Biomaterials Technology: Models of Tissue Engineering for Reperfusion and Implantable Devices for Revascularization
Author and Curator: Larry H Bernstein, MD, FACP
and
Curator: Aviva Lev-Ari, PhD, RN
http://pharmaceuticalintelligence.com/5_04_2013/bernstein_lev-ari/Bioengineering_of_Vascular_and_Tissue_Models
This is the THIRD of a three part series on the evolution of vascular biology and the studies of the effects of biomaterials
in vascular reconstruction and on drug delivery, which has embraced a collaboration of cardiologists at Harvard Medical School , Affiliated Hospitals, and MIT,
requiring cardiovascular scientists at the PhD and MD level, physicists, and computational biologists working in concert, and
an exploration of the depth of the contributions by a distinguished physician, scientist, and thinker.
The FIRST part – Vascular Biology and Disease – covered the advances in the research on
- vascular biology,
- signaling pathways,
- drug diffusion across the endothelium and
- the interactions with the underlying muscularis (media),
- with additional considerations for type 2 diabetes mellitus.
The SECOND part – Stents and Drug Delivery – covered the
- purposes,
- properties and
- evolution of stent technology with
- the acquired knowledge of the pharmacodynamics of drug interactions and drug distribution.
In this THIRD part, on Problems and Promise of Biomaterials Technology, we cover the biomaterials used and the design of the cardiovascular devices, extension of uses, and opportunities for improvement
Biomaterials Technology: Tissue Engineering and Vascular Models –
Problems and Promise
We have thus far elaborated on developments in the last 15 years that have led to significant improvements in cardiovascular health.
First, there has been development of smaller sized catheters that can be introduced into
- not only coronary arteries, but into the carotid and peripheral vasculature;
Second, there has been specific design of coated-stents that can be placed into an artery
- for delivery of a therapeutic drug.
This began with a focus on restenosis, a serious problem after vascular repair, beginning
with the difficult problem of control of heparin activity given intravenously, and was
extended to modifying the heparan-sulfate molecular structure
- to diminish vascular endothelial hyperplasia,
- concurrent with restriction of the anticoagulant activity.
Third, the ability to place stents with medicated biomaterials locally has extended to
- the realm of chemotherapy, and we shall see where this progresses.
The Engineered Arterial Blood Flow Models
Biomedical engineers, in collaboration with physicians, biologists, chemists, physicists, and
mathematicians, have developed models to predict vascular repair by knowledge of
- the impact of interventions on blood flow.
These models have become increasingly sophisticated and precise, and they propel us
toward optimization of cardiovascular therapeutics in general and personalizing treatments
for patients with cardiovascular disease. (1)
The science of vascular biology has been primarily stimulated by the clinical imperative to
- combat complications that ensue from vascular interventions.
Thus, when a novel vascular biological finding or cardiovascular medical/surgical technique
is presented, we are required to ask the 2-fold question:
- what have we learned about the biology of the blood vessel?
- how might this knowledge be used to enhance clinical perspective and treatment?
The innovative method of engineering arterial conduits presented by Campbell et al. in
Circulation Research presents us with just such a challenge, and we deal with it’s biological and clinical ramifications.
Each of four pivotal studies in vascular tissue engineering has been an important advance
in the progression to a tissue-engineered blood vessel that can serve as a
- living graft, responsive to the biological environment as
- a self-renewing tissue with an inherent healing potential.
- Weinberg and Bell taught us that a tissue-engineered graft could be constructed
- and could be composed of human cells.
L’heureux et al demonstrated that the mechanical strength of such a material
- derived in major part from the extracellular matrix and
- production of matrix and integrity of cellular sheets
- could be enhanced by alterations in culture conditions.
Niklason et al. noted that grafts are optimally formed
- when incubated within environmental conditions that they will confront in vivo
- or would have experienced if formed naturally.
Campbell et al. now demonstrate that it is possible to remove
- the immune reaction and acute rejection that may follow cell-based grafting
- by culturing tissues in the anticipated host and
- address a fundamental issue of whether cell source or site of cell placement
- dictates function after cell implantation.
It appears that the vascular matrix can be remodeled by the body according to the needs of the environment. It may
very well be that the ultimate configuration of autologous cell-based vascular graft need not be determined at
outset by the cells that comprise the device, but rather
- by a dynamics that is established by environmental needs, wherein the body molds
- tissue-engineered constructs to meet
- local flow,
- metabolic, and
- inflammatory requirements.
In other words, cell source for tissue reconstruction may be secondary to
cell pliability to environmental influence.
Endothelial and smooth muscle cells from many, perhaps any,
- vascular bed can be used to create new grafts and will then
- achieve secondary function once in place in the artery.
The environmental remodeling observed after implantation
- may modify limitations of grafts that are composed of nonvascular peritoneal cells whose initial structure
is not either venous or arterial. (2)
- The trilaminate vascular architecture provides biochemical regulation and mechanical integrity.
- Yet regulatory control can be regained after injury without recapitulating tertiary structure.
Tissue-engineered (TE) endothelium controls repair even when
- placed in the perivascular space of injured vessels.
It remains unclear from vascular repair studies whether endothelial implants recapitulate the vascular
epithelial lining or expose injured tissues to endothelial cells (ECs) with unique healing potential because
- ECs line the vascular epithelium and the vasa vasorum.
Authors examined this issue in a nonvascular tubular system, asking whether airway repair is controlled by
- bronchial epithelial cells (EPs) or by
- Endothelial Cells (ECs) of the perfusing bronchial vasculature.
Localized bronchial denuding injury
- damaged epithelium, narrowed bronchial lumen, and led to
- mesenchymal cell hyperplasia, hypervascularity, and inflammatory
- cell infiltration. Peribronchial TE constructs embedded with
EPs or ECs limited airway injury, although optimum repair was obtained
- when both cells were present in TE matrices.
EC and EP expression of
- PGE2, TGF1, TGF2, GM-CSF, IL-8, MCP-1, and soluble VCAM-1
- and ICAM-1 was altered by matrix embedding,
but expression was altered most significantly when both,
- EC and EP, cells were present simultaneously.
EPs may provide for functional control of organ injury and fibrous response, and
ECs may provide for preservation of tissue perfusion and the epithelium in particular.
Together the two cells
- optimize functional restoration and healing, suggesting that
- multiple cells of a tissue contribute to the differentiated biochemical function and repair
of a tissue, but need not assume
- a fixed, ordered architectural relationship, as in intact tissues, to achieve these effects. (3)
Matrix-embedded Endothelial Cells (MEECs) Implants
The implantation of matrix-embedded endothelial cells (MEECs)
- is considered to have therapeutic potential in controlling the vascular response to injury and
- maintaining patency in arteriovenous anastomoses.
Authors considered the 3-dimensional microarchitecture of the tissue engineering scaffold to be
a key regulator of endothelial behavior in MEEC constructs.
Notably, Authors found that
- ECs in porous collagen scaffold had a markedly altered cytoskeletal structure with oriented actin
fibers and rearranged focal adhesion proteins, in comparison to cells grown on 2D surfaces.
Examining the immunomodulatory capabilities of MEECs revealed, MEECs were able to reduce the recruitment
of monocytes to an inflamed endothelial monolayer by 5-fold compared to EC on 2D surfaces.
An analysis of secreted factors from the cells revealed
- an 8-fold lower release of Monocyte Chemotactic Protein-1 (MCP-1) from MEECs.
Differences between 3D and 2D cultured cells were abolished in the presence of
- inhibitors to the focal adhesion associated signaling molecule Src, suggesting that
- adhesion-mediated signaling is essential in controlling the potent immunomodulatory
effects of MEEC. (4)
Cardiogenesis is regulated by a complex interplay between transcription factors. How do these interactions
regulate the transition from mesodermal precursors to cardiac progenitor cells (CPCs)?
Yin Yang 1 (YY1), a member of the GLI-Kruppel
- family of DNA-binding zinc finger transcription factor (TF), can
- activate or inhibit transcription in a context-dependent manner.
