Risks for Patients’ and Physician’s Health in the Cath Lab
Reporter and Curator: Aviva Lev-Ari, PhD, RN
| On Thursday, June 27th, 2013, Bayer HealthCare, Nuance® Healthcare, and The Mount Sinai Hospital held a live webinar outlining how one of America’s leading Radiology Departments is pioneering the next generation of imaging informatics. If you were unable to watch it live, or would like to view it again, it is now available online here. | ||
| The Mount Sinai Hospital in New York has taken Contrast Dose Management and IT interoperability to a new level with two industry-leading forces – Bayer’s Certegra® Informatics Platform and Nuance’s PowerScribe® 360 | Reporting. | ||
|
|||||||||||||||||||||||||
|
Risks for Physician’s Health in the Cath Lab
Brain tumours among interventional cardiologists: a cause for alarm? Report of four new cases from two cities and a review of the literature.
Source
Interventional Cardiology, Rambam Medical Center, Bruce Rappaport Faculty of Medicine, the Technion, Israel Institute of Technology, Haifa, Israel. aroguin@technion.ac.il
Abstract
AIMS:
Interventional cardiologists who work in cardiac catheterisation laboratories are exposed to low doses of ionising radiation that could pose a health hazard. DNA damage is considered to be the main initiating event by which radiation damage to cells results in development of cancer.
METHODS AND RESULTS:
We report on four interventional cardiologists, all with brain malignancies in the left hemisphere. In a literature search, we found five additional cases and thus present data on six interventional cardiologist and three interventional radiologists who were diagnosed with brain tumours. All worked for prolonged periods with exposure to ionising radiation in the catheterisation laboratory.
CONCLUSIONS:
In interventional cardiologists and radiologists, the left side of the head is known to be more exposed to radiation than the right. A connection to occupational radiation exposure is biologically plausible, but risk assessment is difficult due to the small population of interventional cardiologists and the low incidence of these tumours. This may be a chance occurrence, but the cause may also be radiation exposure. Scientific study further delineating occupational risks is essential. Since interventional cardiologists have the highest radiation exposure among health professionals, major awareness of radiation safety and training in radiological protection are essential and imperative, and should be used in every procedure.
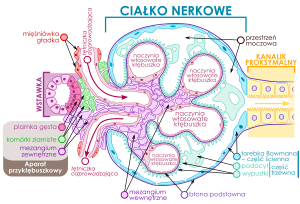
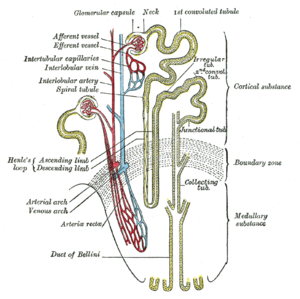
Risks for Patients’ Health in the Cath Lab
Contrast-Induced Nephropathy
- Author: Renu Bansal, MD; Chief Editor: Vecihi Batuman, MD, FACP, FASN
SOURCE
http://emedicine.medscape.com/article/246751-medication#showall
Contrast-induced nephropathy (CIN) is defined as the impairment of renal function and is measured as either a 25% increase in serum creatinine (SCr) from baseline or 0.5 mg/dL (44 µmol/L) increase in absolute value, within 48-72 hours of intravenous contrast administration. (See Etiology.)
For renal insufficiency (RI) to be attributable to contrast administration, it should be acute, usually within 2-3 days, although it has been suggested that RI up to 7 days post–contrast administration be considered CIN; it should also not be attributable to any other identifiable cause of renal failure. A temporal link is thus implied.[1] Following contrast exposure, SCr levels peak between 2 and 5 days and usually return to normal in 14 days. (See Clinical and Workup.)
Complications
CIN is one of the leading causes of hospital-acquired acute renal failure. It is associated with a significantly higher risk of in-hospital and 1-year mortality, even in patients who do not need dialysis.
Nonrenal complications include procedural cardiac complications (eg, Q-wave MI, coronary artery bypass graft [CABG], hypotension, shock), vascular complications (eg, femoral bleeding, hematoma, pseudoaneurysm, stroke), and systemic complications (eg, acute respiratory distress syndrome [ARDS], pulmonary embolism).
There is a complicated relationship between CIN, comorbidity, and mortality. Most patients who develop CIN do not die from renal failure. Death, if it does occur, is more commonly from either a preexisting nonrenal complication or a procedural complication.
Concerns
Many physicians who refer patients for contrast procedures and some who perform the procedure themselves are not fully informed about the risk of CIN. A survey found that less than half of referring physicians were aware of potential risk factors, including diabetes mellitus. (See Differentials.)
