Recent comprehensive review on the role of ultrasound in breast cancer management
Writer, reporter and curator: Dror Nir, PhD

Word Cloud Created by Noam Steiner Tomer 8/10/2020
The paper below by R Hooley is a beautifully written review on how ultrasound could (and should) be practiced to better support breast cancer screening, staging, and treatment. The authors went as well into the effort of describing the benefits from combining ultrasonography with the other frequently used imaging modalities; i.e. mammography, tomosynthesis and MRI. Post treatment use of ultrasound is not discussed although this is a major task for this modality.
I would like to recommend giving attention to two very small (but for me very important) paragraphs: “Speed of Sound Imaging” and “Lesion Annotation”
Enjoy…
Regina J. Hooley, MD, Leslie M. Scoutt, MD and Liane E. Philpotts, MD
Department of Diagnostic Radiology, Yale University School of Medicine, 333 Cedar St, PO Box 208042, New Haven, CT 06520-8042.
Address correspondence to R.J.H. (e-mail: regina.hooley@yale.edu).
Ultrasonography (US) has become an indispensable tool in breast imaging. Breast US was first introduced in the 1950s by using radar techniques adapted from the U.S. Navy (1). Over the next several decades, US in breast imaging was primarily used to distinguish cystic from solid masses. This was clinically important, as a simple breast cyst is a benign finding that does not require further work-up. However, most solid breast lesions remained indeterminate and required biopsy, as US was not adequately specific in differentiating benign from malignant solid breast masses. However, recent advances in US technology have allowed improved characterization of solid masses.
In 1995, Stavros et al (2) published a landmark study demonstrating that solid breast lesions could be confidently characterized as benign or malignant by using high-resolution grays-cale US imaging. Benign US features include few (two or three) gentle lobulations, ellipsoid shape, and a thin capsule, as well as a homogeneously echogenic echotexture. Malignant US features include spiculation, taller-than-wide orientation, angular margins, microcalcifications, and posterior acoustic shadowing. With these sonographic features, a negative predictive value of 99.5% and a sensitivity of 98.4% for the diagnosis of malignancy were achieved. These results have subsequently been validated by others (3,4) and remain the cornerstone of US characterization of breast lesions today. These features are essential in the comprehensive US assessment of breast lesions, described by the Breast Imaging and Reporting Data System (BI-RADS) (5).
US is both an adjunct and a complement to mammography. Advances in US technology include harmonic imaging, compound imaging, power Doppler, faster frame rates, higher resolution transducers, and, more recently, elastography and three-dimensional (3D) US. Currently accepted clinical indications include evaluation of palpable abnormalities and characterization of masses detected at mammography and magnetic resonance (MR) imaging. US may also be used as an adjuvant breast cancer screening modality in women with dense breast tissue and a negative mammogram. These applications of breast US have broadened the spectrum of sonographic features currently assessed, even allowing detection of noninvasive disease, a huge advance beyond the early simplistic cyst-versus-solid assessment. In addition, US is currently the primary imaging modality recommended to guide interventional breast procedures.
The most subtle US features of breast cancers are likely to be best detected by physicians who routinely synthesize findings from multiple imaging modalities and clinical information, as well as perform targeted US to correlate with lesions detected at mammography or MR imaging. Having a strong understanding of the technical applications of US and image optimization, in addition to strong interpretive and interventional US skills, is essential for today’s breast imager.
Optimal Imaging Technique
US is operator dependent, and meticulous attention to scanning technique as well as knowledge of the various technical options available are imperative for an optimized and accurate breast US examination. US is an interactive, dynamic modality. Although breast US scanning may be performed by a sonographer or mammography technologist, the radiologist also benefits greatly from hands-on scanning (Fig 1). Berg et al (6) demonstrated that US interpretive performance was improved if the radiologist had direct experience performing breast US scanning, including rescanning after the technologist. Real-time scanning also provides the opportunity for thorough evaluation of lesions and permits detailed lesion analysis compared with analyzing static images on a workstation. Subtle irregular or indistinct margins, artifacts, and architectural distortions may be difficult to capture on static images. Real-time scanning also allows the operator to assess lesion mobility, location, and relationship to adjacent structures and allows direct assessment of palpable lesions and other clinical findings. Moreover, careful review of any prior imaging studies is imperative to ensure accurate lesion correlation.


The US examination is generally well tolerated by the patient. Gentle but firm transducer pressure and optimal patient positioning are essential, with the patient’s arm relaxed and flexed behind the head. Medial lesions should generally be scanned in the supine position, and lateral lesions, including the axilla, should usually be scanned with the patient in the contralateral oblique position. This allows for elimination of potential artifact secondary to inadequate compression of breast tissue.
Gray-Scale Imaging
Typical US transducers used in breast imaging today have between 192 and 256 elements along the long axis. When scanning the breast, a linear 12–5-MHz transducer is commonly used. However, in small-breasted women (with breast thickness < 3 cm) or when performing targeted US to evaluate a superficial lesion, a linear 17–5-MHz transducer may be used. Such high-frequency transducers provide superb spatial and soft-tissue resolution, permitting substantially improved differentiation of subtle shades of gray, margin resolution, and lesion conspicuity in the background of normal breast tissue (Fig 2). However, the cost of such a high insonating frequency is decreased penetration due to attenuation of the ultrasound beam, making visualization of deep posterior tissue difficult (ie, greater than 3 cm in depth by using a linear 17–5-MHz transducer or greater than 5 cm in depth by using a linear 12–5-MHz transducer).


During the initial US survey of the region of interest in the breast, the depth should be set so that the pectoralis muscle is visualized along the posterior margin of the field of view. Initial gain settings should be adjusted so that fat at all levels is displayed as a midlevel gray. Simple cysts are anechoic. Compared with breast fat, most solid masses are hypoechoic, while the skin, Cooper ligaments, and fibrous tissue are echogenic. Time gain compensation, which adjusts image brightness at different depths from the skin to compensate for attenuation of the ultrasound beam as it penetrates into the breast tissue, may be set manually or, with appropriate equipment, may be adjusted automatically during real-time scanning or even during postprocessing of the image.
When searching for a lesion initially identified at mammography or MR imaging, careful correlation with lesion depth and surrounding anatomic structures is imperative. Lesion location may be affected by the patient’s position, which differs during mammography, US, and MR imaging examinations. Attention to surrounding background tissue may assist in accurate lesion correlation across multiple modalities. If a mass identified at mammography or MR imaging is surrounded entirely by fat or fibroglandular tissue, at US it should also be surrounded by hypoechoic fat or echogenic fibroglandular tissue, respectively. Similarly, careful attention to the region of clinical concern is necessary when scanning a palpable abnormality to ensure that the correct area is scanned. The examiner should place a finger on the palpable abnormality and then place the transducer directly over the region. Occasionally, the US examination may be performed in the sitting position if a breast mass can only be palpated when the patient is upright.
After a lesion is identified, or while searching for a subtle finding, the depth or field of view may be adjusted as needed. The depth should be decreased to better visualize more superficial structures or increased to better visualize deeper posterior lesions. The use of multiple focal zones also improves resolution at multiple depths simultaneously and should be used, if available. Although this reduces the frame rate, the reduction is typically negligible when scanning relatively superficial structures within the breast. If a single focal zone is selected to better evaluate a single lesion, the focal zone should be centered at the same level as the area of interest or minimally posterior to the area of interest, for optimal visualization.
Spatial Compounding, Speckle Reduction, and Harmonic Imaging
Spatial compound imaging and speckle reduction are available on most high-end US units and should be routinely utilized throughout the breast US examination. Unlike standard US imaging, in which ultrasound pulses are transmitted in a single direction perpendicular to the long axis of the transducer, spatial compounding utilizes electronic beam steering to acquire multiple images obtained from different angles within the plane of imaging (7–9). A single composite image is then obtained in real-time by averaging frames obtained by ultrasound beams acquired from these multiple angles (10). Artifactual echoes, including speckle and other spurious noise, as well as posterior acoustic patterns, including posterior enhancement (characteristic of simple cysts) and posterior acoustic shadowing (characteristic of some solid masses), are substantially reduced. However, returning echoes from real structures are enhanced, providing improved contrast resolution (9) so that ligaments, edge definition, and lesion margins, including spiculations, echogenic halos, posterior and lateral borders, as well as microcalcifications, are better defined. Speckle reduction is a real-time postprocessing technique that also enhances contrast resolution, improves border definition, is complementary to spatial compounding, and can be used simultaneously.
When a lesion is identified, harmonic imaging may also be applied—usually along with spatial compounding—to better characterize a cyst or a subtle solid mass. The simultaneous use of spatial compounding and harmonic imaging may decrease the frame rate, although this usually does not impair real-time evaluation. Harmonic imaging relies on filtering the multiple higher harmonic frequencies, which are multiples of the fundamental frequencies. All tissue is essentially nonlinear to sound propagation and the ultrasound pulse is distorted as it travels through breast tissue, creating harmonic frequencies (9). The returning ultrasound signal therefore contains both the original fundamental frequency and its multiples, or harmonics. Harmonic imaging allows the higher harmonic frequencies to be selected and used to create the gray-scale images (8, 9). Lower-frequency superficial reverberation echoes are thereby reduced, allowing improved characterization of simple cysts (particularly if small) through the elimination of artifactual internal echoes often seen in fluid. Harmonic imaging also improves lateral resolution (10) and may also improve contrast between fatty tissue and subtle lesions, allowing better definition of subtle lesion margins and posterior shadowing (Fig 3).