Bioinformatic-based Transcription Factor Genome-wide Sequencing Analysis
These investigators performed a bioinformatic-based transcription factor genome-wide sequencing analysis
- binding site analysis on upstream promoter regions of genes that are enriched in embryonic stem cell–derived CPCs
- to identify novel regulators of mesodermal cardiac lineage
From 32 candidate transcription factors screened, they found that
- Yin Yang 1 (YY1), a repressor of sarcomeric gene expression, is present in CPCs.
They uncovered the ability of YY1 to transcriptionally activate Nkx2.5,
- Nkx2.5 as a key marker of early cardiogenic commitment.
- YY1 regulates Nkx2.5 expression via a 2.1-kb cardiac-specific enhancer as demonstrated by in vitro
- luciferase-based assays,
- in vivo chromatin immunoprecipitation,
- and genome-wide sequencing analysis.
Furthermore, the ability of YY1 to activate Nkx2.5 expression depends on its cooperative interaction with Gata4.
Cardiac mesoderm–specific loss-of-function of YY1 resulted in early embryonic lethality.
This was corroborated in vitro by embryonic stem cell–based assays which showed the
- overexpression of YY1 enhanced the cardiogenic differentiation of embryonic stem cells into CPCs.
The results indicate an essential and unexpected role for YY1
- to promote cardiogenesis as a transcriptional activator of Nkx2.5
- and other CPC-enriched genes. (5)
Proportional Hazards Models to Analyze First-onset of Major
Cardiovascular Disease Events
Various measures of arterial stiffness and wave reflection are considered to be cardiovascular risk markers.
Prior studies have not assessed relations of a comprehensive panel of stiffness measures to prognosis
Authors used Proportional Hazards Models to analyze first-onset of major cardiovascular disease events
- myocardial infarction,
- unstable angina,
- heart failure, or
- stroke
In relation to arterial stiffness measured by
- pulse wave velocity [PWV]
- wave reflection
- augmentation index [AI]
- carotid-brachial pressure amplification [PPA]
- and central pulse pressure [CPP]
in 2232 participants (mean age, 63 years; 58% women) in the Framingham Heart Study.
During median follow-up of 7.8 (range, 0.2 to 8.9) years,
- 151 of 2232 participants (6.8%) experienced an event.
In multivariable models adjusted for
- age,
- sex,
- systolic blood pressure,
- use of antihypertensive therapy,
- total and high-density lipoprotein cholesterol concentrations,
- smoking, and
- presence of diabetes mellitus,
Higher aortic PWV was associated with a 48% increase in
- cardiovascular disease risk
(95% confidence interval, 1.16 to 1.91 per SD; P0.002).
After PWV was added to a standard risk factor model,
- integrated discrimination improvement was 0.7%
(95% confidence interval, 0.05% to 1.3%; P < 0.05).
In contrast, AI, CPP, and PPA were not related to
- cardiovascular disease outcomes in multivariable models.
(1) Higher aortic stiffness assessed by PWV is associated with
- increased risk for a first cardiovascular event.
(2) Aortic PWV improves risk prediction when added to standard risk factors
- and may represent a valuable biomarker of CVD risk in the community. (6)
1. Engineered arterial models to correlate blood flow to tissue biological response. J Martorell, P Santoma, JJ Molins,
AA Garcıa-Granada, JA Bea, et al. Ann NY Acad Sci 2012: 1254:51–56. (Issue: Evolving Challenges in Promoting
Cardiovascular Health) http://dx.doi.org/10.1111/j.1749-6632.2012.06518.x
2. Vascular Tissue Engineering. Designer Arteries. Elazer R. Edelman. Circ Res. 1999; 85:1115-1117
http://www.circresaha.org http://dx.doi.org/10.1161/01.RES.85.12
3. Tissue-engineered endothelial and epithelial implants differentially and synergistically regulate airway repair.
BG Zani, K Kojima, CA Vacanti, and ER Edelman. PNAS 13, 2008; 105(19):7046–7051.
http://www.pnas.org/cgi/doi/10.1073/pnas.0802463105
4. The role of scaffold microarchitecture in engineering endothelial cell immunomodulation.
L Indolfi, AB Baker, ER Edelman. Biomaterials 2012; http://dx.doi.org/10.1016/j.biomaterials.2012.06.052
5. Essential and Unexpected Role of Yin Yang 1 to Promote Mesodermal Cardiac Differentiation. S Gregoire, R Karra,
D Passer, Marcus-André Deutsch, et al. Circ Res. 2013;112:900-910. http://dx.doi.org/10.1161/CIRCRESAHA.113.259259
http://circres.ahajournals.org/doi:10.1161/CIRCRESAHA.113.259259
6. Arterial Stiffness and Cardiovascular Events. The Framingham Heart Study.
GF Mitchell, Shih-Jen Hwang, RS Vasan, MG Larson, et al. Circulation. 2010;121:505-511.
http://circ.ahajournals.org/doi/10.1161/CIRCULATIONAHA.109.886655
Cardiology Diagnosis of ACS and Stents – 2012
The Year in Cardiology 2012: Acute Coronary Syndromes.
Nick E.J. West http://www.medscape.com/viewarticle/779039
The European Society of Cardiology (ESC) produced updated guidance on management of STEMI in 2012.
It also produced a third version of the Universal Definition of Myocardial Infarction.
The importance of early diagnosis is stressed, with first ECG in patients
- with suspected STEMI recommended within 10 min of first medical contact (FMC)
- and primary percutaneous coronary intervention (PPCI) for STEMI
- ideally within 90 min (rated ‘acceptable’ out to a maximum of 120 min).
The guidance highlights the importance of collaborative networks
- to facilitate achievement of such targets.
- the importance of prompt assessment
- management of atypical presentations not always considered under the umbrella of STEMI, including
- left bundle branch block (LBBB),
- paced rhythms, and
- isolated ST-segment elevation in lead aVR,
especially when accompanied by symptoms consistent with myocardial ischaemia.
Therapeutic hypothermia is now recommended for
- all resuscitated patients with STEMI complicated by cardiac arrest
- immediate coronary angiography with a view to follow-on PPCI
- when the ECG demonstrates persistent ST-segment elevation.
In the light of recently published studies and meta-analyses,
- including that of Kalesan et al., drug-eluting stents (DES) are
- now routinely preferred to bare metal stents (BMS) in view of
- the reduced need for repeat revascularization and the lack of
- previously perceived hazard for stent thrombosis.
The more potent antiplatelet agents prasugrel and ticagrelor are also preferred
- to clopidogrel for all STEMI cases, with duration of dual antiplatelet therapy (DAPT)
- ideally for 1 year, but reduced to a strict
- minimum of 6 months for patients receiving DES.
The Third Universal Definition of Myocardial Infarction was published
simultaneously with the STEMI guidance. This guideline endorses
- cardiac troponin as the biomarker of choice to detect myocardial necrosis
- with spontaneously occurring myocardial infarction (MI) defined as an
- elevation above the 99th percentile upper reference value for the assay.
There is further development and clarification of MI in different settings
- to allow standardization across trials and registries
in particular after revascularization procedures: after CABG with normal baseline troponin
- MI is defined as a rise to a value 10 times greater than baseline in the first 48 h, and
- a rise to 5 times greater than 99th percentile upper reference after PCI
in patients with a normal baseline level (or a 20% rise when troponin is elevated and stable or falling pre-procedure).
ACCF/AHA updated guidance on the management of unstable angina/non-STEMI:
angiography with a view to revascularization
- is now recommended within 12–24 h of presentation, with
- DAPT pre-loading prior to PCI procedures also now advocated.
Ticagrelor and prasugrel are cited as acceptable alternatives to clopidogrel.
The maintenance dose of aspirin recommended for the majority of cases is 81 mg daily.
This guideline brings about transatlantic agreement in most areas.
Risk Stratification
Identification and appropriate triage of patients presenting to emergency departments
with acute chest pain remains a difficult dilemma:
- many are low-risk and have a non-cardiac origin
- a significant minority with coronary artery disease may not be picked up
on clinical grounds even when accompanied by appropriate tests,
- including ECG and biomarker estimation used in conjunction
- with a clinical risk score (e.g. GRACE, TIMI).