CIN suffers from a lack of consensus regarding its definition and treatment. Studies differ in regard to the marker used for renal function (SCr vs eGFR), the day of initial measurement and remeasurement of the marker, and the percentage increase used to define CIN. This makes it difficult to compare studies, especially in terms of the efficacy of various treatment modalities. (See Treatment and Medication.)[2]
The reported incidence of CIN might be an underestimation. SCr levels normally rise by day 3 of contrast administration. Most patients do not remain hospitalized for so long and there is no specific protocol to order outpatient SCr levels 3-5 days after the procedure.
Other renal function markers
The use of SCr as a marker of renal function has its limitations. Indicators such as the estimated glomerular filtration rate (eGFR) and cystatin C are increasingly considered to be more reliable and accurate reflectors of existing renal function.[3, 4]
The eGFR can be calculated using the Modification of Diet in Renal Disease (MDRD) formula or the Cockroft-Gault formula. The Cockroft-Gault formula calculates eGFR using age, sex, and body weight, which are factors that, independent of GFR, influence SCr. The MDRD equation also includes blood urea nitrogen (BUN) and serum albumin.
The eGFR works best at low creatinine values. SCr and GFR share a curvilinear relationship. At lower SCr values, doubling SCr is associated with a corresponding 50% decrease in GFR. However, in elderly patients with chronic kidney disease(CKD) who have high SCr values at baseline, a 25% rise in SCr is actually indicative of a relatively modest reduction in GFR. Nonetheless, even a 25% increase in SCr in this situation has been shown to have great impact, especially in terms of inhospital and 1-year mortality.[5]
Serum cystatin C is a serum protein that is secreted by nucleated cells. It is freely filtered by the glomerulus and has been found to be an accurate marker of GFR. Compared with SCr, cystatin C changes much earlier after contrast administration and is not subject to confounding factors, such age, sex, and muscle mass, that influence SCr values independent of the underlying GFR. Cystatin C is increasingly being used as a marker of renal function in cardiac surgical patients.
Patient education
Patients with risk factors for CIN should be educated about the necessity of follow-up care with their physicians with a postprocedure SCr estimation, especially if the initial procedure was done on an outpatient basis.
Etiology
Contrast media (CM) act on distinct anatomic sites within the kidney and exert adverse effects via multiple mechanisms. They cause a direct cytotoxic effect on the renal proximal tubular cells, enhance cellular damage by reactive oxygen species, and increase resistance to renal blood flow. They also exacerbate renal vasoconstriction, particularly in the deeper portions of the outer medulla. This is especially important in patients with CKD, because their preexisting abnormal vascular pathobiology is made worse by the effects of CM.[6, 7]
Renal (particularly medullary) microcirculation depends on a complex interplay of neural, hormonal, paracrine and autocrine influences. Of note are the vasodilator nitric oxide (NO) and the vasoconstrictors vasopressin, adenosine (when it acts via the high affinity A1 receptors), angiotensin II, and endothelins. Prostaglandins cause a redistribution of blood flow to the juxtamedullary cortex and, therefore, are protective.
NO, in particular, seems to be very important, with antiplatelet, vasodilatory, insulin sensitizing, anti-inflammatory, and antioxidant properties. It has been suggested that plasma levels of asymmetrical dimethylarginine (ADMA), which is an endogenous inhibitor of all NO synthase isoforms, can be used as a marker of CIN, especially in patients with unfavorable outcomes.
CM-mediated vasoconstriction is the result of a direct action of CM on vascular smooth muscle and from metabolites such as adenosine and endothelin. Additionally, the osmotic property of CM, especially in the tubular lumen, decreases water reabsorption, leading to a buildup of interstitial pressure. This, along with the increased salt and water load to the distal tubules, reduces GFR and causes local compression of the vasa recta. All of this contributes to worsening medullary hypoxemia and renal vasoconstriction in patients who are already volume depleted.
Finally, CM also increase resistance to blood flow by increasing blood viscosity and by decreasing red cell deformability. This intravascular sludging generates local ischemia and causes activation of reactive oxygen species that result in tubular damage at a cellular level.
Comparison of contrast-agent nephropathy potential
The ability of different classes of CM to cause CIN is influenced by their osmolality, ionicity (the ability of the contrast media to dissociate in water), and molecular structure. Each of these characteristics, in turn, influences their behavior in body fluid and their potential to cause adverse effects. (See Table 1, below.)[8]
Agents are classified as high, low, or iso-osmolar, depending on their osmolality in relation to blood. Low-osmolarity contrast media (LOCM) is actually a misnomer, since these agents have osmolalities of 600-900 mOsm/kg and so are 2-3 times more hyperosmolar than blood. High-osmolarity contrast media (HOCM) are 5-7 times more hyperosmolar than blood, with osmolalities greater than 1500 mOsm/kg.