Speed of Sound Imaging
Conventional US systems set the speed of sound in tissue at a uniform 1540 m/sec (10). However, the speed of sound in tissues of different composition is variable and this variability may compromise US image quality. Breast tissue usually contains fat, and the speed of sound in fat, of approximately 1430–1470 m/sec, is slower than the assumed standard (11). Accurate speed of sound imaging, in which the US transducer may be optimized for the presence of fat within breast tissue, has been shown to improve lateral resolution (12). Additionally, it can be used to better characterize tissue interfaces, lesion margins, and microcalcifications (13) and may also be useful to identify subtle hypoechoic lesions surrounded by fatty breast tissue. Speed of sound imaging is available on most high-end modern US units and is an optional adjustment, depending on whether predominately fatty, predominately dense, or mixed breast tissue is being scanned.
Lesion Annotation
When a mass is identified and the US settings are optimized, the mass should be scanned with US “sweeps” through the entire lesion in multiple planes. Images of the lesion in the radial and antiradial views should be captured and annotated with “right” or “left,” clock face position, and centimeters from the nipple. Radial and anti-radial scanning planes are preferred over standard transverse and sagittal scanning planes because scanning the breast along the normal axis of the mammary ducts and lobar tissues allows improved understanding of the site of lesion origin and better visualization of ductal extension and helps narrow the differential diagnosis (14). Images should be captured with and without calipers to allow margin assessment on static images. Lesion size should be measured in three dimensions, reporting the longest horizontal diameter first, followed by the anteroposterior diameter, then the orthogonal horizontal.
Extended-Field-of-View Imaging
Advanced US technology permits extended-field-of-view imaging beyond the footprint of the transducer. By using a freehand technique, the operator slides the transducer along the desired region to be imaged. The resultant images are stored in real-time and, by applying pattern recognition, a single large-field-of-view image is obtained (7). This can be helpful in measuring very large lesions as well as the distance between multiple structures in the breast and for assessing the relationship of multifocal disease (located in the same quadrant as the index cancer or within 4–5 cm of the index cancer, along the same duct system) and/or multicentric disease (located in a different quadrant than the index cancer, or at a distance greater than 4–5 cm, along a different duct system).
Doppler US
Early studies investigating the use of color, power, and quantitative spectral Doppler US in the breast reported that the presence of increased vascularity, as well as changes in the pulsatility and resistive indexes, showed that these Doppler findings could be used to reliably characterize malignant lesions (15,16). However, other investigators have demonstrated substantial overlap of many of these Doppler characteristics in both benign and malignant breast lesions (17). Gokalp et al (18) also demonstrated that the addition of power Doppler US and spectral analysis to BI-RADS US features of solid breast masses did not improve specificity. While the current BI-RADS US lexicon recommends evaluation of lesion vascularity, it is not considered mandatory (5).
Power Doppler is generally more sensitive than color Doppler to low-flow volumes typical of breast lesions. Light transducer pressure is necessary to prevent occlusion of slow flow owing to compression of the vessel lumen. Currently both power and color Doppler are complementary tools to gray-scale imaging, and power Doppler may improve sensitivity in detecting malignant breast lesions (18,19). Demonstration of irregular branching central or penetrating vascularity within a solid mass raises suspicion of malignant neovascularity (20). Recently, the parallel artery and vein sign has been described as a reliable feature that has the potential to enable prediction of benignity in solid masses so that biopsy may be avoided. In a single study, a paired artery and vein was present in 13.2% of over 1000 masses at US-guided CNB and although an infrequent finding, the specificity for benignity was 99.3% and the false-negative rate was only 1.4%, with two malignancies among 142 masses in which the parallel artery and vein sign was identified (21).
Color and power Doppler US are also useful to evaluate cysts and complex cystic masses that contain a solid component. High-grade invasive cancer and metastatic lymph nodes may occasionally appear anechoic. Demonstration of flow within an otherwise simple appearing cyst, a complicated cyst, or a complex mass confirms the presence of a suspicious solid component, which requires biopsy. In addition, twinkle artifact seen with color Doppler US is useful to identify a biopsy marker clip or subtle echogenic microcalcifications (Fig 4). This Doppler color artifact occurs secondary to the presence of a strong reflecting granular surface and results in a rapidly changing mix of color adjacent to and behind the reflector (22). Care must be taken to avoid mistaking twinkle artifact for true vascular flow and, if in doubt, a spectral Doppler tracing can be obtained, as a normal vascular waveform will not be seen with a twinkle artifact.



Elastography
At physical examination, it has long been recognized that malignant tumors tend to feel hard when compared with benign lesions. US elastography can be used to measure tissue stiffness with the potential to improve specificity in the diagnosis of breast masses. There are two forms of US elastography available today: strain and shear wave. With either technique, acoustic information regarding lesion stiffness is converted into a black-and-white or color-scaled image that can also be superimposed on top of a B-mode gray-scale image.
Strain elastography requires gentle compression with a US probe or natural motion (such as heart beat, vascular pulsation, or respiration) and results in tissue displacement, or strain. Strain (ie, tissue compression and motion) is decreased in hard tissues compared with soft tissue (23). The information obtained with strain elastography provides qualitative information, although strain ratios may be calculated by comparing the strain of a lesion to the surrounding normal tissue. Benign breast lesions generally have lower ratios in comparison to malignant lesions (24,25).
Shear-wave elastography is based on the principle of acoustic radiation force. With use of light transducer pressure, transient automatic pulses can be generated by the US probe, inducing transversely oriented shear waves in tissue. The US system captures the velocity of these shear waves, which travel faster in hard tissue compared with soft tissue (26). Shear-wave elastography provides quantitative information because the elasticity of the tissue can be measured in meters per second or in kilopascals, a unit of pressure.
Elastography features such as strain ratios, size ratios, shape, homogeneity, and maximum lesion stiffness may complement conventional US in the analysis of breast lesions. Malignant masses evaluated with elastography tend to be more irregular, heterogeneous, and typically appear larger at elastography than at grayscale imaging (Fig 5) (27,28). Although malignant lesions generally also exhibit maximum stiffness greater than 80–100 kPa (28,29), caution is necessary when applying these numerical values to lesion analysis. Berg et al (28) reported three cancers among 115 masses with maximum stiffness between 20 and 30 kPa, for a 2.6% malignancy rate; 25 cancers among 281 masses with maximum stiffness between 30 and 80 kPa, for an 8.9% malignancy rate; and 61 cancers among 153 masses with maximum stiffness between 80 and 160 kPa, for a 39.9% malignancy rate (28). Invasive cancers with high histologic grade, large tumor size, nodal involvement, and vascular invasion have also been shown to be significantly correlated with high mean stiffness at shear-wave elastography (30).

Elastography may be useful in improving the specificity of US evaluation of BI-RADS 3 and 4A lesions, including complicated cysts. Berg and colleagues (28) showed that by using qualitative shear-wave elastography and color assessment of lesion stiffness, oval shape, and a maximum elasticity value of less than 80 kPa, unnecessary biopsy of low-suspicion BI-RADS 4A masses could be reduced without a significant loss in sensitivity. Several investigators have proposed a variety of imaging classifications using strain elastography, mostly based on the color pattern (27,31,32). A “bull’s eye” artifact has also been described as a characteristic feature present in benign breast cysts, which may appear as a round or oval lesion with a stiff rim associated with two soft spots, one located centrally and the other posteriorly (33).
Despite these initial promising studies regarding the role of US elastography in the analysis of breast lesions, limitations do exist. Strain and shear-wave elastography are quite different methods of measuring breast tissue stiffness, and the application of these methods varies across different commercial manufacturers. Inter- and intraobserver variability may be relatively high because the elastogram may be affected by differences in degree and method of compression. With strain elastography, a quality indicator that is an associated color bar or numerical value may be helpful to ensure proper light compression. Shear-wave elastography has been shown to be less operator-dependent, as tissue compression is initiated by the US probe in a standard, reproducible fashion (34) and only light transducer pressure is necessary. In addition, there is currently no universal color-coding standard and, depending on the manufacturer and/or operator preference, stiff lesions may be arbitrarily coded to appear red while soft lesions appear blue, or vice versa. Some elastography features such as the “bull’s eye” artifact are only seen on specific US systems. Lesions deeper than 2 cm are less accurately characterized by means of elastography. Moreover, one must be aware that soft cancers and hard benign lesions exist. Therefore, careful correlation of elastography with B-mode US features and mammography is essential. Future studies and further technical advances, including the creation of more uniformity across different US manufacturers, will ultimately determine the usefulness of elastography in clinical practice.
Three-dimensional US
Both handheld and automated high-resolution linear 3D transducers are now available for use in breast imaging. With a single pass of the ultrasound beam, a 3D reconstructed image can be formed in the coronal, sagittal, and transverse planes, potentially allowing more accurate assessment of anatomic structures and tumor margins (Fig 6). Few studies regarding the performance of 3D US in the breast exist, but a preliminary study demonstrated improved characterization of malignant lesions (35). Automated supine whole-breast US using 3D technology is now widely available for use in the screening setting (see section on screening breast US). Three-dimensional US may also be used in addition to computed tomography for image-guided radiation therapy (36) and has a potential role in assessing tumor response to neoadjuvant chemotherapy.