As endorsed in ESC guidance, there has been increasing interest in
- non-typical ECG patterns for the diagnosis of STEMI; although LBBB is
- an accepted surrogate
Widimsky et al. retrospectively analysed 6742 patients admitted to hospital with acute MI
- in patients presenting with right bundle branch block, a blocked epicardial vessel was
- more common (51.7 vs. 39.4%; P < 0.001) and incidence of both shock and mortality
- comparable with LBBB (14.3 vs. 13.1%; P = NS; and 15.8 vs. 15.4%; P = NS, respectively).
Wong et al. demonstrated the importance of ST-elevation in lead aVR,
- often viewed as indicative of left main stem occlusion, having increased mortality
- in patients presenting with both inferior and anterior infarction.
Perhaps the most important data regarding the ECG in 2012 were also the most simple:
- Antoni et al. highlighted a powerful and very simple method of risk stratification;
- heart rate measured on a 12-lead ECG at discharge after Primary PCI (PPCI) is an
- independent predictor of mortality at 1 and 4 years of follow-up.
Patients with a discharge heart rate of ≥70 b.p.m. had a two-fold higher mortality at both follow-up
time points, with every increase of 5 b.p.m. in heart rate
- equating to a 29% increase in mortality at 1 year and 24% at 5 years.
These findings have important implications for the optimization of patient therapies after MI (including the use of
rate-limiting agents such as beta-blockers, calcium channel-blockers, and ivabradine), although large randomized
trials are needed to confirm that
- interventions to reduce heart rate will replicate the benefits observed in this study.
http://img.medscape.com/article/779/039/779039-thumb1.png
Figure 1. Kaplan–Meier time-to-event plots for heart rate at discharge divided by quartiles and all-cause mortality
(A and C) and cardiovascular mortality (B and D) at 1-year (A and B) and 4-year (C and D) follow-up,
demonstrating relationship between discharge heart rate and mortality after PPCI for STEMI.
Modified from Antoni et al.
Coronary Intervention and Cardioprotection in Acute Coronary Syndromes
Microvascular obstruction during PCI for ACS/STEMI is associated with increased infarct size and adverse prognosis;
its pathophysiology is thought to be a combination of
- mechanical distal embolization of thrombus and plaque constituents during PCI, coupled with
- enhanced constriction/hyperreactivity of the distal vascular bed.
The most novel Strategy to Reduce Infarct Size
is the use of a Bare Metal Stent (BMS) covered on its outer surface with a mesh micronet designed to
trap and hold potentially friable material that might embolize distally at the time of PCI.
The MASTER study randomized 433 STEMI patients to PPCI
- with conventional BMS or DES at the operator’s discretion vs.
- the novel MGuard stent (InspireMD, Tel Aviv, Israel);
the primary endpoint of complete ST-segment resolution was better
- in patients receiving MGuard (57.85 vs. 44.7%; P = 0.008), as was
- the achievement of TIMI grade 3 flow in the treated vessel (91.7 vs. 82.9%; P = 0.006).
Nevertheless, median ST-segment resolution did not differ
- between treatment groups,
- myocardial blush grade was no different, and
- safety outcomes at 30 days (death, adverse events) as well as
- overall MRI-determined infarct mass.
Higher TVR rates may accrue with a BMS platform when compared with
- current-generation DES (as now endorsed for PPCI in ESC guidance).
In comparing the four studies in cardioprotection, there remains little to choose between strategies as evidenced by
- the relatively minor differences between surrogate endpoints employed regardless of
- therapeutic intervention chosen (Figure 2).
http://img.medscape.com/article/779/039/779039-fig2.jpg
Figure 2. Comparison of study endpoints for reduction in infarct size in STEMI.
Study endpoints listed on the x-axis. STR, ST-segment resolution; TIMI 3, thrombolysis in
myocardial infarction grade 3 antegrade flow; myocardial blush grade 2/3 (MBG 2/3).
Recent advances in
- PCI equipment,
- peri-procedural pharmacology,
- technique, and safety, as well as
- convergence of national guidance,
are leading to the point where
- even in the highest risk patients such as those presenting with ACS, small improvements
- may be difficult to discern despite large well-designed and -conducted studies.
References
- a. The Task Force on the management of ST-segment elevation acute myocardial infarction
of the European Society of Cardiology. ESC guidelines for the management of acute
myocardial infarction in patients presenting with ST-segment elevation. Eur Heart J
2012;33:2569–2619. b. Management of acute myocardial infarction in patients presenting
with ST-segment elevation. The Task Force on the Management of Acute Myocardial
Infarction of the European Society of Cardiology. Eur Heart J 2003; 24 (1): 28-66.
http://dx.doi.org/10.1093/eurheartj/ehs215
- ESC Guidelines for the management of acute coronary syndromes in patients presenting
without persistent ST-segment elevation: The Task Force for the management of acute
coronary syndromes (ACS) in patients presenting without persistent ST-segment elevation
of the European Society of Cardiology (ESC). http://dx.doi.org/10.1093/eurheartj/ehr236
- Thygesen K, Alpert JS, Jaffe AS, Simoons ML, Chaitman BS, White HD. The Writing Group on
behalf of the Joint ESC/ACCF/AHA/WHF Task Force for the Universal Definition of
Myocardial Infarction. Third universal definition of myocardial infarction.
Eur Heart J 2012;33:2551–2567. http://dx.doi.org/10.1093/eurheartj/ehm355
- Kalesan B, Pilgrim T, Heinimann K, Raber L, Stefanini GG, et al. Comparison of drug-eluting
stents with bare metal stents in patients with ST-segment elevation myocardial infarction.
Eur Heart J 2012;33:977–987.
- Jneid H, Anderson JL, Wright RS, Adams CS, et al. 2012 ACCF/AHA Focused Update of the
Guideline for the Management of Patients with Unstable Angina/Non-ST-Elevation Myocardial
Infraction (Updating the 2007 Guideline and Replacing the 2011 Focused Update). A Report
of the American College of CardiologyFoundation/American Heart Association Task Force
on Practice Guidelines. J Am Coll Cardiol 2012;60:645–681.
- Widimsky P, Rohác F, Stásek J, Kala P, Rokyta R, et al. Primary angioplasty in acute myocardial
infarction with right bundle branch block: should new onset right bundle branch block be added
to future guidelines as an indication for reperfusion therapy? Eur HeartJ 2012;33:86–95.
- Wong CK, Gao W, Stewart RA, French JK, and the HERO-2 Investigators. The prognostic meaning of
the full spectrum of aVR ST-segment changes in acute myocardial infarction.
Eur Heart J 2012;33:384–392.
- Antoni L, Boden H, Delgado V, Boersma E, et al. Relationship between discharge heart rate and mortality
in patients after myocardial infarction treated with primary percutaneous coronary intervention.
Eur Heart J 2012;33:96–102.
- Stone GW, Abizaid A, Silber S, Dizon JM, Merkely B, et al. Prospective, randomised, multicenter evaluation
of a polyethylene terephthalate micronet mesh-covered stent (MGuard) in ST-segment elevation myocardial
infarction. The MASTER Trial. J Am Coll Cardiol. doi:pii:S0735-1097(12)04506-8. 10.1016/j.jacc.2012.09.004.
- Zhou C, Yao Y, Zheng Z, Gong J, Wang W, Hu S, Li L. Stenting technique, gender, and age are associated with
cardioprotection by ischaemic postconditioning in primary coronary intervention: a systematic review of
10 randomized trials. Eur Heart J 2012;33:3070–3077.
Resistant Hypertension.
Robert M. Carey.
Hypertension. 2013;61:746-750. http://dx.doi.org/10.1161/HYPERTENSIONAHA.111.00601
Resistant hypertension is defined as failure to achieve goal blood pressure (BP) <140/90 mm Hg
(or <130/80 mm Hg in patients with diabetes mellitus or chronic kidney disease) in patients with
- hypertension who are compliant with maximum tolerated doses of an appropriate antihypertensive drug regimen consisting of a minimum of 3 agents of different classes, including a diuretic.
- Patients who meet the criteria for resistant hypertension but whose BP can be controlled on maximum tolerated
doses of ≥4 antihypertensive agents are classified as having controlled resistant hypertension.
Although the number of failed antihypertensive drugs required for the classification of resistant hypertension is arbitrary,
- this diagnosis identifies patients at high risk for having a potentially curable form of hypertension, and
- those who may benefit from specific therapeutic approaches to lower BP.