Molecular structure of CM refers to the number of benzene rings. Most CM that were developed in the 1990s are dimers with 2 benzene rings. Dimeric CM, while nonionic and with low osmolarity, have high viscosity, which may influence renal tubular blood flow.
The ratio of iodine to dissolved particles describes an important relationship between opacification and osmotoxicity of the contrast agent. The higher ratios are more desirable. High-osmolar agents have a ratio of 1.5, low-osmolar agents have a ratio of 3, and iso-osmolar agents have the highest ratio, 6.
While the safety of LOCM over HOCM in terms of CIN seems intuitive, clinical evidence of it came from a meta-analysis by Barrett and Carlisle.[9] They showed the benefit of using LOCM over HOCM mostly in high-risk patients. The Iohexol Cooperative Study was a large, prospective, randomized, double-blinded, multicenter trial that compared the risk of developing CIN in patients receiving the low-osmolarity agent iohexol versus the high-osmolarity agent diatrizoate. While the HOCM group was 3.3 times more likely to develop CIN compared with the LOCM group, this was seen only in patients with preexisting CKD (baseline SCr greater than or equal to 1.5 mg/dL). In addition to CKD; diabetes mellitus, male sex, and contrast volume were found to be independent risk factors.
Even within the LOCM category, the risk is not the same for all agents. High-risk patients receiving iohexol have a higher likelihood of developing CIN than do patients receiving another agent (ie, iopamidol) in the same class.
When LOCM were compared with iso-osmolar contrast media (IOCM), the Nephrotoxicity in High-Risk Patients Study of Iso-Osmolar and Low-Osmolar Non-Ionic Contrast Media (NEPHRIC study), arguably the most definitive study in this category to date, found that the odds of developing CIN in high-risk patients were almost 9 times greater for the study’s iohexol group than for the investigation’s iodixanol group (iso-osmolar contrast agent). The incidence of CIN was 3% in the iodixanol group versus 26% in the iohexol group.[10] These results, though promising, were not duplicated in some subsequent studies.
When iodixanol was used, the Rapid Protocol for the Prevention of Contrast-Induced Renal Dysfunction (RAPPID) trial found a 21% incidence of CIN,[11] and the Contrast Media and Nephrotoxicity Following Coronary Revascularization by Angioplasty (CONTRAST) trial found a 33% incidence of CIN.[12] Finally, the Renal Toxicity Evaluation and Comparison Between Visipaque (Iodixanol) and Hexabrix (Ioxaglate) in Patients With Renal Insufficiency Undergoing Coronary Angiography (RECOVER) trial compared the iso-osmolar contrast medium iodixanol to the low-osmolarity agent ioxaglate and found a significantly lower incidence of CIN with iodixanol than with ioxaglate (7.9% vs 17%, respectively).[13]
Thus, although the data are by no means uniform, they seem to suggest that the iso-osmolar contrast agent iodixanol may be associated with smaller increases in SCr and lower rates of CIN when compared with low-osmolar agents, especially in patients with CKD and in those with CKD and diabetes mellitus.[14]
Risk factors
Risk factors for CIN can be divided into patient-related, procedure-related, and contrast-related factors (although the risk factors for CIN are still being identified and remain poorly understood). Patient-related risk factors are as follows:
-
Age
-
CKD
-
Diabetes mellitus
-
Hypertension
-
Metabolic syndrome
-
Anemia
-
Multiple myeloma
-
Hypoalbuminemia
-
Renal transplant
-
Hypovolemia and decreased effective circulating volumes – As evidenced by congestive heart failure (CHF), an ejection fraction (EF) of less than 40%, hypotension, and intra-aortic balloon counterpulsation (IABP) use
Procedure-related risk factors are as follows:
-
Urgent versus elective
-
Arterial versus venous
-
Diagnostic versus therapeutic
Contrast-related risk factors are as follows:
-
Volume of contrast
-
Contrast characteristics, including osmolarity, ionicity, molecular structure, and viscosity
The single most important patient-related risk factor is preexisting CKD, even more so than diabetes mellitus.[15] Patients with CKD in the setting of diabetes mellitus have a 4-fold increase in the risk of CIN compared with patients without diabetes mellitus or preexisting CKD.