US Features of Benign and Malignant Breast Lesions
Cysts
Although for many years the main function of breast US was to differentiate cysts from solid masses, this differentiation can at times be problematic, particularly if the lesion is small or located deep in the breast. Simple cysts are defined as circumscribed, anechoic masses with a thin imperceptible wall and enhanced through transmission (provided spatial compounding is not used). By convention, simple cysts may also contain up to a single thin septation. Simple cysts are confidently characterized with virtually 100% accuracy at US (14,37), provided that they are not very small (< 5 mm in size) or not located in deep tissue. Complicated cysts are hypoechoic with no discernable Doppler flow, contain internal echoes, and may also exhibit indistinct margins, and/or lack posterior acoustic enhancement. Clustered microcysts consist of a cluster of tiny (<2–3 mm in size) anechoic foci with thin (< 0.5 mm in thickness) intervening septations.
Complicated cysts are very common sonographic findings and the majority are benign. In multiple studies, which evaluated over 1400 complicated cysts and microcysts, the malignancy rate ranged from 0% to 0.8% (38–44). Most complicated cysts and clustered microcysts with a palpable or mammographic correlate are classified as BI-RADS 3 and require short-interval imaging follow-up or, occasionally, US-guided aspiration. However, in the screening US setting, if multiple and bilateral complicated and simple cysts are present (ie, at least three cysts with at least one cyst in each breast), these complicated cysts can be assessed as benign, BI-RADS 2, requiring no additional follow-up (38).
Complicated cysts should never demonstrate internal vascularity at color Doppler interrogation. The presence of a solid component, mural nodule, thickened septation, or thickened wall within a cystic mass precludes the diagnosis of a benign complicated cyst. These complex masses require biopsy, as some cancers may have cystic components. The application of compound imaging and harmonics, color Doppler, and potentially elastography may help differentiate benign complicated cysts from malignant cystic-appearing masses and reduce the need for additional follow-up or biopsy.
Solid Masses
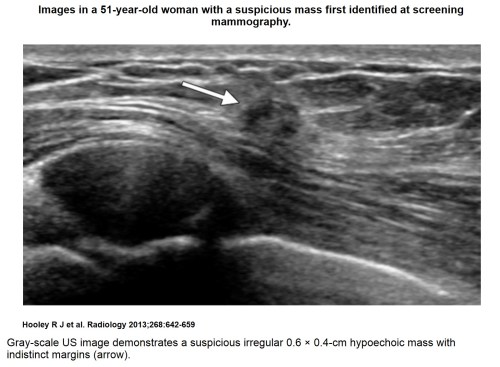
Sonographic features of benign-appearing solid masses include an oval or ellipsoid shape, “wider-than-tall” orientation parallel to the skin, circumscribed margins, gentle and smooth (less than three) lobulations, as well as absence of any malignant features (2,45) (Fig 2b). Lesions with these features are commonly fibroadenomas or other benign masses and can often be safely followed, even if the mass is palpable (46–48). Malignant features of solid masses include spiculations, angular margins, marked hypoechogenicity, posterior acoustic shadowing, microcalcifications, ductal extension, branching pattern, and 1–2-mm microlobulations (2,45) (Figs 1b,5, 6). These are also often taller-than-wide lesions with a nonparallel orientation to the skin and may occasionally be associated with thickened Cooper ligaments and/or or skin thickening. Most cancers have more than one malignant feature, spiculation being the most specific and angular margins the most common (2).
There is, however, considerable overlap between these benign and malignant US features and careful scanning technique, as well as direct correlation with mammography, is essential. For example, some high-grade invasive ductal carcinomas with central necrosis, as well as the well-differentiated mucinous and medullary subtypes, may present as circumscribed, oval, hypoechoic masses that may look like complicated cysts with low-level internal echoes at US. Benign focal fibrous breast tissue or postoperative scars can appear as irregular shadowing masses on US images. Furthermore, while echogenic lesions are often benign and frequently represent lipomas or fibrous tissue, echogenic cancers do rarely occur (Figs 7, 8) (49,50). The presence of a single malignant feature, despite the presence of multiple benign features, precludes a benign classification and mandates biopsy, with the exception of fat necrosis and postoperative scars exhibiting typical benign mammographic features. Likewise, a mass with a benign US appearance should be biopsied if it exhibits any suspicious mammographic features.


Ductal Carcinoma in Situ
Ductal carcinoma in situ (DCIS) is characteristically associated with microcalcifications detected at mammography, but may also be detected at US since they are often associated with a subtle hypoechoic mass, which may indicate an invasive mammographically occult component. US features associated with DCIS most commonly include a hypoechoic mass with an irregular shape, microlobulated margins, no posterior acoustic features, and no internal vascularity. Ductal abnormalities, intracystic lesions, and architectural distortions may also be present (51–53). Noncalcified DCIS manifesting as a solid mass at US is more frequently found in non–high-grade than high-grade DCIS, which is more often associated with microcalcifications and ductal changes (54). US can depict microcalcifications, particularly those in clusters greater than 10 mm in size and located in a hypoechoic mass or a ductlike structure (Fig 9) (55). Malignant calcifications are more likely to be detected sonographically than are benign calcifications, which may be obscured by surrounding echogenic breast tissue (55,56). Although US is inferior to mammography in the detection of suspicious microcalcifications, the main benefit of US detection of DCIS is to identify the invasive component and guide biopsy procedures.


Breast US in Clinical Practice
Current indications for breast US as recommended by the American College of Radiology Practice Guidelines include the evaluation of palpable abnormalities or other breast symptoms, assessment of mammographic or MR imaging–detected abnormalities, and evaluation of breast implants (57). Additionally, US is routinely used for guidance during interventional procedures, treatment planning for radiation therapy, screening in certain groups of women, and evaluation of axillary lymph nodes. Much literature has been written on these uses and a comprehensive discussion is beyond the scope of this article. A few important and timely topics, however, will be reviewed.
BI-RADS US
The BI-RADS US lexicon was introduced in 2003, and subsequently, there have been several studies assessing the accuracy of BI-RADS US classification of breast lesions. Low to moderate interobserver agreement has been found in the description of margins (especially noncircumscribed margins), echogenicity, and posterior acoustic features. Abdullah et al (58) reported low interobserver agreement especially for small masses and for malignant masses. Given the importance of margin analysis in the characterization of benign and malignant lesions, this variability is potentially problematic. Studies have also shown variable results in the use of the final assessment categories. In clinical settings, Raza et al (46) showed inconsistent use of the BI-RADS 3 (probably benign) category in 14.0% of cases when biopsy was recommended. Abdullah et al also demonstrated fair and poor interobserver agreement for BI-RADS 4 (suspicious for malignancy) a, b, and c subcategories (58). However, Henig et al (59) reported more promising results, with malignancy rates in categories 3, 4, and 5 to be similar to those seen with mammographic categorization (1.2%, 17%, and 94%, respectively).
Evaluation of Mammographic Findings
Targeted US is complementary to diagnostic mammography because of its ability to differentiate cystic and solid lesions.US is also useful in the work up of subtle asymmetries, as it can help identify or exclude the presence of an underlying mass. True hypoechoic lesions can often be differentiated from prominent fat lobules by scanning in multiple planes, because true lesions usually do not blend or elongate into adjacent tissue. With the introduction of digital breast tomosynthesis for mammographic imaging, US will play yet another important role. As mammographic lesions can often be detected, localized, and have adequate margin assessment on 3D images, patients with lesions detected on digital breast tomosynthesis images at screening may often be referred directly to US, avoiding additional mammographic imaging and its associated costs and radiation exposure (Fig 10). This will place an even greater importance on high-quality US.



Evaluation of the Symptomatic Patient:Palpable Masses, Breast Pain, and Nipple Discharge
US is essential in the evaluation of patients with the common clinical complaint of either a palpable mass or focal persistent breast pain. Unlike focal breast pain, which may be occasionally associated with benign or malignant lesions, diffuse breast pain (bilateral or unilateral), as well as cyclic breast pain, requires only clinical follow-up, as it is usually physiologic with an extremely low likelihood of malignancy (60,61). In patients with isolated focal breast pain, the role of sonography may be limited to patient reassurance (61). In women younger than 30 years of age, with a palpable lump or focal breast pain, US is the primary imaging test, with a sensitivity and negative predictive value of nearly 100% (62). Symptomatic women older than 30 years usually require both US and mammography, and in these patients, the negative predictive value approaches 100% (63,64). Lehman et al (65) demonstrated that in symptomatic women aged 30–39 years, the risk of malignancy was 1.9% and the added value of adjunct mammography in addition to US was low. Identification of a benign-appearing solid lesion at US in a symptomatic woman can negate the need for needle biopsy, as many of these masses can safely be monitored with short-interval follow-up US (46–48), usually performed at 6 months. A suspicious mass identified at US can promptly undergo biopsy with US guidance.
US can also be used as an alternative or an addition to ductography in patients who present with unilateral, spontaneous bloody, clear, or serosanguinous nipple discharge (66). Among women with worrisome nipple discharge, ductography can demonstrate an abnormality in 59%–82% of women (67,68), MR imaging may demonstrate a suspicious abnormality in 34% of women (68), and US has been shown to demonstrate a subareolar mass or an intraductal mass or filling defect in up to 14% of women (67). If US can be used to identify a retroareolar mass or an intraductal mass, US-guided biopsy can be performed and ductography may be avoided (Fig 11). US may be limited, however, as small peripherally located intraductal masses or masses without an associated dilated duct may not be identified. Therefore, galactography, MR imaging, and/or major duct excision may still be necessary in the symptomatic patient with a negative US examination.

