Posts Tagged ‘Hypertension’
Protected: Flywheel iNO, Three Novel Adult Patient Inhaled Nitric Oxide Product Concepts by Justin D. Pearlman MD ME PhD FACC
Posted in Medical Devices R&D Investment, Medical Imaging Technology, Image Processing/Computing, MRI, CT, Nuclear Medicine, Ultra Sound, Nitric Oxide in Health and Disease, Technology Transfer: Biotech and Pharmaceutical, tagged Heart Failure, Hypertension, nitric oxide, Nitric oxide synthase, Pulmonary hypertension, Torr on June 3, 2013|
Prediction of Occurrence of Cardiovascular Events Independently of Left Ventricular Mass in Hypertensive Patients: Monitoring of Timing of Korotkoff Sounds as Indicator of Arterial Stiffness
Posted in Atherogenic Processes & Pathology, Frontiers in Cardiology and Cardiovascular Disorders, HTN, Origins of Cardiovascular Disease, tagged American Heart Association, Antihypertensive drug, Arterial stiffness, Blood pressure, Cardiovascular disease, Hypertension on May 20, 2013| 1 Comment »
- Original Article
HYPERTENSIONAHA.113.01039 Published online before print May 20, 2013,doi: 10.1161/HYPERTENSIONAHA.113.01039
Arterial Stiffness From Monitoring of Timing of Korotkoff Sounds Predicts the Occurrence of Cardiovascular Events Independently of Left Ventricular Mass in Hypertensive Patients
+Author Affiliations
From the Department of Cardiology and Hypertension, University Hospital of Bordeaux, Bordeaux, France.
- Correspondence to Philippe Gosse, Department of Cardiology and Hypertension, University Hospital of Bordeaux, Hopital Saint Andre, 1 Rue Jean Burguet, 33075 Bordeaux, France. E-mail philippe.gosse@chu-bordeaux.fr
Abstract
Several studies have established that the increase in arterial stiffness (AS) is a cardiovascular risk factor but to date no studies have evaluated in hypertensive patients its prognostic value in comparison with another powerful risk factor, left ventricular mass (LVM) as measured by echocardiography. We prospectively evaluated the prognostic value of AS and LVM in patients with essential hypertension. The population studied comprised 793 patients (56% men) aged 54±14 years. For 519 patients, baseline measurements were made before any antihypertensive treatment, for 274 patients, the measurement were obtained during the follow-up period under antihypertensive treatment. AS was assessed from ambulatory monitoring of blood pressure and timing of Korottkoff sounds. Left ventricular mass was measured in 523 patients. After a mean follow-up of 97 months, 122 cardiovascular events were recorded in the whole population and 74 in the group with LVM determination. AS as continuous or discontinuous variable was independently related to cardiovascular events. The existence or not of antihypertensive treatment at the time of its measurement did not affect its prognostic value. When LVM was forced in the model, AS remained significantly related to cardiovascular events. Thus, AS has an independent prognostic value in the hypertensive, whether measured before or after the administration of antihypertensive treatment. This prognostic value persists after taking LVM into account.
Key Words:
- Received January 10, 2013.
- Revision received March 25, 2013.
- Accepted April 22, 2013.
- © 2013 American Heart Association, Inc.
Walking and Running: Similar Risk Reductions for Hypertension, Hypercholesterolemia, DM, and possibly CAD
Posted in Atherogenic Processes & Pathology, Frontiers in Cardiology and Cardiovascular Disorders, HTN, Origins of Cardiovascular Disease, tagged CHD, Coronary disease, Diabetes Mellitus, Hartford Hospital, Hypercholesterolemia, Hypertension, Lawrence Berkeley National Laboratory on April 4, 2013| 2 Comments »
Reporter: Aviva Lev-Ari, PhD, RN
Walking Versus Running for Hypertension, Cholesterol, and Diabetes Mellitus Risk Reduction
From the Life Sciences Division, Lawrence Berkeley National Laboratory, Berkeley, CA (P.T.W.); and Division of Cardiology, Hartford Hospital, Hartford, CT (P.D.T.).
Correspondence to Paul T. Williams, PhD, Life Sciences Division, Lawrence Berkeley National Laboratory, Donner 464, 1 Cycloton Rd, Berkeley, CA 94720. E-mailptwilliams@lbl.gov
Abstract
Objective—To test whether equivalent energy expenditure by moderate-intensity (eg, walking) and vigorous-intensity exercise (eg, running) provides equivalent health benefits.
Approach and Results—We used the National Runners’ (n=33 060) and Walkers’ (n=15 945) Health Study cohorts to examine the effect of differences in exercise mode and thereby exercise intensity on coronary heart disease (CHD) risk factors. Baseline expenditure (metabolic equivant hours per day [METh/d]) was compared with self-reported, physician-diagnosed incident hypertension, hypercholesterolemia, diabetes mellitus, and CHD during 6.2 years follow-up. Running significantly decreased the risks for incident hypertension by 4.2% (P<10−7), hypercholesterolemia by 4.3% (P<10−14), diabetes mellitus by 12.1% (P<10−5), and CHD by 4.5% per METh/d (P=0.05). The corresponding reductions for walking were 7.2% (P<10−7), 7.0% (P<10−8), 12.3% (P<10−4), and 9.3% (P=0.01). Relative to <1.8 METh/d, the risk reductions for 1.8 to 3.6, 3.6 to 5.4, 5.4 to 7.2, and ≥7.2 METh/d were as follows: (1) 10.0%, 17.7%, 25.1%, and 34.9% from running and 14.0%, 23.8%, 21.8%, and 38.3% from walking for hypercholesterolemia; (2) 19.7%, 19.4%, 26.8%, and 39.8% from running and 14.7%, 19.1%, 23.6%, and 13.3% from walking for hypertension; and (3) 43.5%, 44.1%, 47.7%, and 68.2% from running, and 34.1%, 44.2% and 23.6% from walking for diabetes mellitus (walking >5.4 METh/d excluded for too few cases). The risk reductions were not significantly different for running than walking for diabetes mellitus (P=0.94), hypercholesterolemia (P=0.06), or CHD (P=0.26), and only marginally greater for walking than running for hypercholesterolemia (P=0.04).