Table: Physiochemical Properties of Contrast Media
Although the data is by no means uniform, they seem to suggest that the iso-osmolar contrast agent iodixanol may be associated with smaller increases in SCr and lower rates of CIN when compared with low-osmolar agents, especially in patients with CKD and in those with CKD and diabetes mellitus.[14] Guidelines from the American Heart Association (AHA)/American College of Cardiology (ACC) for the management of acute coronary syndromes patients with CKD recommend the use of IOCM (Class I, level of Evidence).
Table 1. Physiochemical Properties of Contrast Media[16] (Open Table in a new window)
| Class of Contrast Agent | Type of Contrast Agent | Iodine Dose(mg/mL) | Iodine/Particle Ratio | Viscosity(cPs at 37°C) | Osmolality(mOsm/kg H2 O) | Molecular Weight (Da) |
| High-osmolar monomers(ionic) | Diatrizoate (Renografin)Ioxithalamate (Telebrix) | 370350 | 1.51.5 | 2.32.5 | 18702130 | 636643 |
| Low-osmolar dimers(ionic) | Ioxaglate (Hexabrix) | 320 | 3 | 7.5 | 600 | 1270 |
| Low-osmolar monomers(nonionic) | Iohexol (Omnipaque)Iopamidol (Isovue)Iomeprol (Iomeron)
Ioversol (Optiray) Iopromide (Ultravist) Iopentol (Imagopaque) |
350370400
350 370 350 |
333
3 3 3 |
10.49.412.6
9 10 12 |
780790620
790 770 810 |
821777778
807 791 835 |
| Iso-osmolar dimers(nonionic) | Iodixanol (Visipaque)Iotrolan (Isovist) | 320320 | 66 | 11.88.5 | 290290 | 15501620 |
Epidemiology
Occurrence in the United States
CIN is the third leading cause of hospital-acquired renal failure. Decreased renal perfusion and surgery (or in some studies, nephrotoxic medications) are the number one and number two causes, respectively.
An analysis of 15 prospective and retrospective studies from 1976-1996 report an incidence of CIN of 3.1-31%. The number varies depending on the definition used for CIN; the contrast agent characteristics, including the type, amount, duration, and route of administration; preexisting risk factors; and length of follow-up (including the day of measurement of postcontrast serum creatinine).
In patients without risk factors, the incidence may be as low as 2%. With the introduction of risk factors, like diabetes, the number rises to 9%, with incidences being as high as 90% in diabetics with CKD. Therefore, the number and the type of preexisting risk factors directly influence the incidence of renal insufficiency. It is also procedure dependant, with 14.5% overall in patients undergoing coronary interventions compared to 1.6-2.3% for diagnostic intervention, as reported in literature.[17]
Race- and age-related demographics
While African Americans with diabetic nephropathy have a faster acceleration of end-stage renal disease (ESRD), independent of other variables, race has not been found to be a risk factor for CIN.
The incidence of CIN in patients older than age 60 years has been variously reported as 8-16%. It has also been shown that in patients with acute MI who have undergone coronary intervention, an age of 75 years or older is an independent risk factor for CIN.
Prognosis
CIN is normally a transient process, with renal functions reverting to normal within 7-14 days of contrast administration. Less than one-third patients develop some degree of residual renal impairment.
Dialysis is required in less than 1% of patients, with a slightly higher incidence in patients with underlying renal impairment (3.1%) and in those undergoing primary PCI for myocardial infarction (MI) (3%). However, in patients with diabetes and severe renal failure, the rate of dialysis can be as high as 12%.
Of the patients who need dialysis, 18% end up on permanent dialysis therapy. However, many of these patients will have had advanced renal insufficiency and concomitant diabetic nephropathy and will have been destined for dialysis regardless of the episode of CIN.
A growing body of knowledge indicates that acute kidney injury after contrast medium can be a harbinger of CKD or ESRD. In one observational study, the population studied appeared representative of the general population undergoing angiography and the rate of acute kidney ingury was consonant with other studies. The finding that persistent kidney damage can occur after contrast-induced acute kidney injury highlights the potential for acceleration of the progression of kidney injury in individuals with pre-existing CKD.[18]
Mortality
Patients who require dialysis have a considerably worse mortality rate, with reported rates of 35.7% inhospital mortality (compared with 7.1% in the nondialysis group) and a 2-year survival rate of only 19%.
CIN by itself may be an independent mortality risk factor. Following invasive cardiology procedures, patients with normal baseline renal function who develop CIN have reduced survival compared with patients with baseline chronic CKD who do not develop CIN.
Gadolinium-based agents
Gadolinium-based CM (used for magnetic resonance imaging [MRI]), when compared with iodine-based CM, have a similar, if not worse, adverse effect profile in patients with moderate CKD and eGFR of less than 30 mL/min. Their use has been implicated in the development of nephrogenic systemic fibrosis, a chronic debilitating condition with no cure.