Summary
The first portion of this document shows the impact that ER Edelman and his peers have had in the development
of interventional cardiology, and in carrying out studies to test, validate, or reject assumptions about the interaction of
biomaterials with
- vascular and smooth muscle tissue in the repair of injured vessels, by
- trauma
- inflammatory injury
- stent placement.
In the second portion of this discussion, I introduce current views about complications in implanted devices, evolving
standards, and the current definitions of stable, unstable, and previously unclassified ACS risk.
Pushing Drug-Eluting Stents Into Uncharted Territory
Simpler Than You Think—More Complex Than You Imagine
Campbell Rogers, MD; Elazer R. Edelman, MD, PhD. Circulation 2006; 113: 2262-2265.
http://dx.doi.org/10.1161/CIRCULATIONAHA.106.623470
Mechanical failure is a characteristic of a material or a device and not necessarily an indication of inadequacy. All devices
will fail under some specific stress. It is only failure at the lowest levels of stress that may represent inadequacy. Stress on
a material, for example, rises with strain until a critical load is exceeded, at which point the material fatigues and loses
mechanical integrity. Failure analysis, the science by which these conditions are rigorously defined, is an important
component of device design, development, and use. Once the transition point to failure is identified, material use can be
restricted to the zone of safety or modified so as to have this zone expanded. Just as the characterization of a material is
incomplete unless pushed to the limits of load bearing, characterization of an implantable device is incomplete unless
preclinical and clinical environments test the limits of device functionality. It was in this light in 1999 that the Authors noted the impossibility of defining the functional limits of novel bare metal stents in head-to-head trials, which, by necessity, could only include lesions into which the predicate device (the Palmaz-Schatz stent, Cordis, Warren, NJ) could have be placed.
New School Percutaneous Interventions
Over the past 5 years, the number of percutaneous interventions has grown by 40%. This expansion derives from an
increased breadth of cases, as percutaneous interventions are now routinely performed in diabetic, small-vessel, multilesion,
diffuse disease, and acute coronary syndrome settings. Contemporaneously, widespread adoption of drug-eluting stents has emboldened clinicians and provided greater security in the use of these devices in lesions or patients previously thought to
Head-to-head randomized trial data have accumulated so that analysis may demonstrate differences among drug-eluting stents. The playing field for prospective randomized trials could enhance the weight of evidence to unanswered questions about what underlying factors determine device failure.
Complexity Simplified
Drug-eluting stent “failure” can be defined operationally in the same way as material failure:
- inadequate function in the setting of a given load or strain.
The inability to withstand stress may take many forms that can change over time. Failure may be manifest acutely as
- the inability to deliver a stent to the desired location,
- subacutely as stent thrombosis or
- postprocedural myonecrosis, and later as
- restenosis
“Simple lesions” are those in which few devices should fail;“Complex” lesions have a heightened risk of failure. To be of value, each scale of advancing complexity must provoke higher failure rates. For any device may fail sooner than another along one such “complexity” scale and later along another. As advanced drug-eluting stent designs have enhanced deliverability and reduced restenosis rates, 7 randomized trials comparing directly the two Food and Drug Administration (FDA)-approved drug-eluting stents, Cypher (Cordis-Johnson and Johnson) and Taxus (Boston Scientific, Boston, Mass), have been reported. These trials report a broad range of restenotic failure as evidenced by the need for revascularization. Across these trials, driven by a variety of factors, revascularization rates vary quite widely.
The clinical end point of target lesion revascularization (TLR) becomes
- a single measure of device failure.
When the 7 trials are depicted in order of increasing TLR, the rate of failure increases more slowly with 1 device than
the other. This gives two regression plots for Taxus vs Cypher with different slopes, as complexity increases, and the
- separation between the failure rates of the two devices broadens plotted against “degree of complexity” assigned by the slopes of the lines.
Finally, the correlation between TLR rates for Taxus and Cypher stents indicates that trial-specific events and conditions determined TLR (with a sharp slope of Taxus vs Cypher (r-sq = 0.85). The ratio of TLR (the slope) wasgreater than 3, suggesting that although both devices are subject to increasing failure as complexity increases,
- one device becomes ever-more likely than the other to fail when applied in settings with ever-higher TLR risk.
In other words, composite medical devices with a wide range of
- structural,
- geometric, and
- pharmacological differences
- can be shown to produce different clinical effects
- as the environments in which they are tested become increasingly complex.
What the Individual Trials Cannot Tell Us
The progressive difference between the performances of the 2 FDA-approved drug-eluting stents as they are pushed into
more complex settings is precisely what one would anticipate from medical devices with different performance signatures.
Most randomized trials, even if they include high complexity, are unable to identify predictors of failure because of the low numbers of patients enrolled, and the problem gets worse as the number of subsets increase. Consequently, device development, and clinical practice, knowing which patient or lesion characteristics confer higher failure rates is critical.
This analysis has centered on restenosis. Other failure modes to be considered are
- stent thrombosis,
- postprocedural myonecrosis
- late plaque rupture
- vascular disease away from the site
- heightened inflammatory reaction
- are no less critical and may be determined by
- completely different device or patient characteristics.
Well-executed registry or pooled data
It is in this light that the registry report of Kastrati et al. in the current issue of Circulation is of greatest value. There are
two ways in which well-executed registry or pooled data can be most complementary to randomized trials.
First, large numbers of patients provide a higher incidence of rare failure modes as well as allow more granular determination of lesion- or patient-specific predictors of failure (meta-analysis or better, combined data file). A pooled analysis of several head-to-head randomized bare metal stent trials allowed identification of clear risk factors for stent thrombosis that had eluded analysis of the individual (smaller) trials.
Second, registry or pooled data may incorporate a broader range of patient characteristics, allowing greater discrimination between devices. The report of Kastrati et al may fall into this category as well, as it includes “high risk” populations from several randomized trials. They report on more than 2000 lesions in 1845 patients treated with either Taxus or Cypher drug-eluting stents at two hospitals. The study population is from a series of randomized trials comparing Taxus and Cypher stents. Using multivariate analysis to identify what lesion and patient characteristics predict failure (restenosis), they identified risk factors that included
- prior history of coronary bypass surgery
- calcification
- smaller vessel size
- greater degree of prestent and poststent stenosis.
Use of a Cypher rather than Taxus stent was independently associated with lower restenosis risk.
An interesting negative finding was the absence of diabetes as a significant predictor, at odds with strong suggestions from several other analyses. A better understanding from preclinical or clinical studies of the effect of diabetic states on restenosis is critical.
Author’s opinion voiced:
This Author (LHB), considers the study underpowered to answer that question because of further partitioning with several variables. Pooled data with
- rigorous ascertainment and
- careful statistical methodology, taken
- together with randomized trial data, open a door to device choice based on the knowledge that risk of failure (complexity) does vary, and
- the higher the complexity, the greater the incremental benefit of choosing one device over another.
A decision algorithm is therefore possible, whereby multiple failure modes and risk factors are weighed, and
- an optimum stent choice made which balances
- safety and efficacy based on the totality of evidence, rather than anecdote and loose comparisons of disparate subgroups from individual trials.
Evaluating Clinical Trials
The subject of trial(s) is difficult… the aim and meaning of all the trials… is
- to let people know what they ought to do or what they must believe
It was perhaps naïve to imagine that devices as different one from another as the two current FDA-approved drug-eluting
stents would produce identical clinical results. If so, it ought not to come as a surprise that head-to-head randomized trial
data from many different countries in complex settings are now indicating just how differently the 2 devices may perform.
Future trials should be designed and evaluated to examine why these differences exist. Trials residing
only in previous safety and complexity domains
- are unlikely to offer deeper insights into
- device performance,
- patient care decisions, or
- discrimination of alternative therapies.
We look forward to more trials that will examine what we currently believe to be the limits of
- drug-eluting stents and interventional cardiology and to
help define in simple terms differences
- between complex devices applied to complex problems.
This 2009 article was an excellent demonstration of comparing two commonly used coated-stents, and then extending the argument to the need for more data to further delineated the factors that explain the differences they found. In the previous article, the SECOND in the three article series, Stents and Drug Delivery
we concentrated on stents and drug delivery, and not on stent failure. But the following article in J Control Release,
was published the following year, and is another example of this method of explanatory approach to the problem.