Conclusions—Equivalent energy expenditures by moderate (walking) and vigorous (running) exercise produced similar risk reductions for hypertension, hypercholesterolemia, diabetes mellitus, and possibly CHD.
http://atvb.ahajournals.org/content/early/2013/04/04/ATVBAHA.112.300878.abstract.html?papetoc
Secondary Hypertension caused by Aldosterone-producing Adenomas caused by Somatic Mutations in ATP1A1 and ATP2B3
Posted in Biological Networks, Gene Regulation and Evolution, Biomarkers & Medical Diagnostics, CANCER BIOLOGY & Innovations in Cancer Therapy, Cardiovascular Pharmacogenomics, Cell Biology, Signaling & Cell Circuits, Chemical Genetics, Computational Biology/Systems and Bioinformatics, Frontiers in Cardiology and Cardiovascular Disorders, Genome Biology, Genomic Testing: Methodology for Diagnosis, HTN, Molecular Genetics & Pharmaceutical, Personalized and Precision Medicine & Genomic Research, tagged adenoma, Cancer - General, Conditions and Diseases, Hypertension, mutation, Secondary hypertension on February 21, 2013| Leave a Comment »
Reporter: Aviva Lev-Ari, PhD, RN
Somatic mutations in ATP1A1 and ATP2B3 lead to aldosterone-producing adenomas and secondary hypertension
- Felix Beuschlein,
- Sheerazed Boulkroun,
- Andrea Osswald,
- Thomas Wieland,
- Hang N Nielsen,
- Urs D Lichtenauer,
- David Penton,
- Vivien R Schack,
- Laurence Amar,
- Evelyn Fischer,
- Anett Walther,
- Philipp Tauber,
- Thomas Schwarzmayr,
- Susanne Diener,
- Elisabeth Graf,
- Bruno Allolio,
- Benoit Samson-Couterie,
- Arndt Benecke,
- Marcus Quinkler,
- Francesco Fallo,
- Pierre-Francois Plouin,
- Franco Mantero,
- Thomas Meitinger,
- Paolo Mulatero,
- Xavier Jeunemaitre,
- Richard Warth,
- Bente Vilsen,
- Maria-Christina Zennaro,
- Tim M Strom
- & Martin Reincke
- Published online 17 February 2013
- Mutations affecting a pair of related enzyme-coding genes can contribute to the
risk of benign glandular tumors
- called adenomas and secondary hypertension, a new
- study suggests. An international team led by investigators in Germany performed
- on matched tumor and normal samples from nine individuals with forms of adenoma that enhance aldosterone hormone production. This leads to a type of so-called aldosteronism that can bump up blood pressure and cause other adverse symptoms.
When researchers sorted through the exome sequence data, they saw ties between aldosterone-producing adenoma and mutations in two ATPase genes — ATP1A1 and ATP2B3 — that participate in sodium/potassium and calcium signaling, respectively. Somatic ATP1A1 mutations turned up in more than 5 percent of 308 aldosterone-producing adenoma samples screened subsequently, the team noted, while 1.6 percent of those tumors contained ATP2B3 alterations.
“[T]hese findings expand the spectrum of somatic alterations leading to [aldosterone-producing adenomas] to two members of the P-type ATPase pump family, extend knowledge of the molecular mechanism leading to [aldosterone-producing adenoma],” the Ludwig Maximilian University of Munich researcher Martin Reincke, the study’s corresponding author, and colleagues wrote, “and indicate new potential therapeutic targets for the most frequent secondary form of arterial hypertension.”
SOURCE:
http://www.genomeweb.com//node/1194476?hq_e=el&hq_m=1505701&hq_l=6&hq_v=6fcaf1aef4
Somatic mutations in ATP1A1 and ATP2B3 lead to aldosterone-producing adenomas and secondary hypertension
Primary aldosteronism is the most prevalent form of secondary hypertension. To explore molecular mechanisms of autonomous aldosterone secretion, we performed exome sequencing of aldosterone-producing adenomas (APAs). We identified somatic hotspot mutations in the ATP1A1 (encoding an Na+/K+ ATPase α subunit) and ATP2B3 (encoding a Ca2+ ATPase) genes in three and two of the nine APAs, respectively. These ATPases are expressed in adrenal cells and control sodium, potassium and calcium ion homeostasis. Functional in vitro studies of ATP1A1 mutants showed loss of pump activity and strongly reduced affinity for potassium. Electrophysiological ex vivo studies on primary adrenal adenoma cells provided further evidence for inappropriate depolarization of cells with ATPase alterations. In a collection of 308 APAs, we found 16 (5.2%) somatic mutations in ATP1A1 and 5 (1.6%) in ATP2B3.
Mutation-positive cases showed
- male dominance,
- increased plasma aldosterone concentrations and
- lower potassium concentrations compared with mutation-negative cases.
In summary, dominant somatic alterations in two members of the ATPase gene family result in autonomous aldosterone secretion.
Author information
SOURCE:
http://www.nature.com/ng/journal/vaop/ncurrent/full/ng.2550.html
An Important Marker of Hypertension in Young Adults: Plasma Renin
Posted in Biomarkers & Medical Diagnostics, HTN in Youth, tagged Conditions and Diseases, Hypertension, Pheochromocytoma on February 9, 2013| 5 Comments »
An Important Marker of Hypertension in Young Adults: Plasma Renin
Author: Manuela Stoicescu, MD, PhD
Original research
Manuela Stoicescu, MD, PhD
Consultant Internal Medicine, Assistant Professor
Faculty of Medicine and Pharmacy, Medical Disciplines Department
University of Oradea, Romania
ABSTRACT
Introduction: Plasma renin level is an important marker of hypertension in the young adults. The purpose of this study was to determine the role of increased levels of plasmatic renin in the pathogenesis of hypertension in the young adults and to highlight the main conditions underlying the pathogenesis of hypertension in the young people in these circumstances.
Material and methods: The group of patients taking part in the study was of 121 young hypertensive adults (selected from a group of 321 young hypertensive adults), with the age between 18-35 years, with elevated blood pressure exceeding 140/90mmHg in at least three repeated measurements at intervals of one week to exclude white coat phenomenon, or had a blood pressure value greater than 170/100mmHg at the first measurement and increased plasma renin levels above the 4,3ng/ml.
