators until recently. Measurements, only now becoming publically available, seem to confirm vulnerability of some modern designs to longitudinal deformation. Stent designs now provide high flexibility by reducing the number of connecting links between stent segments and by allowing the connecting links to easily change their length. However, this design results in reduced longitudinal strength with the unintended effect of inducing some risk of longitudinal compression of the stent (the “concertina effect”). While contemporary coronary stents are much more deliverable than older-generation stents, longitudinal deformation has emerged as a “new” complication in modern coronary stent platforms. This is more frequently associated with a particular stent design: the “offset peak-to-peak” stent design. Thin-strut stents improve deliverability and conformability. There is only limited evidence that thinner struts may result in less vessel wall damage reducing risk of restenosis. The trend of thinner strut platforms has triggered innovative designs to maintain stent radial strength. The development of longer, thinner, more flexible, and easier-to-deliver stent platforms made percutaneous coronary intervention (PCI) possible even in the most tortuous anatomy and calcified vessels. Longitudinal stent deformation, the distortion or shortening of a stent in the longitudinal axis is the effect of a longitudinal compression force on the stent rings, causing them to nest or concertinate.
The fourth question is the effect of stent design on stent thrombosis. A recent retrospective analysis provided further valuable information on the frequency and mechanisms of longitudinal stent deformation. The study involved 4455 interventional cases performed during a 4-year period. Stent deformation occurred in a total of 9 cases (0.2%) and affected 0.097% of stents deployed. Longitudinal stent deformation is probably not a “class effect,” but highly dependent on a particular stent design.
Stent thrombosis (ST), while infrequent, remains a dreaded complication of percutaneous coronary revascularization because of the associated rates of
- major myocardial infarction (60%-70%) and
- early mortality (20%-25%).1
the emergence of ST redirected the efforts of the cardiology community to mitigate or eliminate this potentially catastrophic event by
- stent design and strut thinness,
- the advent of drug-eluting stent (DES) options, and
- more potent antithrombotic therapy
DESs have been associated with higher ST rates as compared to their bare-metal counterparts, particularly when utilized among high-risk groups and high-risk lesions.
The overall occurrence of Academic Research Consortium (ARC)-defined definite ST at 12 months was 1.2% (46 events). After correction for baseline differences between study groups and other variables deemed to influence the occurrence of ST, Dores et al concluded that the
- use of first-generation DESs was associated with a 2.4-fold increase in the risk of definite ST. Among the cases receiving a first-generation DES,
- the risk of ST was higher for paclitaxel-eluting versus sirolimus-eluting stents.
It should not be a surprise that patients presenting with an ACS are known to be more vulnerable to early ST than patients with chronic stable disease. The initial plaque rupture of ACS triggers a prothrombotic avalanche of events, from platelet activation to local thrombus formation and occlusion, spasm, and distal embolization of microcirculatory debris.6 It is interesting to note in the Dores et al. registry that patients receiving second-generation DESs more often presented with an ACS, making their observations reassuring that ST rates can be kept low. Patients who had early ST were characterized by diffuse atherosclerosis, angiography, inadequate pharmacotherapy, and had a higher incidence of renal insufficiency and insulin-dependent diabetes mellitus. The ACUITY subanalysis found that the rate of ST within 30 days was 1.4%, significantly higher than the 0.3%-0.5% ST rates reported among patients with stable coronary artery disease.
Among the most critical factors in mitigating the risk of ST are adequate and consistent dual-antiplatelet therapy (DAPT). Among patients with ACS, the need for more rapid and potent pharmacological suppression of platelet reactivity in the prevention of early ST is highlighted in clinical trials testing newer antiplatelet therapies. In the Platelet Inhibition and Patient Outcomes (PLATO) study, there was a significant reduction in ST in the ticagrelor group vs the clopidogrel group, with definite ST rates of 1.3% and 1.9%, respectively.