Finally, in the pregnant or lactating patient who presents with a palpable breast mass, focal breast pain, or bloody nipple discharge, US is also the initial imaging modality of choice. Targeted US examination in these patients can be used to identify most benign and malignant masses, including fibroadenomas, galactocoeles, lactating adenomas, abscesses, and invasive carcinomas. In a recent study by Robbins et al (69), a negative predictive value of 100% was found among 122 lesions evaluated with US in lactating, pregnant, or postpartum women. This is much higher than the pregnancy-associated breast cancer sensitivity of mammography, which has been reported in the range of 78%–87% (70,71). The diminished sensitivity of mammography is likely due to increased parenchymal density seen in these patients. However, since lactating breast parenchyma is more echogenic than most breast masses, hypoechoic breast cancers are more readily detected at US in pregnant patients.
Supplemental Screening Breast US
Because of the known limitations of mammography, particularly in women with dense breast tissue, supplemental screening with whole-breast US, in addition to mammography, is increasingly gaining widespread acceptance. Numerous independent studies have demonstrated that the addition of a single screening or whole-breast US examination in women with dense breast tissue at mammography will yield an additional 2.3–4.6 mammographically occult cancers per 1000 women (72–80). Mammographically occult cancers detected on US images are generally small node-negative invasive cancers (Fig 12) (81). However, few studies have investigated the performance of incident screening breast US, and the optimal screening US interval is unknown. Berg and colleagues (82) recently demonstrated that incident annual supplemental screening US in intermediate- and high-risk women with mammographically dense breast tissue enabled detection of an additional 3.7 cancers per 1000 women screened.

Handheld screening breast US is highly operator-dependent and the majority of screening breast US studies have relied on physician-performed examinations. As per the ACRIN 6666 protocol, a normal screening US examination should consist of a minimum of one image in each quadrant and one behind the nipple (83). Two studies have also demonstrated that technologist-performed handheld screening breast US can achieve similar cancer detection rates (76,78).
Automated whole-breast US is a recently developed alternative to traditional handheld screening breast US, in which standardized, uniform image sets may be readily obtained by a nonradiologist. Automated whole-breast US systems may utilize a standard US unit and a linear-array transducer attached to a computer-guided mechanical arm or a dedicated screening US unit with a 15-cm wide transducer (84,85). With these systems, over 3000 overlapping sagittal, transverse, and coronal images are obtained and available for later review by the radiologist, with associated 3D reconstruction. The advantages include less operator dependence, increased radiologist efficiency, and increased reproducibility, which could aid in follow-up of lesions.
A multi-institutional study has shown that supplemental automated whole-breast US can depict an additional 3.6 cancer per 1000 women screened, similar to physician-performed handheld screening US (85). However, disadvantages include the limited ability to scan the entire breast, particularly posterior regions in large breasts, time-consuming review of a large number of images by the radiologist, and the need to recall patients for a second US examination to re-evaluate indeterminate findings. Moreover, few investigators have compared the use of handheld with automated breast US screening. A single small recent study by Chang et al (86) demonstrated that of 14 cancers initially detected at handheld screening, only 57%–79% were also detected by three separate readers on automated whole-breast US images, with the two cancers missed by all three readers at automated whole-breast US, each less than 1 cm in size.
The use of supplemental screening breast US, performed in addition to mammography, remains controversial despite proof of the ability to detect small mammographically occult cancers. US has limited value for the detection of small clustered microcalcifications without an associated mass lesion. Low positive predictive values of biopsies performed of less than 12% have been consistently reported (77,87). No outcome study has been able to demonstrate a direct decrease in patient mortality due to the detection of these additional small and mammographically occult cancers. This would require a long, randomized screening trial, which is not feasible. Rationally, however, the early detection and treatment of additional small breast cancers should improve outcomes and reduce overall morbidity and mortality. Many insurance companies will not reimburse for screening breast US and historically, this examination has not been widely accepted in the United States.
Nevertheless, because of both the known efficacy of supplemental screening breast US and overall increased breast cancer awareness, more patients and clinicians are requesting this examination. In fact, some states now mandate that radiologists inform women of their breast density and advise them to discuss supplemental screening with their doctors. Although supplemental screening breast MR imaging is usually preferred for women who are at very high risk for breast cancer (ie, women with a lifetime risk of over 20%, for example those women who are BRCA positive or have multiple first-degree relatives with a history of premenopausal breast cancer), screening breast US should be considered in women at very high risk for breast cancer who cannot tolerate breast MR imaging, as well as those women with dense breast tissue and intermediate risk (ie, lifetime risk of 15%–20%, for example those women whose only risk factor is a personal history of breast cancer or previous biopsy of a high-risk lesion), or even average risk. Future studies are needed to establish strategies to reduce false-positive results and continue to optimize both technologist-performed handheld screening US and automated whole-breast US in women with mammographically dense breast tissue.
Use of US for MR Imaging–depicted Abnormalities
MR imaging of the breast is now an integral part of breast imaging, most commonly performed to screen high-risk women and to further assess the stage in patients with newly diagnosed breast cancers. While MR has a higher sensitivity than mammography for detecting breast cancer, the specificity is relatively low (88). Lesions detected on MR images are often mammographically occult, but many can be detected with targeted US (Fig 13). Besides further US characterization of an MR imaging–detected lesion, US may be used to guide intervention for lesions initially detected at MR imaging. US-guided biopsies are considerably less expensive, less time consuming, and more comfortable for the patient than MR imaging–guided biopsies.


Some suspicious lesions detected at MR imaging will represent invasive ductal or lobular cancers, but many may prove to be intraductal disease, which can be challenging to detect at US. Meticulous scanning technique is required for an MR imaging–directed US examination, with knowledge of subtle sonographic signs and close correlation with the MR imaging findings and location. Precontrast T1 images are helpful to facilitate localization of lesions in relation to fibroglandular tissue (89). Because MR imaging abnormalities tend to be vascular, increased vascularity may also assist in detection of a subtle sonographic correlate (90). Having the MR images available for simultaneous review while performing the US examination will ideally permit such associative correlation. At the authors’ facility, computer monitors displaying images from the picture archiving and communication system are available in all US rooms for this purpose.
Recent studies have shown that 46%–71% of lesions at MR imaging can be detected with focused US (90–94). Enhancing masses detected on MR images are identified on focused US images in 58%–65% of cases compared with nonmass enhancement, which is identified on focused US images in only 12%–32% of cases (90–92). Some studies have shown that US depiction of an MR imaging correlate was independent of size (91,93,95). However, Meissnitzer et al (92) showed that size dependence is also important: For masses 5 mm or smaller, only 50% were seen, versus 56% for masses 6–10 mm, 73% for masses 11–15 mm, and 86% for masses larger than 15 mm. Likewise, this study also demonstrated that for nonmass lesions, a US correlate was found for 13% of those measuring 6–10 mm, 25% of those 11–15 mm, and 42% of those larger than 15 mm (92). In addition, many of these studies determined that when a sonographic correlate was discovered, the probability of malignancy was increased (90–92). Since typical US malignant features such as spiculation and posterior shadowing may be absent and the pretest probability is higher for MR imaging–detected lesions, a lower threshold for biopsy should be considered when performing MR imaging–directed US compared with routine targeted US (90) or screening US.
Because lesions are often very subtle at MR-directed US examination and because of differences in patient positioning during the two examinations, careful imaging–histologic correlation is required when performing US-guided biopsy of MR imaging–detected abnormalities. For lesions sampled with a vacuum-assisted device and US guidance, Sakamoto et al (96) found a higher rate of false-negative biopsy results for MR imaging–detected lesions than for US-detected lesions, suggesting that precise US-MR imaging correlation may not have occurred. Meissnitzer et al (92) showed that although 91% of MR imaging–detected lesions had an accurate US correlate, 9% were found to be inaccurate. With ever-improving techniques and experience in breast US, the US visualization of MR imaging–detected abnormalities will likely continue to improve. Nevertheless, if a suspicious lesion is not identified sonographically, MR imaging–guided biopsy should still be performed, because the malignancy rate of sonographically occult MR imaging–detected lesions has been shown to range from 14% to 22% (91,95).
Preoperative Staging of Cancer with US
Breast MR imaging has been shown to be more sensitive than US in the detection of additional foci of mammographically occult disease in women with newly diagnosed breast cancer (97–99). Nevertheless, when a highly suspicious mass is identified at mammography and US, immediate US evaluation of the remainder of the ipsilateral breast, the contralateral breast, and the axilla should be considered. If additional lesions are identified, preoperative staging with MR imaging can be avoided and US-guided biopsy can be promptly performed, saving the patient valuable time and expense (100). In a study by Moon et al (101), of 201 patients with newly diagnosed breast cancer, staging US demonstrated mammographically occult multifocal or multicentric disease in 28 patients (14%) and contralateral breast cancers in eight patients (4%), resulting in a change in therapy in 32 patients (16%).
US can also be used to identify abnormal axillary, supraclavicular, and internal mammary lymph nodes. Abnormal lymph nodes characteristically demonstrate focal or diffuse cortical thickening (≥3 mm in thickness), a round (rather than oval or reniform) shape, loss of the echogenic fatty hilum and/or nonhilar, disorganized, irregular blood vessels (102,103) (Fig 14). A positive US-guided CNB or fine-needle aspiration of a clinically abnormal axillary lymph node in a patient with a known breast cancer can aid patient management, by avoiding the need for sentinel node biopsy and allowing the patient instead to proceed directly to axillary lymph node dissection or neoadjuvant chemotherapy.