Results and discussion: Of the 121 young hypertensive patients with increased plasma renin levels, 49 were cases of renal artery stenosis representing 40.50% (p<0.001), 8 cases were represented by small unilateral kidneys representing 6.61% (p<0.001), renal cell carcinoma (previously known as “hypernephron” – Grawitz tumor) was responsible for the younger group of patients studied of 4 cases representing 3.30% (p <0.001) of the cases of hypertension in the young adults, and 60 cases representing 49.59% were represented by pheochromocytoma.
Conclusions: The results show the role of plasma renin dosing as being particularly important in the pathogenesis of secondary hypertension in the young adults
Keywords: Renin, arterial hypertension (HBP), young adults
INTRODUCTION
Renin is an enzyme secreted by the juxtaglomerular apparatus to maintain electrolyte balance and blood pressure in the appropriate limits. Plasma renin level is an important marker of hypertension in the young adults. The purpose of this study was to determine the role of increased levels of plasma renin in the pathogenesis of hypertension in the young adults and highlight the main conditions underlying the pathogenesis of hypertension in the young adults with increased plasmatic renin. The principal diseases which had increased plasma renin levels were: renal artery stenosis, pheochromocytoma, congenital unilateral small kidney, primary reninoma (renal cell carcinoma or Grawitz tumor), situations in which renin is secreted in excess, the highest values being in cases of renal cell carcinoma, of 320ng/ml.
MATERIAL AND METHODS
The group of patients taking parts in the study was of 121 hypertensive young adults, with the ages between 18-35 years with elevated blood pressure over 140/90mmHg in at least three repeated measurements at intervals of one week to exclude white coat phenomenon frequently encountered in the young, or had a severely increased blood pressure of >170/100mmHg on the first measurement and plasma renin levels greater than 4,3 ng/ml.
Parameters for assessment of the diseases which had increased plasma rennin levels were clinical, radiological, biological and histopathological. The study was done after the diagnosis of hypertension and staging according OMS. All patients were investigated clinically and fully analyzed paraclinically. They agreed to participate in the trial after they were explained the criteria of professional ethics, scientific and terms of confidentiality. All patients participating in the study had plasmatic renin levels above 4.3ng /ml. The statistical analysis was done with the help of EPIINFO application, version 6.0, program of the Center for Disease Control and Prevention-CDC in Atlanta, suitable for processing of medical statistics. Averages were calculated for the parameters, frequency ranges, standard deviations, tests of statistical significance by Student method (t test) and χ ².
RESULTS
The group of young hypertensive patients with ages 18-35 years, with elevated blood pressure >140/90mmHg, with increased plasma renin levels over 4.3ng/ml we found 49 cases of renal artery stenosis representing 40.50%, 8 cases of congenital small kidney representing 6.61%, 4 cases of Grawitz tumors (renal cell carcinoma) representing 3.30% and 60 cases of pheochromocytoma representing 49.59%. Table No.1
Table 1. The main conditions that were present in the group of young hypertensive patients with increased plasma renin level.
|
Diseases |
No. of cases |
Percentage of cases [%] |
|
Vascular pathology |
49 |
40.50% |
|
Renal parenchymatous pathology Congenital small kidney |
8 |
6.61% |
|
Renal carcinoma (Gravitz tumor) |
4 |
3.30% |
|
60 |
49.59% |
The positive criteria’s of diagnostic for the diseases were included in the study was:
I. Renal artery stenosis
- The increased value of diastolic blood pressure over 110mmHg.
- Paraombilical systolic murmur.
- Imaging of arteriography.
- Increased plasmatic renin level > 4.3 ng / ml.
II. Congenital small kidney
- Values of blood pressure over 140/90mmHg.
- Arteriography – put in evidence the small kidney
- The abdominal MRI
- Increased plasmatic renin level > 4.3 ng / ml.
III. Renal carcinoma (Gravitz tumor)
- Unilateral lumbar pain
- Loss of appetite
- Weight loss
- Macroscopic hematuria (blood in the urine)
- The value of plasma rennin level increased > 4.3ng /ml, mentioning that in this situation the plasma renin values reached the highest values up to 320ng/ml
- The abdominal MRI
- The renal biopsy
IV. Pheochromocytoma
- The paroxysmal outbursts of severe blood pressure over 220/120mmHg
- Headache
- Tremor of extremities
- Nervousness
- Increased serum catecholamine levels above 260pg/ml
- Increased urinary catecholamine values above the 7.4 mg/24 hrs
- Increased plasmatic renin level > 4.3 ng/ml.
- The abdominal MRI used in the detection of adrenal tumors .
Table 2. Diagnostic criteria’s met by patients
|
Diseases |
No. of cases |
Diagnostic criteria’s met by patients |
|
Vascular pathology Renal artery stenosis |
49 |
24 cases 4 of 4 25 cases 3 of 4 |
|
Renal parenchymatous pathology Congenital small kidney |
8 |
8 cases 4 of 4
|
|
Renal carcinoma (Gravitz tumor) |
4 |
2 cases 7 of 7 2 cases 6 of 7 |
|
Pheochromocytoma |
60 |
38 cases 8 of 8 12 cases 7 of 8 10 cases 6 of 8 |
Of the group of young hypertensive patients studied with increased plasma renin activity, 49 of the cases were renal artery stenosis representing 40.50% (p <0.001). The parameters of the clinical assessment were the increased value of diastolic blood pressure over 110mmHg, paraombilical systolic murmur and an imaging of arteriography. Figure 1.
Figure 1. Arteriography of the right renal artery stenosis (M.I. aged 21 years with HBP = 170/120mmHg)
Of the group of young patients participating in the study, we found 8 cases of unilateral small kidney representing 6.61% (p<0.001). The pathogenic mechanism of hypertension was ischemic, in that all cases arterial high blood pressure evolved along with hyperreninemia in congenital unilateral small kidney. The early diagnosis of renal disease is very important, in the best cases before the hypertension causes severe nephroangiosclerosis on the contralateral kidney, leading to nephrectomy which then can then lead to the disappearance of hypertension. The parameters of assessment in this case were the clinical blood pressure values above 140/90mmHg, imaging methods to put in evidence the small kidney: arteriography Figure2 abdominal MRI Figura3 and biological-increased plasmatic renin activity> 4.3 ng / ml.