Lesion Complexity Determines Arterial Drug Distribution After Local Drug Delivery
AR Tzafriri, N Vukmirovic, VB Kolachalama, I Astafieva, ER Edelman. J Control Release. 2010; 142(3): 332–338.
http://:dx. doi:.org/10.1016/j.jconrel.2009.11.007 PMCID: PMC2994187
Local drug delivery from endovascular stents has transformed how we treat coronary artery disease. Yet, few drugs are in fact effective when delivered from endovascular implants and those that possess a narrow therapeutic window. The width of this window is predicated to a great degree upon the extent of drug deposition and distribution through the arterial wall.
- Drugs that are retained within the blood vessel are far more effective than those that are not.
Thus, for example, heparin regulates virtually every aspect of the vascular response to injury, but it is so soluble and diffusible that it simply cannot stay in the artery for more than minutes after release.
- Heparin has no effect on intimal hyperplasia when eluted from a stent.
- Paclitaxel and sirolimus in contradistinction are far smaller compounds with perhaps more narrow and specific effects than heparin.
These drugs bind tenaciously to tissue protein elements and specific intracellular targets and remain beneath stent struts long after release.
The clinical efficacy of paclitaxel and sirolimus at reducing coronary artery restenosis rates following elution from stents appears incontrovertible. Emerging clinical and preclinical data suggest that the benefit of the local release of these drugs is beset by significant complications, that rise with lesion complexity as
- the native composition and layered ultrastructure of the native artery is more significantly disrupted.
Virmani and others have hypothesized that the attraction of lipophilic drugs like paclitaxel and sirolimus to fat should affect their retention within and effects upon atheromatous lesions.
Though stents are deployed in diseased arteries drug distribution has only been quantified in intact, non-diseased vessels.
Authors @ MIT, correlated steady-state arterial drug distribution with tissue ultrastructure and composition in abdominal aortae from atherosclerotic human autopsy specimens and rabbits
- with lesions induced by dietary manipulation and controlled injury.
Drug and compositional metrics were quantified and correlated at a compartmental level, in each of the tunica layers, or at an intra-compartmental level. All images were processed to
- eliminate backgrounds and artifacts, and
- pixel values between thresholds were extracted for all zones of interest.
Specific algorithms analyzed each of the histo/immuno-stained arterial structures. Intra-compartmental analyses were
- performed by sub-dividing arterial cross-sections into 2–64 equal sectors and
- evaluating the pixel-average luminosity for each sector.
Linear regression of drug versus compositional luminosities asymptotically approached steady state after subdivision into 16 sectors. This system controlled delivered dose and removed the significant unpredictability in release that is imposed by variability
- in stent position relative to the arterial wall,
- inflation techniques and stent geometry.
As steady state tissue distribution results were obtained under constant source conditions, without washout by flowing blood,
- they constitute upper bounds for arterial drug distribution
- following transient modes of in vivo drug delivery wherein
- only a fraction of the eluted dose is absorbed by the artery
Paclitaxel, everolimus, and sirolimus deposition in human aortae was maximal in the media and scaled inversely with lipid content.
Net tissue paclitaxel and everolimus levels were indistinguishable in mildly injured rabbit arteries independent of diet. Yet, serial sectioning of cryopreserved arterial segments demonstrated
- a differential transmural deposition pattern that was amplified with disease and
- correlated with expression of their intracellular targets, tubulin and FKBP-12.
Tubulin distribution and paclitaxel binding increased with
- vascular injury and macrophage infiltration, and
- were reduced with (reduced) lipid content.
Sirolimus analogues and their specific binding target FKBP-12 were less sensitive to alterations of diet
in mildly injured arteries, presumably reflecting a faster transient response of FKBP-12 to injury.
The idea that drug deposition after balloon inflation and stent implantation within diseased, atheromatous and sclerotic vessels tracks so precisely with specific tissue elements is
- an important consideration of drug-eluting technologies and
- may well require that we consider diseased rather than naïve tissues in preclinical evaluations.
Another publication in the same year reveals the immense analytical power used in understanding the complexities
of drug-eluting stents.
Luminal Flow Amplifies Stent-Based Drug Deposition in Arterial Bifurcations
Kolachalama VB, Levine EG, Edelman ER. PLoS ONE 2009; 4(12): e8105.
http://dx.doi.org/10.1371/journal.pone.0008105
Treatment of arterial bifurcation lesions using drug-eluting stents (DES) is now common clinical practice.
Arterial drug distribution patterns become challenging to analyze if the lesion involves more than a vessel
such as in the case of bifurcations. As use extends to nonstraightforward lesions and complex geometries,
questions abound
- regarding DES longevity and safety
Indeed, there is no consensus on best stent placement scenario, no understanding as to
- whether DES will behave in bifurcations as they do in straight segments, and
- whether drug from a main-branch (MB) stent can be deposited within a side-branch (SB).
It is not evident how to
- efficiently determine the efficacy of local drug delivery and
- quantify zones of excessive drug that are
- harbingers of vascular toxicity and thrombosis,
- and areas of depletion that are associated
- with tissue overgrowth and
- luminal re-narrowing.
Geometry modeling and governing equations
Authors @MIT constructed two-phase computational models of stent-deployed arterial bifurcations
- simulating blood flow and drug transport to investigate the
- factors modulating drug distribution when the main-branch (MB) was treated using a DES.
The framework for constructing physiologically realistic three dimensional computational models of single
and bifurcated arterial vessels was SolidWorks (Dassault Systemes) (Figs. 1A–1B, Movie S1). The geometry
generation algorithm allowed for controlled alteration of several parameters including
- stent location
- strut dimensions
- stent-cell shape
- lumen diameter to arterial tissue thickness ratio
- lengths of the arterial branches
- extent of stent apposition and
- the bifurcation angle.
For the current study, equal lengths (2LS) were assumed for the proximal and distal sections of the MB from the bifurcation. The SB was constructed at an angle of 300. The inlet conditions were based on
- mean blood flow and
- diameter measurements
obtained from human left anterior descending coronary artery (LAD).
The diameter of the lumen (DMB) and thickness (TMB) for the MB were defined such that DMB=TMB~10 and
- this ratio was also maintained for the SB.
Schematics of the computational models used for the study. A stent of length LS is placed at the upstream section of the arterial vessel in the (A) absence and in the (B) presence of a bifurcation, respectively.
- Insets in (B) denote delta wing stent design (i),
- strut thickness (d) (ii), and
- the outlets of the side-branch in (iii) and
- and the main-branch in (iv).
A delta wing-shaped cell design belonging to the class of slotted-tube stents was used for all simulations.
The length (LS) and diameter (DS) were
- fixed at 9|10-2 m and 3|10-2 m, respectively, for the MB stent.
All stents were assumed to be perfectly apposed to the lumen of MB and the intrinsic strut shape was modeled as
- square with length 10-4 m.
The continuity and momentum equations were solved within the arterial lumen, where
vf , rho~1060 kg=m3, P and m are
- velocity
- density
- pressure and the
- viscosity of blood.
In order to capture boundary layer effects at the lumen-wall (or mural) surface, a Carreau model was employed for
- all the simulations to account for shear thinning behavior of blood at low shear rates
In the arterial lumen, drug transport followed advection-diffusion process. Similar to the momentum transport in the arterial lumen, the continuity equation was solved within the arterial wall by assuming it as a porous medium.
A finite volume solver (Fluent, ANSYS Inc.) was utilized to perform the coupled flow and drug transport simulations. The semi-implicit method for pressure-linked equations-consistent (SIMPLEC) algorithm was used with second order spatial accuracy. A second order discretization scheme was used to solve the pressure equation and second order upwind schemes were used for the momentum and concentration variables.
Simulations for each case were performed
- for at least 2500 iterations or
- until there was a 1028 reduction in the mass transport residual.
Drug distribution in non-bifurcating vessels
Constant flow simulations generate local recirculation zones juxtaposed to the stent which in turn act as
- secondary sources of drug deposition and
- induce an asymmetric tissue drug distribution profile in the longitudinal flow direction.