A review of 3 series and 4 case reports suggested that the risk of renal insufficiency with gadolinium is similar to that of iodinated radiocontrast dye. The reported incidence varies from 4% in stage 3 CKD to 20% in stage 4 CKD. It may even be worse, as suggested by some investigators. A prospective study of 57 patients found that acute renal failure was seen in 28% of patients in the gadolinium group, compared with 6.5% of patients in the iodine group, despite prophylactic saline and N-acetylcysteine (NAC).
The risk factor profile is similar to that for iodinated CM; increased incidence of acute renal failure is seen in older patients and in those with lower baseline creatinine clearance, diabetic nephropathy, anemia, and hypoalbuminemia.
Risk stratification scoring systems
CIN is the result of a complex interplay of many of the above risk factors. The presence of 2 or more risk factors is additive, and the likelihood of CIN rises sharply as the number of risk factors increases. Researchers have tried to objectively quantify and predict the contribution of each risk factor to the ultimate outcome of CIN.
Risk stratification scoring systems have been devised to calculate an individual patient’s risk of developing CIN. This has mostly been done in patients undergoing percutaneous coronary intervention (PCI), especially those with preexisting risk factors. Mehran et al developed the following scoring system based on points awarded to each of 7 multivariate predictors[19] :
-
Hypotension = 5 points
-
IABP use = 5 points
-
CHF = 5 points
-
SCr of greater than 1.5 mg/dL = 4 points
-
Age greater than 75 years = 4 points
-
Anemia = 3 points
-
Diabetes mellitus = 3 points
-
Contrast volume = 1 point for each 100 cc used
Based on the total calculated score, patients were divided into low-risk (score of less than or equal to 5), moderate-risk (score of 6-10), high-risk (score of 11-15), and very–high-risk (score of greater than or equal to 16) categories. The rate of CIN and the requirement for dialysis were 7.5 and 0.04%, 14 and 0.12%, 26.1 and 1.09%, and 57.3 and 12.6%, respectively, for each of the 4 groups.
Bartholomew et al worked to create another scoring system and took into consideration 8 variables, including creatinine clearance of less than 60 mL/min, IABP use, urgent coronary procedure, diabetes mellitus, CHF, hypertension, peripheral vascular disease (PVD), and volume of contrast used.[20]
History and Physical Examination
History
Patients usually present with a history of contrast administration 24-48 hours prior to presentation, having undergone a diagnostic or therapeutic procedure (eg, PCI). The renal failure is usually nonoliguric.
Physical examination
A physical examination is useful for ruling out other causes of acute nephropathy, such as cholesterol emboli (eg, blue toe, livedo reticularis) or drug-induced interstitial nephritis (eg, rash). Patients may have evidence of volume depletion or may be in decompensated CHF.
Diagnostic Considerations
Conditions to consider in the differential diagnosis of CIN include the following:
-
Atheroembolic renal failure – More than 1 week after contrast, blue toes, livedo reticularis, transient eosinophilia, prolonged course, and lower recovery
-
Acute renal failure (includes prerenal and postrenal azotemia) – There may also be associated dehydration from aggressive diuresis, exacerbated by preexisting fluid depletion; the acute renal failure is usually oliguric, and recovery is anticipated in 2-3 weeks
-
Acute interstitial nephritis (triad of fever, skin rash, and eosinophilia) – Also eosinophiluria; the nephritis is usually from drugs such as penicillin, cephalosporins, and nonsteroidal anti-inflammatory drugs (NSAIDs)
-
Acute tubular necrosis – Ischemia from prerenal causes; endogenous toxins, such as hemoglobin, myoglobin, and light chains; exogenous toxins, such as antibiotics, chemotherapeutic agents, organic solvents, and heavy metals
Approach Considerations
SCr concentration usually begins to increase within 24 hours after contrast agent administration, peaks between days 3 and 5, and returns to baseline in 7-10 days. Serum cystatin C (which has been suggested as a surrogate marker of renal function in lieu of SCr) is increased in patients with CIN.
Nonspecific formed elements can appear in the urine, including renal tubular epithelial cells, pigmented granular casts, urate crystals, and debris. However, these urine findings do not correlate with severity.
Urine osmolality tends to be less than 350 mOsm/kg. The fractional excretion of sodium (FENa) may vary widely. In the minority of patients with oliguric CIN, the FENa is low in the early stages, despite no clinical evidence of volume depletion.