Figure2. Arteriography evidence the congenital small left kidney (D.R. of 19 years old with HBP = 165/110mmHg)
Figure 3. MRI – scan with contrast substance putting in evidence the left renal hypoplasia (D.R. of 19 years old with HBP = 165/110mmHg)
Renal cell carcinoma (renal carcinoma, previously “hypernephroma” – Grawitz tumor) was responsible for the younger group of patients studied, 4 cases representing 3,30% (p<0.001) of the HBP young cases. All had severely elevated blood pressure values over 200/100mmHg. The diagnostic was based on clinical parameters: unilateral lumbar pain, loss of appetite, weight loss, but only two cases had macroscopic hematuria (blood in the urine), biological – the value of increased plasma rennin level > 4.3ng /ml, mentioning that in this situation the plasma renin values reached the highest values up to 320ng/ml. Imaging parameters are represented in the abdominal MRI by Figure 4.
Figure 4. MRI-scan with bilateral renal tumor (F.R.of 34 years with malignant HBP=220/130mmHg — worked with pesticides)
Histopathological parameters were put into evidence in all four cases in which renal biopsy was performed and the histopathological results of which are outlined below:
Two cases were clear cell renal carcinoma based on the histopathology results after renal biopsy – histological preparation with H&E staining with the objective of 10X is shown in (Figure 5a) and (Figure 5b)
Figure 5a. Clear cell renal carcinoma (objective 10x) – H&E stain (M.I. 21 years with paroxysmal HBP=200/110mmHg)
Figure 5b. Clear cell renal carcinoma (objective 10x) – H&E stain (P.R. 28 years with severe form HBP=210/110mmhHg)
The other two cases of renal carcinoma are represented in the following H&E stained images, after the renal biopsy (Figure 6) and (Figure 7).
Figure 6. Renal carcinoma (objective 10x). H & E stain (I.G. 31 years with paroxysmal HBP=210/115 mmHg)
Figure 7. Renal carcinoma (objective 10x). H & E stain (F.R. 34 years with malignant HBP=220/130mmHg — worked with pesticides)
Hypertension in the young patients with renal cell carcinoma took the form of severe paroxysmal HBP=200/110mmHg or above in all four cases, due to excessive secretion of renin produced in large quantities by the tumor and it was the one form which attracted most the clinical attention when it was not manifested by macroscopic hematuria.
Of the group of hypertensive young adults studied, 60 of the cases were of pheochromocytoma representing 49.59%.
The diagnostic criteria used in this clinical situation were: paroxysmal outbursts of severe blood pressure values over 220/120mmHg accompanied by headache, tremor of extremities, nervousness, biological parameters represented by increased serum catecholamine levels above 260pg/ml, increased urinary catecholamine values above the 7.4 mg/24 hrs, imaging parameters which were used in the detection of adrenal tumors by performing an abdominal MRI. Figure 8.
Figure 8. Abdominal MRI-scan with pheochromocytoma of the right adrenal gland (G.R. 24 years with paroxysmal HBP=220/130mmHg)
DISCUSSIONS
The importance of this study was to measure the level of plasma renin of hypertensive young patients with ages between 18-35 years to determine its role in the pathogenesis of secondary hypertension in the young adults. Also the conditions in which plasma renin level is increased in the context of secondary hypertension in the young patients.
Renovascular hypertension was one of the important causes of secondary hypertension in the young, its frequency in the group of patients studied was of 49 cases with renal artery stenosis representing 40.50% (p <0.001), in all these cases the renin plasma level was increased above 4,3ng/ml.
Safian R.D. and Textor S.C. [1] found the frequency of renal artery stenosis in a group of young hypertensive patients with increased plasma renin activity, as being 42.36%, which is slightly higher than in our study, this difference could be explained by a better paraclinical investigation of the young patients with hypertension.
Of the group of young hypertensive patients participating in the study we found 8 cases of unilateral congenital small kidney, representing 6.61% (p<0.001).
Goddard C, et al. [2] found that the incidence of hypertension in young people with kidney hypoplasia was 25%. They suggested that the renin-angiotensin-aldosterone system plays an important role in the pathogenesis of hypertension in the situation of renal hypoplasia. This difference could be explained by the fact that young patients in other countries had an increased teratogenic risk compared with the young in our country.
Renal cell carcinoma (Grawitz tumor) was responsible for the younger group of patients studied, 4 cases representing 3.30% (p<0.001) of all the young hypertensive patients. Two cases were clear cell renal cell carcinoma histopathology analyzed after a renal biopsy. The data obtained are slightly lower than those in the literature (5%) Sukarochana [3] and (4%) Gangurly [4]. This difference could be explained by the fact that our country carcinogenic risk factors are lower.
Rose HJ, Pruitt AW [5] reported the case of a young patient with severe hypertension of 190/110mmHg, which after further investigations had found increased plasma renin levels and after paraclinical investigations a solitary simple kidney cyst was found.
DW Robertson et al. [6] reported the case of a young man who had elevated blood pressure (HBP=180/120mmHg) and after investigations increased plasma renin level was found and a left renal tumor (primary reninoma) was found, whose blood pressure values were normalized after tumor resection.
Pheochromocytoma was found in 60 of the cases representing 49.59% of cases of the young hypertensive adults.
Abrams HL [7] found that the incidence of pheochromocytoma in the young hypertensive cases was 21.03%, Bravo EL [8] found 42.38% cases of pheochromocytoma, and Bravo EL, Gifford RWJr [9] 46.03 % of young hypertensive patients with pheochromocytoma. These results are lower than those obtained in our study. This could be explained by the risk factors in this geographical area and dominant genetic factor has an important role in the etiology of pheochromocytoma.
CONCLUSIONS
- Plasmatic renin level is an important marker of hypertension in the young adults.
- The highest plasmatic renin levels up to 320ng/ml were found in the cases of renal cell carcinoma, because the kidney tumor cells secrete increased amounts of renin.
- This situation suggests that hypertension in the young adults is hyperreninemia hypertension in the most cases dominated by a vasoconstriction and increased peripheral vascular resistance due hyperactivity of the sympathetic nervous system, being a rapidly evolving form of hypertension with vascular complications.
- The results of plasma renin dosing shows its important role in the pathogenesis in secondary hypertension of the young adults, these conditions are not quite as rare as one might think but not enough investigated.
- This marker should be routinely performed in young patients with hypertension, especially those with medium and severe forms of blood pressure > 170/100mmHg, having a role in establishing the etiology of hypertension in the young, however presently it is still not made often enough, but situation must to be change in the future.
REFERENCES:
[1] Safian R.D, Textor S.C, Renal-artery stenosis, N Engl J Med 2001, 344(6):431-42.