Interventional Breast US
US-guided interventional procedures have increased in volume in recent years and US is now the primary biopsy guidance technique used in many breast imaging centers. Most palpable lesions, as well as lesions detected at mammography, MR imaging, or screening US, can be sampled with US. With current high-resolution transducers, even suspicious intraductal microcalcifications may be detected and sampled.
While US-guided procedures require technical skills that must be developed and can be challenging, once mastered this technique allows precise real-time sampling of the lesion, which is not possible with either stereotactic or MR imaging–guided procedures. US-guided procedures do not require ionizing radiation or intravenous contrast material. US procedures are more tolerable for patients than stereotactic (104) or MR imaging–guided procedures because US-guided procedures are faster and more comfortable, as breast compression and uncomfortable biopsy coils or tables are not necessary and the procedure may be performed with the patient supine (104–106).
Most literature has shown that automated 14-gauge CNB devices are adequate for the majority of US-guided biopsies (107–115). Image-guided CNB is preferable to fine-needle aspiration cytology of breast masses because of superior sensitivity, specificity, and diagnostic accuracy (116). DCIS, malignant invasion, and hormone receptor status of invasive breast cancers can be determined with CNB samples, but not with fine-needle aspiration cytology. Fine-needle aspiration may be performed, however, in complicated cysts and symptomatic simple cysts. In these cases, the cyst aspirate fluid can often be discarded; cytology is usually only necessary if the fluid is frankly bloody (117).
The choice of performing fine-needle aspiration or CNB of a suspicious axillary lymph node depends on radiologist preference and the availability of an experienced cytopathologist, although CNB is usually more accurate than fine-needle aspiration biopsy (118,119). Fine-needle aspiration may be preferred for suspicious deep lymph nodes in proximity to the axillary vessels, whereas CNB may be preferred in large nodes with thickened cortices, particularly if determination of hormone receptor status or immunohistochemistry is desired, since more tissue is required for these assays. If lymphoma is suspected, a core should be placed in saline and also in conventional formalin.
While the underestimation rate of malignancy can be considerable for high-risk lesions such as atypical hyperplasia, such histology is not commonly found in lesions undergoing US-guided CNB. Multiple studies have shown a false-negative rate for US CNB biopsy of around 2%–3% (107–115). Although the contiguous and larger samples obtained with a vacuum-assisted biopsy device undoubtedly reduce sampling error, the vacuum-assisted biopsy is a more expensive and more invasive procedure (109). In the authors’ experience, vacuum-assisted US biopsy is to be considered for small masses, intraductal or intracystic lesions, or lesions with subtle microcalcifications. These may be difficult to adequately sample with a spring-loaded automatic firing device. Alternatively, for more accurate sampling of such challenging cases, as well as some axillary lymph nodes and masses smaller than 1 cm in size, automated CNB needles designed to place the inner trough of the needle within a lesion before firing can be utilized (Fig 15). With this technique, the sampling trough of the CNB needle can be clearly visualized within the lesion before the overlying outer sheath is fired. Regardless of needle choice, a postbiopsy clip marker should be placed followed by a postbiopsy mammogram to document clip position. This will assist with follow-up imaging, facilitating mammography and/or MR imaging correlation.