Our3D computational model predicts a far more extensive fluid mechanic effect on drug deposition than previously appreciated in two-dimensional (2D) domains.
Within the stented region, drug deposition on the mural interface quantified as
- the area-weighted average drug concentration (AWAC)
- in the distal segment of the stent is 12% higher than the proximal segment
Total drug uptake in the arterial wall denote as volume-weighted average concentration (VWAC) is highest in the middle segment of the stent and 5% higher than the proximal stent region
Increased mural drug deposition along the flow direction in a non-bifurcating arterial vessel.
Inset shows a high magnification image of drug pattern in the distal stent segment outlined by black dashed line.
The entire stent is divided into three equal sections denoted as proximal, middle and distal sections, respectively
and the same notation is followed for subsequent analyses.
http://dx.doi.org/10.1371/journal.pone.0008105.g002
These observations indicate that the flow-mediated effect induced by the presence of the stent in the artery
- is maximal on the mural surface and
- increases in the longitudinal flow direction.
Further, these results suggest that transmural diffusion-mediated transport sequesters drug from both
- the proximal and distal portions of the stent
- into the central segment of the arterial wall beneath the stent.
Predicted levels of average drug concentration varied exponentially
- with linear increments of inlet flow rate
but maintained similar relationship between the inter-segment concentration levels within the stented region.
Stent position influences drug distribution in bifurcated beds
The location of the stent directly modulates
- the extent to which drug is deposited on the arterial wall as well as
- spatial gradients that are established in arterial drug distribution.
Similar to the non-bifurcating vessel case,
- peaks in drug deposition occur directly beneath the stent struts regardless of the relative location of the SB with respect to the stent. However,
- drug distribution and corresponding spatial heterogeneity within inter-strut regions depend on the stent location with respect to the flow divider.
- Mural drug deposition is a function of relative stent position with respect to the side-branch and Reynolds number in arterial bifurcations.
Impact of flow on drug distribution in bifurcations
One can appreciate how blood flow and flow dividers affect arterial drug deposition, and especially on inter-strut drug deposition.
- Drug deposition within the stented-region of MB and the entire SB significantly decreases with flow acceleration regardless of stent placement.
- extensive flow-mediated drug delivery in bifurcated vascular beds where the drug distribution patterns are heterogeneous and sensitive to relative stent position and luminal flow.
A single DES in the MB coupled with large retrograde luminal flow on the lateral wall of the side-branch (SB) can provide drug deposition on the SB lumen-wall interface, except
- when the MB stent is downstream of the SB flow divider.
- the presence of the SB affects drug distribution in the stented MB.
Fluid mechanic effects play an even greater role than in the SB
Summary
We presented the hemodynamic effects on drug distribution patterns using a
- simplified uniform-cell stent design, though our methodology is adaptable to
several types of stents with variable design features.
Variability in arterial drug distribution due to other geometric and morphologic aspects such as
- bifurcation angle, arterial taper as well as presence of a trifurcation can also be understood using our computational framework.
Further, performance of a candidate DES using other commonly used stenting procedures for bifurcation lesions such as culotte and crush techniques can be quantified based on their resulting drug distribution patterns.
Other Related Articles that were published on this Open Access Online Scientific Journal include the following:
Vascular Repair: Stents and Biologically Active Implants
Larry H Bernstein, MD, FACP and Aviva Lev-Ari, RN,
PhD, 5/4/2013
Modeling Targeted Therapy
Larry H Bernstein, MD, FACP 3/2/2013
Quantum Biology And Computational Medicine
Larry H Bernstein, MD, FACP 4/3/2013
Virtual Biopsy – is it possible?
Larry H Bernstein, MD, FACP 3/3/2013
Reprogramming cell fate 3/2/2013
Larry H Bernstein, MD, FACP
How Methionine Imbalance with Sulfur-Insufficiency Leads to Hyperhomocysteinemia
Larry H Bernstein, MD, FACP 4/4/2013
Amyloidosis with Cardiomyopathy
Larry H Bernstein, MD, FACP 3/31/2013
Nitric Oxide, Platelets, Endothelium and Hemostasis
Larry H Bernstein, MD, FACP 11/8/2012
Mitochondrial Damage and Repair under Oxidative Stress
Larry H Bernstein, MD, FACP 10/28/2012
Endothelial Function and Cardiovascular Disease
Larry H Bernstein, MD, FACP 10/25/2012
Endothelial Dysfunction, Diminished Availability of cEPCs, Increasing CVD Risk for Macrovascular Disease –Therapeutic Potential of cEPCs
Aviva Lev-Ari, PhD, RN 8/27/2012
Prostacyclin and Nitric Oxide: Adventures in Vascular Biology – A Tale of Two Mediators
Aviva Lev-Ari, RN, PhD, 4/30/2013
Genetics of Conduction Disease: Atrioventricular (AV) Conduction Disease (block): Gene Mutations – Transcription, Excitability, and Energy Homeostasis
Aviva Lev-Ari, PhD, 4/28/2013
Genetics of Conduction Disease: Atrioventricular (AV) Conduction Disease (block): Gene Mutations – Transcription, Excitability, and Energy Homeostasis
Revascularization: PCI, Prior History of PCI vs CABG
Aviva Lev-Ari, PhD, 4/25/2013
Revascularization: PCI, Prior History of PCI vs CABG
Revascularization: PCI, Prior History of PCI vs CABG
Aviva Lev-Ari, PhD, RN 4/25/2013
http://pharmaceuticalintelligence.com/2013/04/25/revascularization-pci-prior-history-of-pci-vs-cabg/
Cholesteryl Ester Transfer Protein (CETP) Inhibitor: Potential of Anacetrapib to treat Atherosclerosis and CAD
Aviva Lev-Ari, PhD, RN 4/7/2013
http://pharmaceuticalintelligence.com/2013/04/07/cholesteryl-ester-transfer-protein-cetp-inhibitor-potential-of-anacetrapib-to-treat-atherosclerosis-and-cad/
Hypertriglyceridemia concurrent Hyperlipidemia: Vertical Density Gradient Ultracentrifugation a Better Test to Prevent Undertreatment of High-Risk Cardiac Patients
Aviva Lev-Ari, PhD, RN 4/4/2013
http://pharmaceuticalintelligence.com/2013/04/04/hypertriglyceridemia-concurrent-hyperlipidemia-vertical-density-gradient-ultracentrifugation-a-better-test-to-prevent-undertreatment-of-high-risk-cardiac-patients/
Fight against Atherosclerotic Cardiovascular Disease: A Biologics not a Small Molecule – Recombinant Human lecithin-cholesterol acyltransferase (rhLCAT) attracted AstraZeneca to acquire AlphaCore
Aviva Lev-Ari, PhD, RN 4/3/2013
http://pharmaceuticalintelligence.com/2013/04/03/fight-against-atherosclerotic-cardiovascular-disease-a-biologics-not-a-small-molecule-recombinant-human-lecithin-cholesterol-acyltransferase-rhlcat-attracted-astrazeneca-to-acquire-alphacore/
High-Density Lipoprotein (HDL): An Independent Predictor of Endothelial Function & Atherosclerosis, A Modulator, An Agonist, A Biomarker for Cardiovascular Risk
Aviva Lev-Ari, PhD, RN 3/31/2013
http://pharmaceuticalintelligence.com/2013/03/31/high-density-lipoprotein-hdl-an-independent-predictor-of-endothelial-function-artherosclerosis-a-modulator-an-agonist-a-biomarker-for-cardiovascular-risk/
Acute Chest Pain/ER Admission: Three Emerging Alternatives to Angiography and PCI
Aviva Lev-Ari, PhD, RN 3/10/2013
http://pharmaceuticalintelligence.com/2013/03/10/acute-chest-painer-admission-three-emerging-alternatives-to-angiography-and-pci/
Genomics & Genetics of Cardiovascular Disease Diagnoses: A Literature Survey of AHA’s Circulation Cardiovascular Genetics, 3/2010 – 3/2013
Lev-Ari, A. and L H Bernstein 3/7/2013
http://pharmaceuticalintelligence.com/2013/03/07/genomics-genetics-of-cardiovascular-disease-diagnoses-a-literature-survey-of-ahas-circulation-cardiovascular-genetics-32010-32013/
The Heart: Vasculature Protection – A Concept-based Pharmacological Therapy including THYMOSIN
Aviva Lev-Ari, PhD, RN 2/28/2013
http://pharmaceuticalintelligence.com/2013/02/28/the-heart-vasculature-protection-a-concept-based-pharmacological-therapy-including-thymosin/
Arteriogenesis and Cardiac Repair: Two Biomaterials – Injectable Thymosin beta4 and Myocardial Matrix Hydrogel
Aviva Lev-Ari, PhD, RN 2/27/2013
http://pharmaceuticalintelligence.com/2013/02/27/arteriogenesis-and-cardiac-repair-two-biomaterials-injectable-thymosin-beta4-and-myocardial-matrix-hydrogel/
Coronary artery disease in symptomatic patients referred for coronary angiography: Predicted by Serum Protein Profiles
Aviva Lev-Ari, PhD, RN 12/29/2012
http://pharmaceuticalintelligence.com/2012/12/29/coronary-artery-disease-in-symptomatic-patients-referred-for-coronary-angiography-predicted-by-serum-protein-profiles/
Special Considerations in Blood Lipoproteins, Viscosity, Assessment and Treatment
Bernstein, HL and Lev-Ari, A. 11/28/2012
http://pharmaceuticalintelligence.com/2012/11/28/special-considerations-in-blood-lipoproteins-viscosity-assessment-and-treatment/
Peroxisome proliferator-activated receptor (PPAR-gamma) Receptors Activation: PPARγ transrepression for Angiogenesis in Cardiovascular Disease and PPARγ transactivation for Treatment of Diabetes
Aviva Lev-Ari, PhD, RN 11/13/2012
http://pharmaceuticalintelligence.com/2012/11/13/peroxisome-proliferator-activated-receptor-ppar-gamma-receptors-activation-pparγ-transrepression-for-angiogenesis-in-cardiovascular-disease-and-pparγ-transactivation-for-treatment-of-dia/
Clinical Trials Results for Endothelin System: Pathophysiological role in Chronic Heart Failure, Acute Coronary Syndromes and MI – Marker of Disease Severity or Genetic Determination?