[2] Goddard C , Vallothon MB, Broyer M, Plasma rennin activity in segmental hypoplasia of kidneys with hypertension, Nephron 2003, 11:308-17.
[3] Sukarochana K, Nephroblastoma and hypertension J Surg 2005, 7- 573.
[5] Rose HJ, Pruitt AW, Hypertension, hyperreninemia and a solitary renal cyst in an adolescent, Am J Med 2004, 61; 579-82.
[6] Robertson DW, Klidjiana A, Harding KK, Walters G, Lee MR, Robb-Smith AHT, Hypertension due to a renin-secreting renal tumor, Am Med 2005, 43 (9) 63-76.
[8] Bravo EL, Pheochromocytoma: New concepts and future trends, Kidney Int 1991, 40:544-556.
[9] Bravo EL, Gifford RWJr, Pheochromocytoma: Diagnosis, localization and management, N Engl J. Med 1984, 311-1298.
Corresponding author
Manuela Stoicescu, Internal Medicine Department, University of Oradea, Faculty of Medicine and Pharmacy, Oradea, Romania: County Hospital of Oradea, Phone 0723019951, e-mail: manuela_stoicescu@yahoo.com
Arterial Hypertension in Young Adults: An Ignored Chronic Problem
Posted in Biomarkers & Medical Diagnostics, HTN in Youth, tagged Blood pressure, Cardiovascular Disorders, Conditions and Diseases, Hypertension on February 9, 2013| 4 Comments »
Arterial Hypertension in Young Adults: An Ignored Chronic Problem
Author: Manuela Stoicescu, MD, PhD
Manuela Stoicescu, MD, PhD
Consultant Internal Medicine, Assistant Professor
Faculty of Medicine and Pharmacy, Medical Disciplines Department
University of Oradea, Romania
ABSTRACT
Introduction:
Don’t ignore the young patients: being young does not necessary mean being healthy.
Objectives:
The objectives in this study was to analyzed the principal clinical aspects and conduct laboratory investigations with young people in group of age 18-35 years. Attracting attention to the diagnosis of hypertension in the young in the early stages of life. I choice this topic because high blood pressure in the young, in particularly, in this group age was insufficiently studied while a high frequency of cases presented every day was continuously increasing.
Material and method:
The study was performed in the Ambulatory Specialty of the Internal Diseases Department in the County Hospital in Oradea, Romania. Study period was 1 October 2006 to 31 July 2009. Included in the study were 321 patients with hypertension exceeding 140/90 mmHg which was maintained higher after three consecutive determinations in intervals of one week to exclude the “white coat phenomenon”, an effect noted very frequent in young people, especially in young women, because a young persons have hyperactivity of simpatico nervous systemic, or the value of blood pressure was higher more than 170/110 mmHg from first determination.
Results
1. Importance of the genetic factors in the etiologies of disease was suggests that family prevalence of hypertension in the young people and another family diseases like hyperaldosteronism, polycystic kidney and multiple endocrine neoplasias MEN2a.
2. Importance of personal pathologic antecedents demonstrated in my study that repetitive Streptococcus angina with Streptococcus β hemolytic group A originated in the first place as a cause in hypertension in the young people in context of acute streptococcal renal parenchymatous diseases.
3. Renin plasmatic level is a very important marker of high blood pressure in the young. It was high in 121 cases (37.69%). This situation suggests that hypertension in the young is hyperreninemic hypertension in many cases because a young person has a systemic hyperactivity of simpatico nervous.
4. Left ventricular hypertrophy is been detected in X-ray, ECG and echocardiography. In my studied I detected left ventricular hypertrophy in 35 patients representing 10.49%.
5. Proteinuria was represented in 96 cases (29.90%) has two meanings:
- nephropathy complication of hypertension or
- acute glomerulonephritis or
- nephritis syndrome accompaniment with hematuria 38 cases (11.83%).
6. The eye ground findings of young people with hypertension are frequently normal. In the absence of prior readings, one needs to look for evidence of target organ damage that may suggest chronicity. In my study this changes appeared for 86 cases 23.3 %, hemorrhages and exudates I rarely found in 9 cases represent 2.8% and papilla edema was presented in 2 cases even when hypertension was very severe more than 200/120 mmHg and complicate with hypertensive encephalopathy.
Conclusions: Guidelines for hypertension treatment with young patients group of ages 18-35 was developed, which I hope will help the activity of physicians in general specialties in their practice, to use for diagnosis and easy work. This is new and hypertension in the young in this group of ages was insufficiently studied.
Key words: hypertension diagnosis, young adults.
INTRODUCTION
Prior to the last twenty years it was impossible to accept the idea that hypertension and atherosclerosis begin in adolescence and even earlier in childhood. Current concepts concerning the nature of hypertension in the young are changing. Earlier clinical impressions indicated that hypertension in the young was secondary and the essential hypertension occurred only rarely.
In my recent study, involving young people, of the age group of age 18-35 year old, indicated that young with high levels of blood pressure often the cause is known and often is unknown. When high BP (HBP) is found in the young the young compared with their peers, it is likely that the HBP will continue to Adulthood. My study has indicated that the level of blood pressure in young is closely related to the occurrence of hypertension in adulthood. Thus, changing concepts suggest that essential hypertension begins in early life. Considerable information is now known about the clinical and pathologic features of hypertension in adults.
We understand clinical diagnoses, the pathophysiology and humoral background, and the consequences of end stage renal disease (ESRD). We are even beginning to consider that essential hypertension may represent more than one disease. By contrast, little is known about the early natural development of essential hypertension. For example, how can hypertension in young be defined? We cannot equate level of blood pressure with cardiovascular damage as in adults (cardiovascular, cerebral, and renal disease). Furthermore, there is little specific information that can be used to predict development of adult hypertension. As a beginning, descriptive studies of the early natural development of essential hypertension are needed. It is logical to assume that prevention would be most successful if the disease process could be understood and treated in its earliest phase.
OBJECTIVES
Don’t ignore the young adult patient. Being young does not necessary means being healthy. Key objectives in my study was to analyze the principal clinical aspects and laboratory tests performed on young adults in the group age 18-35 year old, to advocate for the attention needed for diagnosis of hypertension in the young adults in the early stages of the disease.