There has been recent interest in the percutaneous removal of benign breast lesions by using US-guided vacuum-assisted biopsy. While in general, proved benign concordant lesions can safely remain in the breast, some patients desire removal. Percutaneous US-guided removal with a vacuum-assisted biopsy device can replace surgical removal in some cases, particularly for small lesions (1 cm in size or less). Several reports have shown promising results demonstrating rates of complete lesion excision, varying from 61% to 94% (120–124). Dennis et al (125) demonstrated that vacuum-assisted US-guided biopsy could be used to excise intraductal lesions resulting in resolution of problematic nipple discharge in 97% of patients. Even on long-term follow-up, most studies show low rates of residual masses, more commonly observed in larger fibroadenomas.
Intraoperative Breast US
The use of two-dimensional and 3D intraoperative US may decrease the incidence of positive margins and decrease re-excision rates (126–130) particularly in the setting of lumpectomy for palpable cancers, when US is used to assess the adequacy of surgical margins to determine the need for additional tissue removal. Similarly, intraoperative US has also been utilized to improve detection and removal of metastatic lymph nodes during sentinel lymph node assessment (131).
Intravenous US microbubble contrast agents have been used to enhance US diagnosis by means of analysis, enhancement patterns, the rates of uptake and washout, and identification of tumor angiogenesis. In addition, preliminary research has shown that intravenous US contrast agents may be able to depict tissue function with the potential to deliver targeted gene therapy to selected tumor cells (132). However, there are currently no intravenous US contrast agents approved for use in breast imaging by the U.S. Food and Drug Administration. Other potential advances in breast US include fusion imaging, which involves the direct overlay of correlative MR imaging with targeted US. Another evolving area is that of US computer-aided detection, which may be of particular benefit when combined with automated whole-breast screening US.
Technical advances in US now allow comprehensive US diagnosis, management, and treatment of breast lesions. Optimal use of US technology, meticulous scanning technique with careful attention to lesion morphology, and recognition and synthesis of findings from multiple imaging modalities are essential for optimal patient management. In the future, as radiologists utilize US for an ever-increasing scope of indications, become aware of the more subtle sonographic findings of breast cancer, and apply newly developing tools, the value of breast US will likely continue to increase and evolve.
Essentials
- • Breast US is operator dependent; knowledge and understanding of the various technical options currently available are important for image optimization and accurate diagnosis.
- • US is an interactive, dynamic modality and real-time scanning is necessary to assess subtle findings associated with malignancy.
- • Ability to synthesize the information obtained from the breast US examination with concurrent mammography, MR imaging, and clinical breast examination is necessary for accurate diagnosis.
- • The use of screening breast US in addition to mammography, particularly in women with dense breast tissue, is becoming more widely accepted in the United States.
- • Breast US guidance is the primary biopsy method used in most breast imaging practices, and the radiologist should be familiar with various biopsy devices and techniques to adequately sample any breast mass identified at US.
Disclosures of Conflicts of Interest: R.J.H. No relevant conflicts of interest to disclose. L.M.S. Financial activities related to the present article: none to disclose. Financial activities not related to the present article: educational consultant in vascular US to Philips Healthcare; payment for lectures on breast US from Educational Symposia; payment for development of educational presentations from Philips Healthcare. Other relationships: none to disclose. L.E.P. Financial activities related to the present article: none to disclose. Financial activities not related to the present article: consultant to Hologic. Other relationships: none to disclose.
Abbreviations:
BI-RADS = Breast Imaging and Reporting Data System
CNB = core needle biopsy
DCIS = ductal carcinoma in situ
3D = three dimensional
References
- ↵
- Dempsey PJ
. The history of breast ultrasound. J Ultrasound Med2004;23(7):887–894.
- ↵
- Stavros AT,
- Thickman D,
- Rapp CL,
- Dennis MA,
- Parker SH,
- Sisney GA
. Solid breast nodules: use of sonography to distinguish between benign and malignant lesions. Radiology 1995;196(1):123–134.
- ↵
- Mainiero MB,
- Goldkamp A,
- Lazarus E,
- et al
. Characterization of breast masses with sonography: can biopsy of some solid masses be deferred? J Ultrasound Med 2005;24(2):161–167.
- ↵
- Graf O,
- Helbich TH,
- Hopf G,
- Graf C,
- Sickles EA
. Probably benign breast masses at US: is follow-up an acceptable alternative to biopsy? Radiology2007;244(1):87–93.
- ↵
- Mendelson EB,
- Baum JK,
- Berg WA,
- Merritt CR,
- Rubin E
. Breast Imaging Reporting Data System. BI-RADS: Ultrasound. Reston, Va: American College of Radiology, 2003.
- ↵
- Berg WA,
- Blume JD,
- Cormack JB,
- Mendelson EB
. Training the ACRIN 6666 Investigators and effects of feedback on breast ultrasound interpretive performance and agreement in BI-RADS ultrasound feature analysis. AJR Am J Roentgenol 2012;199(1):224–235.
- ↵
- Stafford RJ,
- Whitman GJ
. Ultrasound physics and technology in breast imaging. Ultrasound Clin 2011;6(3):299–312.
- ↵
- Weinstein SP,
- Conant EF,
- Sehgal C
. Technical advances in breast ultrasound imaging. Semin Ultrasound CT MR 2006;27(4):273–283.
- ↵
- Athanasiou A,
- Tardivon A,
- Ollivier L,
- Thibault F,
- El Khoury C,
- Neuenschwander S
. How to optimize breast ultrasound. Eur J Radiol2009;69(1):6–13.
- ↵
- Kremkau FW
. Sonography principles and instruments. 8th ed. St Louis, Mo: Elsevier-Saunders, 2011.
- ↵
- Goss SA,
- Johnston RL,
- Dunn F
. Comprehensive compilation of empirical ultrasonic properties of mammalian tissues. J Acoust Soc Am1978;64(2):423–457.
- ↵
- Napolitano D,
- Chou CH,
- McLaughlin G,
- et al
. Sound speed correction in ultrasound imaging. Ultrasonics 2006;44(Suppl 1):e43–e46.
- ↵
- Barr RG,
- Rim A,
- Graham R,
- Berg W,
- Grajo JR
. Speed of sound imaging: improved image quality in breast sonography. Ultrasound Q2009;25(3):141–144.
- ↵
- Stavros AT
. Breast ultrasound. Philadelphia, Pa: Lippincott, Williams & Wilkins, 2004.
- ↵
- Cosgrove DO,
- Kedar RP,
- Bamber JC,
- et al
. Breast diseases: color Doppler US in differential diagnosis. Radiology 1993;189(1):99–104.
- ↵
- Sehgal CM,
- Arger PH,
- Rowling SE,
- Conant EF,
- Reynolds C,
- Patton JA
.Quantitative vascularity of breast masses by Doppler imaging: regional variations and diagnostic implications. J Ultrasound Med 2000;19(7):427–440;quiz 441–442.
- ↵
- Birdwell RL,
- Ikeda DM,
- Jeffrey SS,
- Jeffrey RB Jr.
. Preliminary experience with power Doppler imaging of solid breast masses. AJR Am J Roentgenol1997;169(3):703–707.
- ↵
- Gokalp G,
- Topal U,
- Kizilkaya E
. Power Doppler sonography: anything to add to BI-RADS US in solid breast masses? Eur J Radiol 2009;70(1):77–85.
- ↵
- Tozaki M,
- Fukuma E
. Does power Doppler ultrasonography improve the BI-RADS category assessment and diagnostic accuracy of solid breast lesions?Acta Radiol 2011;52(7):706–710.
- ↵
- Mehta TS,
- Raza S,
- Baum JK
. Use of Doppler ultrasound in the evaluation of breast carcinoma. Semin Ultrasound CT MR 2000;21(4):297–307.
- ↵
- Horvath E,
- Silva C,
- Fasce G,
- et al
. Parallel artery and vein: sign of benign nature of breast masses. AJR Am J Roentgenol 2012;198(1):W76–W82.
- ↵
- Campbell SC,
- Cullinan JA,
- Rubens DJ
. Slow flow or no flow? Color and power Doppler US pitfalls in the abdomen and pelvis. RadioGraphics2004;24(2):497–506.
- ↵
- Schaefer FK,
- Heer I,
- Schaefer PJ,
- et al
. Breast ultrasound elastography: results of 193 breast lesions in a prospective study with histopathologic correlation. Eur J Radiol 2011;77(3):450–456.
- ↵
- Zhao QL,
- Ruan LT,
- Zhang H,
- Yin YM,
- Duan SX
. Diagnosis of solid breast lesions by elastography 5-point score and strain ratio method. Eur J Radiol2012;81(11):3245–3249.
- ↵
- Stachs A,
- Hartmann S,
- Stubert J,
- et al
. Differentiating between malignant and benign breast masses: factors limiting sonoelastographic strain ratio.Ultraschall Med 2013;34(2):131–136.
- ↵
- Bercoff J,
- Tanter M,
- Fink M
. Supersonic shear imaging: a new technique for soft tissue elasticity mapping. IEEE Trans Ultrason Ferroelectr Freq Control2004;51(4):396–409.
- ↵
- Itoh A,
- Ueno E,
- Tohno E,
- et al
. Breast disease: clinical application of US elastography for diagnosis. Radiology 2006;239(2):341–350.
- ↵
- Berg WA,
- Cosgrove DO,
- Doré CJ,
- et al
. Shear-wave elastography improves the specificity of breast US: the BE1 multinational study of 939 masses.Radiology 2012;262(2):435–449.
- ↵
- Athanasiou A,
- Tardivon A,
- Tanter M,
- et al
. Breast lesions: quantitative elastography with supersonic shear imaging—preliminary results. Radiology2010;256(1):297–303.
- ↵
- Evans A,
- Whelehan P,
- Thomson K,
- et al
. Invasive breast cancer: relationship between shear-wave elastographic findings and histologic prognostic factors.Radiology 2012;263(3):673–677.
- ↵
- Fleury Ede F,
- Fleury JC,
- Piato S,
- Roveda D Jr.
. New elastographic classification of breast lesions during and after compression. Diagn Interv Radiol 2009;15(2):96–103.
- ↵
- Tozaki M,
- Fukuma E
. Pattern classification of ShearWave™ Elastography images for differential diagnosis between benign and malignant solid breast masses. Acta Radiol 2011;52(10):1069–1075.