Aviva Lev-Ari, PhD, RN 10/19/2012
http://pharmaceuticalintelligence.com/2012/10/19/clinical-trials-results-for-endothelin-system-pathophysiological-role-in-chronic-heart-failure-acute-coronary-syndromes-and-mi-marker-of-disease-severity-or-genetic-determination/
Endothelin Receptors in Cardiovascular Diseases: The Role of eNOS Stimulation
Aviva Lev-Ari, PhD, RN 10/4/2012
http://pharmaceuticalintelligence.com/2012/10/04/endothelin-receptors-in-cardiovascular-diseases-the-role-of-enos-stimulation/
Inhibition of ET-1, ETA and ETA-ETB, Induction of NO production, stimulation of eNOS and Treatment Regime with PPAR-gamma agonists (TZD): cEPCs Endogenous Augmentation for Cardiovascular Risk Reduction – A Bibliography
Aviva Lev-Ari, PhD, RN 10/4/2012
http://pharmaceuticalintelligence.com/2012/10/04/inhibition-of-et-1-eta-and-eta-etb-induction-of-no-production-and-stimulation-of-enos-and-treatment-regime-with-ppar-gamma-agonists-tzd-cepcs-endogenous-augmentation-for-cardiovascular-risk-reduc/
Positioning a Therapeutic Concept for Endogenous Augmentation of cEPCs — Therapeutic Indications for Macrovascular Disease: Coronary, Cerebrovascular and Peripheral
Aviva Lev-Ari, PhD, RN 8/29/2012
http://pharmaceuticalintelligence.com/2012/08/29/positioning-a-therapeutic-concept-for-endogenous-augmentation-of-cepcs-therapeutic-indications-for-macrovascular-disease-coronary-cerebrovascular-and-peripheral/
Cardiovascular Outcomes: Function of circulating Endothelial Progenitor Cells (cEPCs): Exploring Pharmaco-therapy targeted at Endogenous Augmentation of cEPCs
Aviva Lev-Ari, PhD, RN 8/28/2012
http://pharmaceuticalintelligence.com/2012/08/28/cardiovascular-outcomes-function-of-circulating-endothelial-progenitor-cells-cepcs-exploring-pharmaco-therapy-targeted-at-endogenous-augmentation-of-cepcs/
Endothelial Dysfunction, Diminished Availability of cEPCs, Increasing CVD Risk for Macrovascular Disease – Therapeutic Potential of cEPCs
Aviva Lev-Ari, PhD, R N 8/27/2012
http://pharmaceuticalintelligence.com/2012/08/27/endothelial-dysfunction-diminished-availability-of-cepcs-increasing-cvd-risk-for-macrovascular-disease-therapeutic-potential-of-cepcs/
Vascular Medicine and Biology: CLASSIFICATION OF FAST ACTING THERAPY FOR PATIENTS AT HIGH RISK FOR MACROVASCULAR EVENTS Macrovascular Disease – Therapeutic Potential of cEPCs
Aviva Lev-Ari, PhD, RN 8/24/2012
http://pharmaceuticalintelligence.com/2012/08/24/vascular-medicine-and-biology-classification-of-fast-acting-therapy-for-patients-at-high-risk-for-macrovascular-events-macrovascular-disease-therapeutic-potential-of-cepcs/
Cardiovascular Disease (CVD) and the Role of agent alternatives in endothelial Nitric Oxide Synthase (eNOS) Activation and Nitric Oxide Production
Aviva Lev-Ari, PhD, RN 7/19/2012
http://pharmaceuticalintelligence.com/2012/07/19/cardiovascular-disease-cvd-and-the-role-of-agent-alternatives-in-endothelial-nitric-oxide-synthase-enos-activation-and-nitric-oxide-production/
Resident-cell-based Therapy in Human Ischaemic Heart Disease: Evolution in the PROMISE of Thymosin beta4 for Cardiac Repair
Aviva Lev-Ari, PhD, RN 4/30/2012
http://pharmaceuticalintelligence.com/2012/04/30/93/
Triple Antihypertensive Combination Therapy Significantly Lowers Blood Pressure in Hard-to-Treat Patients with Hypertension and Diabetes
Aviva Lev-Ari, PhD, RN 5/29/2012
http://pharmaceuticalintelligence.com/2012/05/29/445/
Macrovascular Disease – Therapeutic Potential of cEPCs: Reduction Methods for CV Risk
Aviva Lev-Ari, PhD, RN 7/2/2012
http://pharmaceuticalintelligence.com/2012/07/02/macrovascular-disease-therapeutic-potential-of-cepcs-reduction-methods-for-cv-risk/
Mitochondria Dysfunction and Cardiovascular Disease – Mitochondria: More than just the “powerhouse of the cell”
Aviva Lev-Ari, PhD, RN 7/9/2012
http://pharmaceuticalintelligence.com/2012/07/09/mitochondria-more-than-just-the-powerhouse-of-the-cell/
Bystolic’s generic Nebivolol – positive effect on circulating Endothelial Proginetor Cells endogenous augmentation
Aviva Lev-Ari, PhD, RN 7/16/2012
http://pharmaceuticalintelligence.com/2012/07/16/bystolics-generic-nebivolol-positive-effect-on-circulating-endothilial-progrnetor-cells-endogenous-augmentation/
Arteriogenesis and Cardiac Repair: Two Biomaterials – Injectable Thymosin beta4 and Myocardial Matrix Hydrogel
Aviva Lev-Ari, PhD, RN 2/27/2013
http://pharmaceuticalintelligence.com/2013/02/27/arteriogenesis-and-cardiac-repair-two-biomaterials-injectable-thymosin-beta4-and-myocardial-matrix-hydrogel/
Cardiac Surgery Theatre in China vs. in the US: Cardiac Repair Procedures, Medical Devices in Use, Technology in Hospitals, Surgeons’ Training and Cardiac Disease Severity”
Aviva Lev-Ari, PhD, RN 1/8/2013
http://pharmaceuticalintelligence.com/2013/01/08/cardiac-surgery-theatre-in-china-vs-in-the-us-cardiac-repair-procedures-medical-devices-in-use-technology-in-hospitals-surgeons-training-and-cardiac-disease-severity/
Heart Remodeling by Design – Implantable Synchronized Cardiac Assist Device: Abiomed’s Symphony
Aviva Lev-Ari, PhD, RN 7/23/2012
http://pharmaceuticalintelligence.com/2012/07/23/heart-remodeling-by-design-implantable-synchronized-cardiac-assist-device-abiomeds-symphony/
Acute Chest Pain/ER Admission: Three Emerging Alternatives to Angiography and PCI
Aviva Lev-Ari, PhD, RN 3/10/2013
http://pharmaceuticalintelligence.com/2013/03/10/acute-chest-painer-admission-three-emerging-alternatives-to-angiography-and-pci/
Dilated Cardiomyopathy: Decisions on implantable cardioverter-defibrillators (ICDs) using left ventricular ejection fraction (LVEF) and Midwall Fibrosis: Decisions on Replacement using late gadolinium enhancement cardiovascular MR (LGE-CMR)
Aviva Lev-Ari, PhD, RN 3/10/2013
http://pharmaceuticalintelligence.com/2013/03/10/dilated-cardiomyopathy-decisions-on-implantable-cardioverter-defibrillators-icds-using-left-ventricular-ejection-fraction-lvef-and-midwall-fibrosis-decisions-on-replacement-using-late-gadolinium/