Histology
CM cause direct toxic effects on renal tubular epithelial cells, characterized by cell vacuolization, interstitial inflammation, and cellular necrosis. In a study, these characteristic changes, called osmotic nephrosis, were observed in 22.3% of patients undergoing renal biopsy, within 10 days of contrast exposure.[21]
Approach Considerations
Hydration therapy is the cornerstone of CIN prevention. Renal perfusion is decreased for up to 20 hours following contrast administration. Intravascular volume expansion maintains renal blood flow, preserves nitric oxide production, prevents medullary hypoxemia, and enhances contrast elimination.
However, a number of other CIN therapies have been investigated, including the use of statins, bicarbonate, N-acetylcysteine (NAC), ascorbic acid, the adenosine antagonists theophylline and aminophylline, vasodilators, forced diuresis, and renal replacement therapy. Patients with CIN should be managed in consultation with a nephrologist.
Hydration Therapy
The first study revealing the benefit of hydration in CIN prevention came from Solomon et al.[22] They also found forced diuresis to be inferior to hydration with 0.45% saline. Fluids with different compositions and tonicity have since been studied, including bicarbonate and mannitol.
Normal saline has been found to be superior to half-normal saline in terms of its enhanced ability in intravascular volume expansion. It also causes increased delivery of sodium to the distal nephron, prevents rennin-angiotensin activation, and thus maintains increased renal blood flow. In terms of route of administration, oral fluids, while beneficial, are not as effective as intravenous hydration.[23, 24]
The CIN Consensus Working Panel found that adequate intravenous volume expansion with isotonic crystalloids (1-1.5 mL/kg/h), 3-12 hours before the procedure and continued for 6-24 hours afterward, decreases the incidence of CIN in patients at risk. The panel studied 6 clinical trials with different protocols for volume expansion. The studies differed in the type of fluid used for hydration (isotonic vs half-normal saline), route, duration, timing, and amount of fluid used.[25]
For hospitalized patients, volume expansion should begin 6 hours prior to the procedure and be continued for 6-24 hours postprocedure. For outpatients, administration of fluids can be initiated 3 hours before and continued for 12 hours after the procedure. Postprocedure volume expansion is more important than preprocedure hydration. It has been suggested that a urine output of 150 mL/h should guide the rate of intravenous fluid replacement, although the CIN Consensus Working Panel did not find it useful to recommend a target urine output.
CHF poses a particular challenge. Patients with compensated CHF should still be given volume, albeit at lower rates. Uncompensated CHF patients should undergo hemodynamic monitoring, if possible, and diuretics should be continued. In emergency situations, one’s clinical judgment should be used, and, in the absence of any baseline renal function, adequate postprocedure hydration should be carried out.
What is interesting, however, is that, while hydration remains the cornerstone for CIN prevention, a randomized, controlled trial comparing a strategy of volume expansion with no volume expansion has not been performed to date.
Statins
Statins are widely used in coronary artery disease (CAD) for their pleiotropic effects (favorable effects on endothelin and thrombus formation, plaque stabilization, and anti-inflammatory properties), and it was believed that, given the vascular nature of CIN, they might have similar renoprotective effects. The data for statin use, however, are retrospective and anecdotal; they are taken mostly from patients already on statins who underwent PCI.[26]
A significantly lower incidence of CIN was found in patients treated with statins preoperatively (CIN incidence of 4.37% in the statin group vs 5.93% in the nonstatin group). However, prospective trials looking at statin use in patients undergoing noncardiac procedures are needed to better qualify this initial promise.
Bicarbonate Therapy
Bicarbonate therapy alkalinizes the renal tubular fluid and, thus, prevents free radical injury. Hydrogen peroxide and an oxygen ion (from superoxide) react to form a hydroxide ion, all agents of free radical injury. This reaction, called the Harber-Weiss reaction, is activated in an acidic environment. Bicarbonate, by alkalinizing the environment, slows down the reaction. It also scavenges reactive oxygen species (ROS) from NO, such as peroxynitrite.
Bicarbonate protocols most often include infusion of sodium bicarbonate at the rate of 3 mL/kg/hour an hour before the procedure, continued at 1 mL/kg/hour for 6 hours after. Some investigators have used 1 mL/kg/hour for 24 hours, starting 12 hours before the procedure. The exact duration, however, remains a matter of debate. Hydration with sodium bicarbonate has been found by some researchers to be more protective than normal saline alone.
Treatment controversy
A 2008 retrospective cohort study at the Mayo Clinic assessed the risk of CIN associated with the use of sodium bicarbonate, NAC, and the combination of sodium bicarbonate with NAC and found that, compared with no treatment, sodium bicarbonate used alone was associated with an increased risk of CIN. NAC alone or in combination with sodium bicarbonate did not significantly affect the incidence of CIN. The results were obtained after adjusting for confounding factors, including total volume of hydration, medications, baseline creatinine, and contrast iodine load.[27] Given the above new information, it is recommended that the use of sodium bicarbonate to prevent CIN should be further evaluated.