MATERIAL AND METHOD
The study was performed in the Ambulatory Specialty of the Internal Diseases Department at the County Hospital in Oradea, Romana. Study period was 1 October 2006 to 31 July 2009. Study participants:
- 321 young patients,
- group of ages 18-35, patients with high blood pressure more than 140/90 mmHg
- after three consecutive determinations in interval one week maintain higher than 140/90 mmHg to exclude the “white coat phenomenon”, effect very frequently encountered with young adults especially with young women, because young person have a hyperactivity of sympathetic nervous system, or
- the value of blood pressure was high more than 170/110mmHg from first determination.
The patients had a comprehensive physical examination (clinical and par clinical) and diagnosed with hypertension in different stages.
The study consideration was done after having confirmed the diagnosis of hypertension and the standardization according to the phenomenon of high blood pressure and the classification of OMS.
The patients agreed to participate after being introduced in the study after they have been explained the deontological and preserving of the confidentiality criteria.
For statistics data I has been used the EPIINFO application, 6.0 version, a program of The Center of Disease Control and Prevention- Atlanta, with the Student method (test t) and χ²
RESULTS AND DISCUSSIONS
We observed that a 1/5 of the patients studied have in family antecedents of young adults hypertensive member of the family:hypertension in 70 cases (21.80%), stroke in 46 cases (14.33%), myocardial infarction in 55 cases (17.13%), peripheral vascular disease in 23 cases ( 7.16%) obesity 38 (11.83%), pre-eclamptic toxemia in 31 cases(9.65% ), hyperaldosteronism in 18 cases (5.60%), polycystic kidney in 26 cases (8.09%), multiple endocrine neoplasias MEN2a in 14 cases (4.36%) Distribution of cases according to family history. See, Table 1.
Table 1: Distribution of Cases according to Family History
|
Consideration |
No. of cases |
Percent |
|
Hypertension for parents, grandparents, aunts, uncles and cousins |
70 |
21.80% |
|
Family antecedents of stroke |
46 |
14.33 % |
|
Family antecedents of myocardial infarction |
55 |
17.13% |
|
Family antecedents of peripheral vascular disease |
23 |
7.16% |
|
Family antecedents of obesity |
38 |
11.83% |
|
Pre-eclamptic toxemia |
31 |
9.65% |
|
Hyperaldosteronism |
18 |
5.60% |
|
Polycystic kidney |
26 |
8.09% |
|
Multiple endocrine neoplasias MEN2a |
14 |
4.36% |
A significant numbers of patients in my studies did not have any antecedents of hypertension in their family history. That fact demonstrates that not only genetic factors have an important role in the etiology of the disease.ENvironmental factors count.
Significant number of hypertensive young patients had diseases in their personal history: Scarlatti in 27 (8.41%), repetitive angina with Streptococcus β hemolytic group A in 88 (27.41%), chronic ORAL infection focus in 35 (10.90%) chronic stomathological focus infections in 19 (5.91%), nephritis in 34(10.59%), endocrine disorders in 16 (4.98% ), physical and psychological in 22 (6.85%), head trauma in 11 (3.42%), therapy with corticosteroids secondary to another disease (for example erythematous systemic lupus) in 5 (1.55%), therapy with AINS drugs in 21 (6.54%), use decongestion nasal in 4 (1.24%) repetitive urinary tract infection in 28 (8.72%), syphilis in 11 (3.42%). See, Table II.
Table II: Distribution of Cases by Illnesses in Personal History
|
Consideration |
No. of cases |
Percent |
|
Scarlatti |
27 |
8.41% |
|
Repetitive Streptococcus angina with Streptococcus β hemolytic group A |
88 |
27.41% |
|
Chronic ORAL infection focus |
35 |
10.90% |
|
Chronic stomathological infection focus |
19 |
5.91% |
|
Nephritis |
34 |
10.59% |
|
Endocrine disorders |
16 |
4.98% |
|
Physical and psychical suprademanding |
22 |
6,85% |
|
Head trauma |
11 |
3,42% |
|
Therapy with corticosteroids |
5 |
1.55% |
|
Therapy with AINS |
21 |
6.54% |
|
Use decongestion at nasal |
4 |
1.24% |
|
Repetitive urinary tract infections |
28 |
8.72% |
|
Syphilis |
11 |
3.42% |
Fig.1: Principal Diseases Etiology for Young Hypertensive Patients
Table III: Laboratory Results
|
Hemoglobin value ↑ |
18 |
5.60% |
|
Hematocrit ↑ |
18 |
5.60% |
|
Value of glucose ↑ |
68 |
21.18% |
|
Cholesterol ↑ |
78 |
24.29% |
|
HDL cholesterol ↑ |
86 |
26.79% |
|
LDL cholesterol ↑ |
77 |
23.67% |
|
Triglycerides ↑ |
105 |
32.71% |
|
Uric acid ↑ |
57 |
17.75% |
|
Creatinina ↑ |
38 |
11.83% |
|
Urea ↑ |
36 |
11.21% |
|
Serum sodium ↑ |
42 |
13.08% |
|
Serum potassium↓ |
42 |
13.08% |
|
Urinalysis -albuminuria+ -hematuria+ |
96 |
29.90% |
|
38 |
11.83% |
|
|
Urine culture with female + |
104 |
32.29% |
Table IV: Laboratory Special Tests
|
Rennin plasmatic↑ |
121 |
37.69% |
|
Vanillyl Mandelic Acid testing (VMA): in urine↑ |
18 |
5.60% |
|
Catecholamine urine↑ |
18 |
5.60% |
|
Cortisol urine ↑ |
9 |
2.80% |
|
Cortisolemia ↑ |
9 |
2.80% |
|
TSH ↑ |
16 |
4.98% |
|
T3 ↑ |
16 |
4.98% |
|
T4 ↑ |
16 |
4.98% |
|
CT abdominal |
114 |
35.51% |
|
RMN abdominal |
158 |
49.92% |
|
Intravenous urogrography |
102 |
31.77% |
Observations on Eye Exam and Retinopathy [The eye ground (eye ground findings)]
Clearly, the most helpful information to have when one is attempting to establish the chronicity of hypertension is past blood pressure readings. Unfortunately, these are by no means always available since routine blood pressure measurement in young adults is not yet uniformly obtained. In the absence of prior readings, one needs to look for evidence of target organ damage that may suggest chronicity. In adolescent with even severe chronic hypertension or hypertensive encephalopathy. In my study this changes appear for the optic fund may show no more than retinal arteriolar narrowing in 103 cases represent 32.09% and arterio-venous nicking in 98 cases represent 30.53%, hemorrhages and exudates I rarely found in 9 cases represent 2.8%, papilla edema may be absent except in 2 cases even when hypertension was very severe more than 200/120 mmHg with complications of encephalopathy and in 109 cases represent 33.96 was normal result of eye ground examination. See Table V and Fig. 2
Just as there may be minimal eye ground findings, there are infrequently cardiac findings that suggest chronicity.