The Heart: Vasculature Protection – A Concept-based Pharmacological Therapy including THYMOSIN
Aviva Lev-Ari, PhD, RN 2/28/2013
http://pharmaceuticalintelligence.com/2013/02/28/the-heart-vasculature-protection-a-concept-based-pharmacological-therapy-including-thymosin/
FDA Pending 510(k) for The Latest Cardiovascular Imaging Technology
Aviva Lev-Ari, PhD, RN 1/28/2013
http://pharmaceuticalintelligence.com/2013/01/28/fda-pending-510k-for-the-latest-cardiovascular-imaging-technology/
PCI Outcomes, Increased Ischemic Risk associated with Elevated Plasma Fibrinogen not Platelet Reactivity
Aviva Lev-Ari, PhD, RN 1/10/2013
http://pharmaceuticalintelligence.com/2013/01/10/pci-outcomes-increased-ischemic-risk-associated-with-elevated-plasma-fibrinogen-not-platelet-reactivity/
The ACUITY-PCI score: Will it Replace Four Established Risk Scores — TIMI, GRACE, SYNTAX, and Clinical SYNTAX
Aviva Lev-Ari, PhD, RN 1/3/2013
http://pharmaceuticalintelligence.com/2013/01/03/the-acuity-pci-score-will-it-replace-four-established-risk-scores-timi-grace-syntax-and-clinical-syntax/
Coronary artery disease in symptomatic patients referred for coronary angiography: Predicted by Serum Protein Profiles
Aviva Lev-Ari, PhD, RN 12/29/2012
http://pharmaceuticalintelligence.com/2012/12/29/coronary-artery-disease-in-symptomatic-patients-referred-for-coronary-angiography-predicted-by-serum-protein-profiles/
Heart Renewal by pre-existing Cardiomyocytes: Source of New Heart Cell Growth Discovered
Aviva Lev-Ari, PhD, RN 12/23/2012
http://pharmaceuticalintelligence.com/2012/12/23/heart-renewal-by-pre-existing-cardiomyocytes-source-of-new-heart-cell-growth-discovered/
Cardiovascular Risk Inflammatory Marker: Risk Assessment for Coronary Heart Disease and Ischemic Stroke – Atherosclerosis.
Aviva Lev-Ari, PhD, RN 10/30/2012
http://pharmaceuticalintelligence.com/2012/10/30/cardiovascular-risk-inflammatory-marker-risk-assessment-for-coronary-heart-disease-and-ischemic-stroke-atherosclerosis/
To Stent or Not? A Critical Decision
Aviva Lev-Ari, PhD, RN 10/23/2012
http://pharmaceuticalintelligence.com/2012/10/23/to-stent-or-not-a-critical-decision/
New Definition of MI Unveiled, Fractional Flow Reserve (FFR)CT for Tagging Ischemia
Aviva Lev-Ari, PhD, RN 8/27/2012
http://pharmaceuticalintelligence.com/2012/08/27/new-definition-of-mi-unveiled-fractional-flow-reserve-ffrct-for-tagging-ischemia/
Ethical Considerations in Studying Drug Safety — The Institute of Medicine Report
Aviva Lev-Ari, PhD, RN 8/23/2012
http://pharmaceuticalintelligence.com/2012/08/23/ethical-considerations-in-studying-drug-safety-the-institute-of-medicine-report/
New Drug-Eluting Stent Works Well in STEMI
Aviva Lev-Ari, PhD, RN 8/22/2012
http://pharmaceuticalintelligence.com/2012/08/22/new-drug-eluting-stent-works-well-in-stemi/
Expected New Trends in Cardiology and Cardiovascular Medical Devices
Aviva Lev-Ari, PhD, RN 8/17/2012
http://pharmaceuticalintelligence.com/2012/08/17/expected-new-trends-in-cardiology-and-cardiovascular-medical-devices/
Coronary Artery Disease – Medical Devices Solutions: From First-In-Man Stent Implantation, via Medical Ethical Dilemmas to Drug Eluting Stents
Aviva Lev-Ari, PhD, RN 8/13/2012
http://pharmaceuticalintelligence.com/2012/08/13/coronary-artery-disease-medical-devices-solutions-from-first-in-man-stent-implantation-via-medical-ethical-dilemmas-to-drug-eluting-stents/
Percutaneous Endocardial Ablation of Scar-Related Ventricular Tachycardia
Aviva Lev-Ari, PhD, RN 7/18/2012
http://pharmaceuticalintelligence.com/2012/07/18/percutaneous-endocardial-ablation-of-scar-related-ventricular-tachycardia/
Competition in the Ecosystem of Medical Devices in Cardiac and Vascular Repair: Heart Valves, Stents, Catheterization Tools and Kits for Open Heart and Minimally Invasive Surgery (MIS)
Aviva Lev-Ari, PhD, RN 6/22/2012
http://pharmaceuticalintelligence.com/2012/06/22/competition-in-the-ecosystem-of-medical-devices-in-cardiac-and-vascular-repair-heart-valves-stents-catheterization-tools-and-kits-for-open-heart-and-minimally-invasive-surgery-mis/
Global Supplier Strategy for Market Penetration & Partnership Options (Niche Suppliers vs. National Leaders) in the Massachusetts Cardiology & Vascular Surgery Tools and Devices Market for Cardiac Operating Rooms and Angioplasty Suites
Aviva Lev-Ari, PhD, RN 6/22/2012
http://pharmaceuticalintelligence.com/2012/06/22/global-supplier-strategy-for-market-penetration-partnership-options-niche-suppliers-vs-national-leaders-in-the-massachusetts-cardiology-vascular-surgery-tools-and-devices-market-for-car/
Related Articles

English: tissue engineered vascular graft Deutsch: tissue engineerte Gefäßprothese (Photo credit: Wikipedia)

English: Cardiovascular disease: PAD therapy with stenting Deutsch: PAVK Therapie: Kathetertherapie mit stenting (Photo credit: Wikipedia)
Endoscopic image of self-expanding metallic stent in esophagus. Photograph released into public domain on permission of patient. — Samir धर्म 07:38, 2 June 2006 (UTC) (Photo credit: Wikipedia)
Like this:
Like Loading...
Read Full Post »





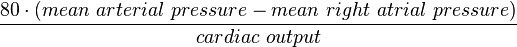
 , where t is time, s is a complex parameter, and A is a complex scalar. Complex values simply mean two dimensional, e.g., magnitude (as in resistance) plus phase shift (to account for reactive components).
, where t is time, s is a complex parameter, and A is a complex scalar. Complex values simply mean two dimensional, e.g., magnitude (as in resistance) plus phase shift (to account for reactive components).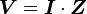 where V and I are the complex scalars in the voltage and current respectively and Z is the complex impedance.
where V and I are the complex scalars in the voltage and current respectively and Z is the complex impedance.