N-acetylcysteine
NAC is acetylated L-cysteine, an amino acid. Its sulfhydryl groups make it an excellent antioxidant and scavenger of free oxygen radicals. It also enhances the vasodilatory properties of nitric oxide. Twelve meta-analyses covering 29 randomized, controlled trials have been published on the effect of NAC therapy in CIN. They all suffer from significant heterogeneity. The standard oral NAC regimen consists of 600 mg twice daily for 24 hours before and on the day of the procedure. Higher doses of 1 g, 1200 mg, and 1500 mg twice daily have also been studied, with no significant dose-related or route-related (oral vs intravenous) difference. NAC has very low oral bioavailability; substantial interpatient variability and inconsistency between the available oral products obscure the picture further.[3, 24, 28]
Treatment controversy
The latest controversy relating to NAC therapy questioned the parameter on which its effectiveness was based. It was suggested that the beneficial effect of NAC in CIN is related to its SCr-lowering ability rather than to improved GFR. It was believed that NAC directly reduces SCr by increasing SCr’s excretion (tubular secretion), decreasing its production (augments activity of creatine kinase), or interfering with its laboratory measurement, enzymatic or nonenzymatic (Jaffe method).
This was supported by a study that demonstrated a significant decrease in SCr after 4 doses of 600 mg of oral NAC in healthy volunteers with normal kidney function and no exposure to radiocontrast media.[29] This would bring doubt into the results of at least 13 randomized, controlled trials that showed NAC to be protective in CIN, with SCr used as the endpoint. However, Haase et al compared the effect of NAC on SCr by simultaneously studying its effect on cystatin C and found that NAC did not artifactually lower SCr when measured by the Jaffe method.[30]
The CIN Working Panel concluded that the existing data on NAC therapy in CIN is sufficiently varied to preclude a definite recommendation.[25] In the practice of medicine, though, it remains part of the standard of care and is routinely administered because of its low cost, lack of adverse effects, and potential beneficial effect, as demonstrated by the relative risk reduction of CIN, ranging from 0.37-0.73, as reported in several meta-analyses.
Renal Replacement Therapy
Less than 1% of patients with CIN ultimately go on to require dialysis, the number being slightly higher in patients with underlying renal impairment (3.1%) and in those undergoing primary PCI for MI (3%). However, in patients with diabetes and severe renal failure, the rate of dialysis can be as high as 12%. Patients who get dialyzed do considerably worse, with inhospital mortality rates of 35.7% (compared with 7.1% in the nondialysis group) and a 2-year survival rate of only 19%.
CM have molecular weights that range between 650 and 1600 mOsm/kg. They have low lipophilicity, low plasma protein binding, and minimal biotransformation. They quickly equilibrate across capillary membranes and have volumes of distribution equivalent to that of the extracellular fluid volume. In patients with normal renal function, CM are excreted with the first glomerular passage and the decrease in their plasma concentration follows a 2-part exponential function, a distribution phase and an elimination phase. However, in patients with renal impairment, the renal clearance values are reduced. For example, 50% of the low-osmolarity contrast agent iomeprol is eliminated within 2 hours in healthy subjects, compared with 16-84 hours in patients with severe renal impairment.
In patients already on dialysis, the commonly sited issues with contrast administration include volume load and direct toxicity of contrast to the remaining nonfunctional nephrons and nonrenal tissues. Thus, the perceived need for emergent dialysis and contrast removal.
Rodby attempted to address these concerns, calculating that the administration of 100 mL of hyperosmolar contrast would move 265 mL of water from the intracellular to the extracellular compartment, resulting in an increase in extracellular volume by 365 mL. The increase in intravascular space would therefore be only a third, or 120 mL. Fluid shifts with LOCM are even less. He also found that extrarenal toxicity of CM was cited in mostly single case reports, and no objective evidence could be identified in 3 prospective studies.[31]
The risk of acute damage from contrast is therefore greatest in patients with CKD. This can be explained by the increase in single nephron GFR and, thus, the filtered load of contrast per nephron. This is akin to a double hit to the remaining nephrons; increased contrast load and prolonged tubular exposure. While this may not seem to be a concern in patients with ESRD who are already on dialysis, residual renal function, in fact, plays a big role in their outcome, more so in patients on peritoneal dialysis. Its preservation is therefore important.[31]
CM can be effectively and efficiently removed by hemodialysis (HD). Factors that influence CM removal include blood flow, membrane surface area, molecular size, transmembrane pressure, and dialysis time. High-flux dialysis membranes with blood flows of between 120-200 mL/min can remove almost 50% of iodinated CM within an hour and 80% in 4 hours. Even in patients with CKD, in whom contrast excretion is delayed, it was found that 70-80% of contrast can be removed by a 4-hour HD treatment. In view of the limited benefit of therapies such as hydration, bicarbonate and NAC, dialysis may seem like the definitive answer.