Table V Distribution of Cases by Eye Exam Findings
|
Normal |
109 |
33.96% |
|
Arteriolar narrowing |
103 |
32.09% |
|
Arterio-venous nicking |
98 |
30.53% |
|
Exudates and hemorrhages |
9 |
2.80% |
|
Papilla edema |
2 |
0.62% |
Fig. 2: Distribution of Cases by Changes of Eye Ground Findings
The heart morphology was not clinically enlarged in many cases and the ECG and chest X-ray were usually unhelpful in detecting left ventricular hypertrophy unless hypertension has been prolonged and severe. In my studies left ventricular hypertrophy was present in 35 cases (10.49%), they are helpful in determining chronicity of hypertension and in 206 cases (64.18%) left ventricular hypertrophy was absent. If negative suggesting nothing about the duration of hypertension. See Table VI and Fig 3
Table VI: Distribution of Cases by Changes in Chest X-Ray
|
Normal |
206 |
64.18% |
|
Elongation and elevated of left inferior arcos |
99 |
30.85% |
|
Cardiomegaly |
12 |
3.73% |
|
Aneurism of thoracic aorta |
4 |
1.24% |
Fig. 3: Distribution of Cases by Changes of chest X-Ray
The echocardiography seems to be more sensitive for evaluating chamber size and wall thickness than the ECG and can be helpful. Left atria hypertrophy and left ventricular hypertrophy (Sokolow -Lyon index) and left axial deviation was possible to detect. In my studies I found 35 cases (10.49%) with LVH, 36 cases (11.21%) with LAH and 35 cases (10.49%) with left axial deviation. Secondary changes of depolarization like ST segment sub elevated and negative T wave I found in 35 cases represent 10.49%. See Table VII and Fig. 4
Table 7: Distribution of Cases by Changes in ECG
|
Normal |
180 |
56.07% |
|
Left axial deviation> -30 |
35 |
10.90% |
|
Left atria hypertrophy |
36 |
11.21% |
|
Left ventricular hypertrophy Sokolov -Lyon index(SV1+RV5/V6>35mm) |
35 |
10.90% |
|
Secondary changes of depolarization – ST segment sub elevated and T wave negative |
35 |
10.90% |
Fig. 4: Distribution of Cases by Changes in ECG
Table VIII: Distribution of cases by Echocardiography of Hearth Examination
|
Normal |
226 |
70.40% |
|
Left ventricular hypertrophy |
40 |
12.46% |
|
Ejection fraction(FE) of left ventricular<55% |
27 |
8.41% |
|
Aortic coarctation |
28 |
8.72% |
Fig. 5: Distribution of cases by Changes in Echocardiography
Table IX: Distribution of Cases by Urine Analysis Results
|
Normal |
187 |
58.27% |
|
Proteinuria |
96 |
29.90% |
|
Hematuria |
38 |
11.83 % |
Proteinuria I detect in 96 cases (29.90%) and hematuria in 38 cases (11.83%). See Fig. 6
Fig.6: Distribution of cases by Urine Analysis Results
OMS stadialization classification high blood pressure in three stages. In my study about hypertension in the young adults, the results are as follows:
- Stages I: 270 cases represents 84.11%,
- Stages II: 40 cases represents 12.46%,
- Stages III: 9 cases (2.80%) and
- Stage IV: malign hypertension 2 cases represents 0.62%.
See, Table IX and Fig.7
Table IX: Distribution of Cases by Stadialization
|
Stages I |
270 |
84.11% |
|
Stages II |
40 |
12.46% |
|
Stages III |
9 |
2.81% |
|
Stages IV |
2 |
0.62% |
Fig. 7: Distribution of Cases by Stadialization
DISCUSSION
1. A 1/5 of group of patients studied have in family antecedents of young hypertensive family member with the following diseases:
- stroke 46 cases (14.33%),
- myocardial infarction 55 cases (17.13%),
- peripheral vascular disease 23 cases (7.16%),
- obesity 38 (11.83%),
- pre-eclamptic toxemia 31 cases (9.65%),
- hiperaldosteronism in 18 cases (5.60%),
- polycystic kidney 26cases (8.09%),
- multiple endocrine diseases II 14 cases (4.36%).
These results are in concordance with observations of Kotchen JM [1] which in a studies about young hypertensive patients concluded that family aggregation of hypertension was very frequent 20.2% (p<0,001) suggesting the importance of a genetic factor in the etiology of hypertension in the young adults.
2. An important number of hypertensive young patients were present in personal pathological antecedents: Scarlatti 27 (8.41%), repetitive angina with Streptococcus β hemolytic group A 88 (27.41%),chronic ORL infection focus 35 (10.90%) chronic stomathological focus infections 19 (5.91%), nephritis 34 (10.59%), endocrine disorders 16 (4.98% ), physical and psychical supra solicitation 22 (6.85%), head trauma 11 ( 3.42% ), therapy with corticosteroizi from another disease (for example erithematous systemic lupus) 5 (1.55%) therapy with AINS drugs 21 (6.54%), use decongestion nasal 4 (1.24%) repetitive urinary tract infection 28 (8.72%), syphilis 11 (3.42%)(p<0,001). Loggie JMH [2] in a studies with hypertension in the young reported the streptococcus infection with Streptococcus β hemolytic group A was 18.2% , chronic ORAL infection focus was 8.9%, chronic stomathological focus infections was 3.98%, glomerulonephritis was 6.2% and physical and psychical supra solicitation was 12.43% in personal pathological antecedents.
3. Changes of retinal vascular were insufficiently studied in young adults. In my study this changes appear for the optic fund may show no more than retinal arteriolar narrowing 103 cases represent 32.09% and arterio venous nicking 98 cases represent 30.53% , hemorrhages and exudates I rarely found from 9 cases represent 2.8%, papilla edema may be absent except 2 cases even with hypertension was very severe more than 200/120mmHg and complicate with encephalopathy and 109 cases represent 33.96% was normal result of funduoscopic examination (p<0.001).