This brings us to ST in randomized trials of DES. There was a much higher rate of late (beyond 30 days to 1 year) definite/probable ST following BMS compared with sirolimus-eluting stents (SES) (1% versus 0.1%, respectively). BMS was associated with a lower rate of very late (beyond 1 year) definite/probable ST compared with SES (0.4% versus 0.9%, respectively) [1]. The different overall rate of definite/ probable ST following BMS compared with SES is nearly equal at 4-year follow-up (1.7% versus 1.5%, respectively), is indeterminate (p=0.7) [1]. The study was underpowered for detection of a difference in rare-by-nature events such as ST.
Finally, Dr. Pearlman analyzes the published studies concerning whether there should be a reduction in the length of dual antiplatelet therapy to six months. Drug eluting stents decrease in stent stenosis from endothelial exuberant growth at the cost of increased propensity to thrombosis, offset by prolonged use of dual anti-platelet medication. The risk has increased due to drug eluting stent prevalence, but that is offset by management with dual anti platelet agents, so the net incidence is reduced. Stent thrombosis is promoted by metal stents until they endothelialize, but drug-eluting stents impede the endothelialization, so minimal dual platelet agent duration in practice is 3 months for BMS, 6-12 months for DES, but benefit fades to 2% at 1 year, 1% at 2 years at which point risk-benefit is unconvincing. Evidence supports that premature discontinuation clopidogrel is the most powerful independent predictor of late ST.
So here we have the status in a nutshell.
- ST has driven the design of stents to be simpler to insert effectively, with a clear goal to minimize ST
- The stent designs have resulted in thinner, and multi-segmented longer insertions as needed.
- The result of improved stent design has been an effect of local vessel distortion.
- The standard of practice is provisional T-branch DES
- The use of dual antiplatelet therapy for not less than 1 year is determined by the time required for endothelialization of the artery.
- There is a risk difference incurred by ACS versus stable disease, and by adequacy of antithrombotic therapy prior to an acute event.
Other related articles published on this Open Access Online Scientific Journal, include the following:
AHA, ACC Change in requirement for surgical support: Class IIb -> Class IIa Level of Evidence A: Supports Nonemergent PCI without Surgical Backup (Change of class IIb, level of Evidence B).
Larry H Bernstein, MD, FCAP and Justin D Pearlman, MD, PhD, FACC
Survivals Comparison of Coronary Artery Bypass Graft (CABG) and Percutaneous Coronary Intervention (PCI) / Coronary Angioplasty
Larry H Bernstein, MD, FCAP and Aviva Lev-Ari, PhD, RN
Coronary Reperfusion Therapies: CABG vs PCI – Mayo Clinic preprocedure Risk Score (MCRS) for Prediction of5. in-Hospital Mortality after CABG or PCI
Larry H. Bernstein, MD, FCAP and Aviva Lev-Ari, PhD, RN
Coronary Artery Disease – Medical Devices Solutions: From First-In-Man Stent Implantation, via Medical Ethical Dilemmas to Drug Eluting Stents
Aviva Lev-Ari, PhD, RN
Absorb™ Bioresorbable Vascular Scaffold: An International Launch by Abbott Laboratories
Aviva Lev-Ari, PhD, RN
CABG or PCI: Patients with Diabetes – CABG Rein Supreme
Aviva Lev-Ari, PhD, RN
To Stent or Not? A Critical Decision
Aviva Lev-Ari, PhD, RN
New Drug-Eluting Stent Works Well in STEMI
Aviva Lev-Ari, PhD, RN
Revascularization: PCI, Prior History of PCI vs CABG
Aviva Lev-Ari, PhD, RN
Drug Eluting Stents: On MIT’s Edelman Lab’s Contributions to Vascular Biology and its Pioneering Research on DES
Larry H Bernstein, MD, FCAP and Aviva Lev-Ari, PhD, RN
Outcomes in High Cardiovascular Risk Patients: Prasugrel (Effient) vs. Clopidogrel (Plavix); Aliskiren (Tekturna) added to ACE or added to ARB
Aviva Lev-Ari, PhD, RN
Read Full Post »