- ↵
- Barr RG,
- Lackey AE
. The utility of the “bull’s-eye” artifact on breast elasticity imaging in reducing breast lesion biopsy rate. Ultrasound Q2011;27(3):151–155.
- ↵
- Cosgrove DO,
- Berg WA,
- Doré CJ,
- et al
. Shear wave elastography for breast masses is highly reproducible. Eur Radiol 2012;22(5):1023–1032.
- ↵
- Kalmantis K,
- Dimitrakakis C,
- Koumpis C,
- et al
. The contribution of three-dimensional power Doppler imaging in the preoperative assessment of breast tumors: a preliminary report. Obstet Gynecol Int 2009;2009:530579.
- ↵
- Chadha M,
- Young A,
- Geraghty C,
- Masino R,
- Harrison L
. Image guidance using 3D-ultrasound (3D-US) for daily positioning of lumpectomy cavity for boost irradiation. Radiat Oncol 2011;6:45.
- ↵
- Hilton SV,
- Leopold GR,
- Olson LK,
- Willson SA
. Real-time breast sonography: application in 300 consecutive patients. AJR Am J Roentgenol1986;147(3):479–486.
- ↵
- Berg WA,
- Sechtin AG,
- Marques H,
- Zhang Z
. Cystic breast masses and the ACRIN 6666 experience. Radiol Clin North Am 2010;48(5):931–987.
-
- Kolb TM,
- Lichy J,
- Newhouse JH
. Occult cancer in women with dense breasts: detection with screening US—diagnostic yield and tumor characteristics.Radiology 1998;207(1):191–199.
-
- Buchberger W,
- DeKoekkoek-Doll P,
- Springer P,
- Obrist P,
- Dünser M
. Incidental findings on sonography of the breast: clinical significance and diagnostic workup. AJR Am J Roentgenol 1999;173(4):921–927.
-
- Berg WA,
- Campassi CI,
- Ioffe OB
. Cystic lesions of the breast: sonographic-pathologic correlation. Radiology 2003;227(1):183–191.
-
- Chang YW,
- Kwon KH,
- Goo DE,
- Choi DL,
- Lee HK,
- Yang SB
. Sonographic differentiation of benign and malignant cystic lesions of the breast. J Ultrasound Med 2007;26(1):47–53.
-
- Daly CP,
- Bailey JE,
- Klein KA,
- Helvie MA
. Complicated breast cysts on sonography: is aspiration necessary to exclude malignancy? Acad Radiol2008;15(5):610–617.
- ↵
- Venta LA,
- Kim JP,
- Pelloski CE,
- Morrow M
. Management of complex breast cysts. AJR Am J Roentgenol 1999;173(5):1331–1336.
- ↵
- Hong AS,
- Rosen EL,
- Soo MS,
- Baker JA
. BI-RADS for sonography: positive and negative predictive values of sonographic features. AJR Am J Roentgenol2005;184(4):1260–1265.
- ↵
- Raza S,
- Chikarmane SA,
- Neilsen SS,
- Zorn LM,
- Birdwell RL
. BI-RADS 3, 4, and 5 lesions: value of US in management—follow-up and outcome. Radiology2008;248(3):773–781.
-
- Harvey JA,
- Nicholson BT,
- Lorusso AP,
- Cohen MA,
- Bovbjerg VE
. Short-term follow-up of palpable breast lesions with benign imaging features: evaluation of 375 lesions in 320 women. AJR Am J Roentgenol 2009;193(6):1723–1730.
- ↵
- Graf O,
- Helbich TH,
- Fuchsjaeger MH,
- et al
. Follow-up of palpable circumscribed noncalcified solid breast masses at mammography and US: can biopsy be averted? Radiology 2004;233(3):850–856.
- ↵
- Linda A,
- Zuiani C,
- Lorenzon M,
- et al
. Hyperechoic lesions of the breast: not always benign. AJR Am J Roentgenol 2011;196(5):1219–1224.
- ↵
- Soon PS,
- Vallentine J,
- Palmer A,
- Magarey CJ,
- Schwartz P,
- Morris DL
.Echogenicity of breast cancer: is it of prognostic value? Breast2004;13(3):194–199.
- ↵
- Moon WK,
- Myung JS,
- Lee YJ,
- Park IA,
- Noh DY,
- Im JG
. US of ductal carcinoma in situ. RadioGraphics 2002;22(2):269–280; discussion 280–281.
-
- Yang WT,
- Tse GM
. Sonographic, mammographic, and histopathologic correlation of symptomatic ductal carcinoma in situ. AJR Am J Roentgenol2004;182(1):101–110.
- ↵
- Izumori A,
- Takebe K,
- Sato A
. Ultrasound findings and histological features of ductal carcinoma in situ detected by ultrasound examination alone. Breast Cancer 2010;17(2):136–141.
- ↵
- Park JS,
- Park YM,
- Kim EK,
- et al
. Sonographic findings of high-grade and non-high-grade ductal carcinoma in situ of the breast. J Ultrasound Med2010;29(12):1687–1697.
- ↵
- Moon WK,
- Im JG,
- Koh YH,
- Noh DY,
- Park IA
. US of mammographically detected clustered microcalcifications. Radiology 2000;217(3):849–854.
- ↵
- Soo MS,
- Baker JA,
- Rosen EL
. Sonographic detection and sonographically guided biopsy of breast microcalcifications. AJR Am J Roentgenol2003;180(4):941–948.
- ↵
- ↵
- Abdullah N,
- Mesurolle B,
- El-Khoury M,
- Kao E
. Breast imaging reporting and data system lexicon for US: interobserver agreement for assessment of breast masses. Radiology 2009;252(3):665–672.
- ↵
- Heinig J,
- Witteler R,
- Schmitz R,
- Kiesel L,
- Steinhard J
. Accuracy of classification of breast ultrasound findings based on criteria used for BI-RADS.Ultrasound Obstet Gynecol 2008;32(4):573–578.
- ↵
- Mansel R
. Management of breast pain. In: Harris JR, Lippman ME, MorrowM, Osborne CK, eds. Diseases of the breast. 4th ed. Philadelphia, Pa:Lippincott Williams & Wilkins, 2010; 52.
- ↵
- Leung JW,
- Kornguth PJ,
- Gotway MB
. Utility of targeted sonography in the evaluation of focal breast pain. J Ultrasound Med 2002;21(5):521–526; quiz 528–529.
- ↵
- Loving VA,
- DeMartini WB,
- Eby PR,
- Gutierrez RL,
- Peacock S,
- Lehman CD
.Targeted ultrasound in women younger than 30 years with focal breast signs or symptoms: outcomes analyses and management implications. AJR Am J Roentgenol 2010;195(6):1472–1477.
- ↵
- Soo MS,
- Rosen EL,
- Baker JA,
- Vo TT,
- Boyd BA
. Negative predictive value of sonography with mammography in patients with palpable breast lesions. AJR Am J Roentgenol 2001;177(5):1167–1170.
- ↵
- Tumyan L,
- Hoyt AC,
- Bassett LW
. Negative predictive value of sonography and mammography in patients with focal breast pain. Breast J2005;11(5):333–337.
- ↵
- Lehman CD,
- Lee CI,
- Loving VA,
- Portillo MS,
- Peacock S,
- DeMartini WB
.Accuracy and value of breast ultrasound for primary imaging evaluation of symptomatic women 30-39 years of age. AJR Am J Roentgenol2012;199(5):1169–1177.
- ↵
- Ballesio L,
- Maggi C,
- Savelli S,
- et al
. Role of breast magnetic resonance imaging (MRI) in patients with unilateral nipple discharge: preliminary study.Radiol Med (Torino) 2008;113(2):249–264.
- ↵
- Sabel MS,
- Helvie MA,
- Breslin T,
- et al
. Is duct excision still necessary for all cases of suspicious nipple discharge? Breast J 2012;18(2):157–162.
- ↵
- Morrogh M,
- Morris EA,
- Liberman L,
- Borgen PI,
- King TA
. The predictive value of ductography and magnetic resonance imaging in the management of nipple discharge. Ann Surg Oncol 2007;14(12):3369–3377.
- ↵
- Robbins J,
- Jeffries D,
- Roubidoux M,
- Helvie M
. Accuracy of diagnostic mammography and breast ultrasound during pregnancy and lactation. AJR Am J Roentgenol 2011;196(3):716–722.
- ↵
- Liberman L,
- Giess CS,
- Dershaw DD,
- Deutch BM,
- Petrek JA
. Imaging of pregnancy-associated breast cancer. Radiology 1994;191(1):245–248.
- ↵
- Ahn BY,
- Kim HH,
- Moon WK,
- et al
. Pregnancy- and lactation-associated breast cancer: mammographic and sonographic findings. J Ultrasound Med2003;22(5):491–497; quiz 498–499.
- ↵
- Kolb TM,
- Lichy J,
- Newhouse JH
. Comparison of the performance of screening mammography, physical examination, and breast US and evaluation of factors that influence them: an analysis of 27,825 patient evaluations. Radiology 2002;225(1):165–175.
-
- Buchberger W,
- Niehoff A,
- Obrist P,
- DeKoekkoek-Doll P,
- Dünser M
. Clinically and mammographically occult breast lesions: detection and classification with high-resolution sonography. Semin Ultrasound CT MR 2000;21(4):325–336.
-
- Crystal P,
- Strano SD,
- Shcharynski S,
- Koretz MJ
. Using sonography to screen women with mammographically dense breasts. AJR Am J Roentgenol2003;181(1):177–182.
-
- Gordon PB,
- Goldenberg SL
. Malignant breast masses detected only by ultrasound: a retrospective review. Cancer 1995;76(4):626–630.
- ↵
- Kaplan SS
. Clinical utility of bilateral whole-breast US in the evaluation of women with dense breast tissue. Radiology 2001;221(3):641–649.
- ↵
- Berg WA,
- Blume JD,
- Cormack JB,
- et al
. Combined screening with ultrasound and mammography vs mammography alone in women at elevated risk of breast cancer. JAMA 2008;299(18):2151–2163.
- ↵
- Hooley RJ,
- Greenberg KL,
- Stackhouse RM,
- Geisel JL,
- Butler RS,
- Philpotts LE
. Screening US in patients with mammographically dense breasts: initial experience with Connecticut Public Act 09-41. Radiology2012;265(1):59–69.
-
- Leconte I,
- Feger C,
- Galant C,
- et al
. Mammography and subsequent whole-breast sonography of nonpalpable breast cancers: the importance of radiologic breast density. AJR Am J Roentgenol 2003;180(6):1675–1679.
- ↵
- Corsetti V,
- Houssami N,
- Ferrari A,
- et al
. Breast screening with ultrasound in women with mammography-negative dense breasts: evidence on incremental cancer detection and false positives, and associated cost. Eur J Cancer2008;44(4):539–544.
- ↵
- Bae MS,
- Han W,
- Koo HR,
- et al
. Characteristics of breast cancers detected by ultrasound screening in women with negative mammograms. Cancer Sci2011;102(10):1862–1867.
- ↵
- Berg WA,
- Zhang Z,
- Lehrer D,
- et al
. Detection of breast cancer with addition of annual screening ultrasound or a single screening MRI to mammography in women with elevated breast cancer risk. JAMA 2012;307(13):1394–1404.
- ↵
- ↵
- Kelly KM,
- Richwald GA
. Automated whole-breast ultrasound: advancing the performance of breast cancer screening. Semin Ultrasound CT MR2011;32(4):273–280.
- ↵
- Kelly KM,
- Dean J,
- Lee SJ,
- Comulada WS
. Breast cancer detection: radiologists’ performance using mammography with and without automated whole-breast ultrasound. Eur Radiol 2010;20(11):2557–2564.
- ↵
- Chang JM,
- Moon WK,
- Cho N,
- Park JS,
- Kim SJ
. Breast cancers initially detected by hand-held ultrasound: detection performance of radiologists using automated breast ultrasound data. Acta Radiol 2011;52(1):8–14.
- ↵
- Berg WA
. Supplemental screening sonography in dense breasts. Radiol Clin North Am 2004;42(5):845–851, vi.
- ↵