Skalina MEL et al. [3] observations: 281 hypertensive young patients 140 have changes for the optic fund arterio venous nicking 93 cases, hemorrhages found from 7 cases and exudates appear from 40 patients. 141 patients have the optic fund examination normal.
4. The heart is not often clinically enlarged and the ECG and chest X-ray are usually unhelpful in detecting left ventricular hypertrophy unless hypertension has been prolonged and severe. In my studies left ventricular hypertrophy was present in 35 cases (10.49%), they are helpful in determining chronicity of hypertension and from 206 cases (64.174%) left ventricular hypertrophy was absent, that suggest that if negative, they tell one nothing about the duration of hypertension. The results are in concordance with observation with Laird WP and Fixler DE [4] who reports after performing chest X-ray for 210 young hypertensive, 103 have normal results, 78 have elongation and elevated of left inferior arcos and 29 present’s cardiomegaly.
5. The echocardiogram seems to be more sensitive for evaluating chamber size and wall thickness than the ECG and can be helpful. Left atria hypertrophy and left ventricular hypertrophy (Sokolow-Lyon index) and left axial deviation it’s possible to detect. In my studies I found 35 cases (10.49%) with LVH, 36 cases (11.21%) with LAH and 35 cases (10.49%) with left axial deviation. Secondary changes of depolarization like ST segment sub elevated and negative T wave I found from 35 cases represent 10.49% (p<0,001).The results are in concordance with observation with Laird WP and Fixler DE [4], who reports than 18% from young hypertensive subject, presents left ventricular hypertrophy detected after echocardiography examination, end in concordance with observation with Schieken RM et al. [5] in Muscatine studies who reports more than 14% from young hypertensive subjects have left ventricular hypertrophy after make echocardiography examination.
6. Proteinuria I detect in 96 cases (29.90%) and hematuria in 38 cases (11.83%).This changes appear in context of acute glomerulonephritis and hypertension was secondary renal.
Schmider et al. [6] sustained that glomerular hyperfiltration is a early marker for nefroangiosclerosis and a sign for subclinical organ affected.
7. OMS stadialization classification high blood pressure in three stages. In my study about hypertension in young adults the results are: in stages I found 270 cases represents 84.11%, in stages II 40 cases represents 12.46%, in stages III 9 cases (2.80%) and malign hypertension 2 cases represents 0.62%.
CONCLUSIONS
- 1. Importance of genetic factors in etiologies of disease is suggested that family aggregation of hypertension in young adults and another familial diseases like hyperaldosteronism, polycystic kidney and multiple endocrine diseases II.
- 2. Importance of personal pathologic antecedents demonstrated in my study that repetitive Streptococcus angina with Streptococcus β hemolytic group A was found in the first place as a cause of hypertension in the young people in context of acute streptococcal renal parenchymatous diseases.
- 3. Except nonspecific symptoms of high blood pressure exist specifically symptoms who suggest etiology of hypertension with young people.
- 4. The fundoscopic findings in the young adult with hypertension are frequently normal. In the absence of prior readings, one needs to look for evidence of target organ damage that may suggest chronicity. In my study this changes appear for 86 cases 23.3 % and hemorrhages and exudates I rarely found from 9 cases represent 2.8% and papilla edema may be absent except 2 cases even with hypertension is very severe more than 200/120mmHg and complication of encephalopathy.
- 5. Left ventricular hypertrophy is possible to detect X-ray, ECG and echocardiography. In my studied I detected left ventricular hypertrophy from 35 patients represent 10.49%.
- 6. Proteinuria 96 cases (29.90%) have two significance: nephropathy complication of high blood pressure, or etiology in context or glomerulonephritis alone or accompaniment with hematuria 38 cases (11.83%) in nephritis syndrome.
- 7. Renin plasmatic level is very important marker of high blood pressure in the young. Was high in 121 cases (37.69%).This situation suggests that hypertension in the young is hiperreninemic hypertension in many cases because young adults have hyperactivity of sympatetic nervous system.
- 8. OMS classification evaluated stages I 270 cases (84.11%), stages II 40 cases (12.46%) in stages III 9 cases (2.80%) and malign hypertension (stages IV) 2 cases (0.62%)
- 9. Finally I make a small guideline about hypertension with young patients group of ages 18-35, which I hope to help activity of every physician indifferent specialties in your practice, to use for diagnosis and easy work.
REFERENCES
1. Kotchen JM “Effect of relative weight on familial blood pressure aggregations“ Am J Epidemiol.1987 105-214.
2. Loggie JMH. “The diagnostic evaluation of adolescents with hypertension.” In Hunt JC, Dreifus LS, Dustan HP et al, eds Dialogues in Hypertension Update II vol 1. Lyndhurst, NJ: Health Learning Systems 1984:43-56.
3. Skalina MEL et al.: Annable WL, Kleigman RM, Fanaroff AA “ Hypertensive retinopathy in the adolescent“ J Adolesc. 1983:103:781-6.
4. Laird WP and Fixler DE. “Left ventricular hypertrophy in adolescents with elevated blood pressure: assessment by chest roentgenography, electrocardiography and echocardiography.” Adolescents 2001;67:255-9.
5.Schieken RM and coauthors: Clarke WR, Lauer RM, “Left ventricular hypertrophy in the young with blood pressures in the upper quin-tile of the distribution: the Muscatine study.” Hypertenesion 2004;3:669-75.
6. Schmider and coauthors: Messerli FH, Garavaglia GE, Nunez BD “Glomerular hyperfiltration indicates target organ disease in essential hypertension.” Circulation 2003; 76: III-273.



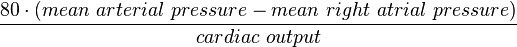
 , where t is time, s is a complex parameter, and A is a complex scalar. Complex values simply mean two dimensional, e.g., magnitude (as in resistance) plus phase shift (to account for reactive components).
, where t is time, s is a complex parameter, and A is a complex scalar. Complex values simply mean two dimensional, e.g., magnitude (as in resistance) plus phase shift (to account for reactive components).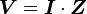 where V and I are the complex scalars in the voltage and current respectively and Z is the complex impedance.
where V and I are the complex scalars in the voltage and current respectively and Z is the complex impedance.