- Morrow M,
- Waters J,
- Morris E
. MRI for breast cancer screening, diagnosis, and treatment. Lancet 2011;378(9805):1804–1811.
- ↵
- Hashimoto BE,
- Morgan GN,
- Kramer DJ,
- Lee M
. Systematic approach to difficult problems in breast sonography. Ultrasound Q 2008;24(1):31–38.
- ↵
- Abe H,
- Schmidt RA,
- Shah RN,
- et al
. MR-directed (“second-look”) ultrasound examination for breast lesions detected initially on MRI: MR and sonographic findings. AJR Am J Roentgenol 2010;194(2):370–377.
- ↵
- Demartini WB,
- Eby PR,
- Peacock S,
- Lehman CD
. Utility of targeted sonography for breast lesions that were suspicious on MRI. AJR Am J Roentgenol 2009;192(4):1128–1134.
- ↵
- Meissnitzer M,
- Dershaw DD,
- Lee CH,
- Morris EA
. Targeted ultrasound of the breast in women with abnormal MRI findings for whom biopsy has been recommended. AJR Am J Roentgenol 2009;193(4):1025–1029.
- ↵
- Candelaria R,
- Fornage BD
. Second-look US examination of MR-detected breast lesions. J Clin Ultrasound 2011;39(3):115–121.
- ↵
- Carbognin G,
- Girardi V,
- Calciolari C,
- et al
. Utility of second-look ultrasound in the management of incidental enhancing lesions detected by breast MR imaging. Radiol Med (Torino) 2010;115(8):1234–1245.
- ↵
- LaTrenta LR,
- Menell JH,
- Morris EA,
- Abramson AF,
- Dershaw DD,
- Liberman L
.Breast lesions detected with MR imaging: utility and histopathologic importance of identification with US. Radiology 2003;227(3):856–861.
- ↵
- Sakamoto N,
- Tozaki M,
- Higa K,
- Abe S,
- Ozaki S,
- Fukuma E
. False-negative ultrasound-guided vacuum-assisted biopsy of the breast: difference with US-detected and MRI-detected lesions. Breast Cancer 2010;17(2):110–117.
- ↵
- Hlawatsch A,
- Teifke A,
- Schmidt M,
- Thelen M
. Preoperative assessment of breast cancer: sonography versus MR imaging. AJR Am J Roentgenol2002;179(6):1493–1501.
-
- Zhang Y,
- Fukatsu H,
- Naganawa S,
- et al
. The role of contrast-enhanced MR mammography for determining candidates for breast conservation surgery.Breast Cancer 2002;9(3):231–239.
- ↵
- Berg WA,
- Gutierrez L,
- NessAiver MS,
- et al
. Diagnostic accuracy of mammography, clinical examination, US, and MR imaging in preoperative assessment of breast cancer. Radiology 2004;233(3):830–849.
- ↵
- Yang W
. Staging of breast cancer with ultrasound. Semin Ultrasound CT MR 2011;32(4):331–341.
- ↵
- Moon WK,
- Noh DY,
- Im JG
. Multifocal, multicentric, and contralateral breast cancers: bilateral whole-breast US in the preoperative evaluation of patients.Radiology 2002;224(2):569–576.
- ↵
- Mainiero MB,
- Cinelli CM,
- Koelliker SL,
- Graves TA,
- Chung MA
. Axillary ultrasound and fine-needle aspiration in the preoperative evaluation of the breast cancer patient: an algorithm based on tumor size and lymph node appearance. AJR Am J Roentgenol 2010;195(5):1261–1267.
- ↵
- Bedi DG,
- Krishnamurthy R,
- Krishnamurthy S,
- et al
. Cortical morphologic features of axillary lymph nodes as a predictor of metastasis in breast cancer: in vitro sonographic study. AJR Am J Roentgenol 2008;191(3):646–652.
- ↵
- Mainiero MB,
- Gareen IF,
- Bird CE,
- Smith W,
- Cobb C,
- Schepps B
. Preferential use of sonographically guided biopsy to minimize patient discomfort and procedure time in a percutaneous image-guided breast biopsy program. J Ultrasound Med 2002;21(11):1221–1226.
-
- Philpotts LE
. Percutaneous breast biopsy: emerging techniques and continuing controversies. Semin Roentgenol 2007;42(4):218–227.
- ↵
- Harvey JA,
- Moran RE,
- DeAngelis GA
. Technique and pitfalls of ultrasound-guided core-needle biopsy of the breast. Semin Ultrasound CT MR2000;21(5):362–374.
- ↵
- Parker SH,
- Jobe WE,
- Dennis MA,
- et al
. US-guided automated large-core breast biopsy. Radiology 1993;187(2):507–511.
-
- Liberman L,
- Drotman M,
- Morris EA,
- et al
. Imaging-histologic discordance at percutaneous breast biopsy. Cancer 2000;89(12):2538–2546.
- ↵
- Philpotts LE,
- Hooley RJ,
- Lee CH
. Comparison of automated versus vacuum-assisted biopsy methods for sonographically guided core biopsy of the breast.AJR Am J Roentgenol 2003;180(2):347–351.
-
- Shah VI,
- Raju U,
- Chitale D,
- Deshpande V,
- Gregory N,
- Strand V
. False-negative core needle biopsies of the breast: an analysis of clinical, radiologic, and pathologic findings in 27 concecutive cases of missed breast cancer. Cancer2003;97(8):1824–1831.
-
- Crystal P,
- Koretz M,
- Shcharynsky S,
- Makarov V,
- Strano S
. Accuracy of sonographically guided 14-gauge core-needle biopsy: results of 715 consecutive breast biopsies with at least two-year follow-up of benign lesions.J Clin Ultrasound 2005;33(2):47–52.
-
- Dillon MF,
- Hill AD,
- Quinn CM,
- O’Doherty A,
- McDermott EW,
- O’Higgins N
. The accuracy of ultrasound, stereotactic, and clinical core biopsies in the diagnosis of breast cancer, with an analysis of false-negative cases. Ann Surg 2005;242(5):701–707.
-
- Povoski SP,
- Jimenez RE,
- Wang WP
. Ultrasound-guided diagnostic breast biopsy methodology: retrospective comparison of the 8-gauge vacuum-assisted biopsy approach versus the spring-loaded 14-gauge core biopsy approach. World J Surg Oncol 2011;9:87.
-
- Bolívar AV,
- Alonso-Bartolomé P,
- García EO,
- Ayensa FG
. Ultrasound-guided core needle biopsy of non-palpable breast lesions: a prospective analysis in 204 cases. Acta Radiol 2005;46(7):690–695.
- ↵
- Youk JH,
- Kim EK,
- Kim MJ,
- Kwak JY,
- Son EJ
. Analysis of false-negative results after US-guided 14-gauge core needle breast biopsy. Eur Radiol2010;20(4):782–789.
- ↵
- Garg S,
- Mohan H,
- Bal A,
- Attri AK,
- Kochhar S
. A comparative analysis of core needle biopsy and fine-needle aspiration cytology in the evaluation of palpable and mammographically detected suspicious breast lesions. Diagn Cytopathol2007;35(11):681–689.
- ↵
- Ciatto S,
- Cariaggi P,
- Bulgaresi P
. The value of routine cytologic examination of breast cyst fluids. Acta Cytol 1987;31(3):301–304.
- ↵
- Rao R,
- Lilley L,
- Andrews V,
- Radford L,
- Ulissey M
. Axillary staging by percutaneous biopsy: sensitivity of fine-needle aspiration versus core needle biopsy. Ann Surg Oncol 2009;16(5):1170–1175.
- ↵
- Gong JZ,
- Snyder MJ,
- Lagoo AS,
- et al
. Diagnostic impact of core-needle biopsy on fine-needle aspiration of non-Hodgkin lymphoma. Diagn Cytopathol2004;31(1):23–30.
- ↵
- Ko ES,
- Han H,
- Lee BH,
- Choe H
. Sonographic changes after removing all benign breast masses with sonographically guided vacuum-assisted biopsy.Acta Radiol 2009;50(9):968–974.
-
- Slanetz PJ,
- Wu SP,
- Mendel JB
. Percutaneous excision: a viable alternative to manage benign breast lesions. Can Assoc Radiol J 2011;62(4):265–271.
-
- Yom CK,
- Moon BI,
- Choe KJ,
- Choi HY,
- Park YL
. Long-term results after excision of breast mass using a vacuum-assisted biopsy device. ANZ J Surg2009;79(11):794–798.
-
- Kim MJ,
- Park BW,
- Kim SI,
- et al
. Long-term follow-up results for ultrasound-guided vacuum-assisted removal of benign palpable breast mass. Am J Surg2010;199(1):1–7.
- ↵
- Wang ZL,
- Liu G,
- Li JL,
- et al
. Sonographically guided percutaneous excision of clinically benign breast masses. J Clin Ultrasound 2011;39(1):1–5.
- ↵
- Dennis MA,
- Parker S,
- Kaske TI,
- Stavros AT,
- Camp J
. Incidental treatment of nipple discharge caused by benign intraductal papilloma through diagnostic Mammotome biopsy. AJR Am J Roentgenol 2000;174(5):1263–1268.
- ↵
- Bouton ME,
- Wilhelmson KL,
- Komenaka IK
. Intraoperative ultrasound can facilitate the wire guided breast procedure for mammographic abnormalities.Am Surg 2011;77(5):640–646.
-
- Fisher CS,
- Mushawah FA,
- Cyr AE,
- Gao F,
- Margenthaler JA
. Ultrasound-guided lumpectomy for palpable breast cancers. Ann Surg Oncol2011;18(11):3198–3203.
-
- Krekel NM,
- Lopes Cardozo AM,
- Muller S,
- Bergers E,
- Meijer S,
- van den Tol MP
.Optimising surgical accuracy in palpable breast cancer with intra-operative breast ultrasound: feasibility and surgeons’ learning curve. Eur J Surg Oncol2011;37(12):1044–1050.
-
- Olsha O,
- Shemesh D,
- Carmon M,
- et al
. Resection margins in ultrasound-guided breast-conserving surgery. Ann Surg Oncol 2011;18(2):447–452.
- ↵
- DeJean P,
- Brackstone M,
- Fenster A
. An intraoperative 3D ultrasound system for tumor margin determination in breast cancer surgery. Med Phys2010;37(2):564–570.
- ↵
- Hsu GC,
- Ku CH,
- Yu JC,
- Hsieh CB,
- Yu CP,
- Chao TY
. Application of intraoperative ultrasound to nonsentinel node assessment in primary breast cancer. Clin Cancer Res 2006;12(12):3746–3753.
- ↵
- Kiessling F,
- Fokong S,
- Koczera P,
- Lederle W,
- Lammers T
. Ultrasound microbubbles for molecular diagnosis, therapy, and theranostics. J Nucl Med2012;53(3):345–348.
Like this:
Like Loading...
Read Full Post »













































![CT (A) of 78-year-old male who was status post–left lobe lobectomy and left upper lobe wedge resection shows recurrent nodule at the surgical resection site. Fused PET/CT (B) demonstrates increased [18F]FDG uptake in the corresponding nodule at the surgical resection site consistent with recurrent tumor.](https://i0.wp.com/pharmaceuticalintelligence.com/wp-content/uploads/2013/01/fig-10-lung.jpg?resize=317%2C326)
![CT (A) demonstrates new left upper lobe mass representing new primary NSCLC in a patient who had a status post–right pneumonectomy for a prior NSCLC. CT (B) obtained in the same patient 2 weeks after radiofrequency ablation (RFA) demonstrates the postablation density in the left upper lobe. Fused PET/CT (C) obtained 4 months after RFA demonstrates mild [18F]FDG uptake at RFA site in the left upper lobe consistent with posttreatment inflammation. Fused PET/CT (D) obtained 7 months after RFA demonstrates new focal [18F]FDG uptake at post-RFA-opacity consistent with recurrent tumor.](https://i0.wp.com/pharmaceuticalintelligence.com/wp-content/uploads/2013/01/fig-11-lung.jpg?resize=283%2C842)