However, an excellent meta-analysis by Cruz et al—8 trials (6 randomized and 2 nonrandomized, controlled studies) were included in the analysis, with a pooled sample size of 412 patients—indicated that periprocedural extracorporeal blood purification (ECBP) does not significantly reduce the incidence of CIN in comparison with standard medical therapy. ECBP in the study consisted of HD (6 trials), continuous venovenous hemofiltration (1 trial), and continuous venovenous hemodiafiltration (1 trial).[32]
Cruz et al found that the incidence of CIN in the standard medical therapy group was 35.2%, compared with 27.8% in the ECBP group. Renal death (combined endpoint of death or dialysis dependence) was 12.5% in the standard medical therapy group, compared with 7.9% in the ECBP group.
An important consideration is the role of ECBP therapy in patients with severe renal impairment (ie, stage 5 CKD) not yet on maintenance dialysis. A study by Lee et al indicated that in patients with chronic renal failure who are undergoing coronary angiography, prophylactic HD can improve renal outcome. The study included 82 patients with stage 5 CKD who were not on dialysis and who were referred for coronary angiography.[33] The patients were randomly assigned to either undergo prophylactic HD (initiated within 81 ± 32 min) or to receive intravenous normal saline (control group).
The baseline creatinine of the dialysis group was 13.2 mL/min/1.73 m2, comparable to that of the control group (12.6 mL/min/1.73 m2). The investigators’ primary endpoint was change in creatinine clearance in the 2 groups on day 4, which was found to be statistically significant (0.4 ± 0.9 mL/min/1.73 m2 in the dialysis group vs 2.2 ± 2.8 mL/min/1.73 m2 in the control group).
Lee et al found that 35% of the control group required temporary renal replacement therapy, compared with 2% of the dialysis group. In addition, long-term, postdischarge dialysis was required in 13% of the control patients but in none of the dialysis patients. Among those patients who did not require chronic dialysis, an increase in SCr at discharge of over 1 mg/dL from baseline was found in 13 patients in the control group and in 2 patients in the dialysis group.
The study, though hopeful, does raise some concerns. While the change in creatinine clearance on day 4 from baseline was statistically significant, the day 4 creatinine clearance itself was not significantly different between the 2 groups. Also, the results were not expressed as CIN incidence. This patient population is very fragile and is already on the verge of dialysis. How much time off dialysis a single HD session was able to buy these patients was not discussed. The duration of follow-up was also not clear.
Marenzi et al found better outcomes in patients who received venovenous hemofiltration both pre- and post-CM administration than in patients who received post-CM hemofiltration or no hemofiltration at all. These outcomes included a lower likelihood of CIN, no need for HD, and no 1-year mortality, in the pre-/post-CM group.[34]
The biggest confounder in studies of continuous renal replacement therapy (CRRT) is that the outcome measure (SCr) is affected by the treatment itself. While the advantage of CRRT is the lack of delay in its institution, contrast clearance rates would be 1 L/h (16.6 mL/min provided a maximal sieving coefficient for contrast across the hemofiltration membrane of 1), substantially less than standard HD.
Furthermore, continuous venovenous hemofiltration is expensive, highly invasive, and requires trained personnel; the procedure itself needs to be performed in the intensive care unit (ICU). In the face of equivocal benefit of a highly invasive and expensive procedure, the role of continuous venovenous hemofiltration has yet to be accepted as a prophylactic treatment for avoiding CIN.
Dialysis immediately after contrast administration has been suggested for patients already on long-term HD and for those at very high risk of CIN. Three studies looked at its necessity and found that LOCM can be given safely to patients with ESRD who are being maintained on HD without the added expense or inconvenience of emergent postprocedural HD.
The only condition in which HD might be argued to have a beneficial role is in patients on peritoneal dialysis who rely on their residual renal function. In this setting, HD performed soon after CM administration may provide enhanced removal and therefore protect residual renal function. It should be noted, however, that these patients on peritoneal dialysis would therefore need an additional HD procedure with concomitant vascular access, as the clearance with peritoneal dialysis would be far too slow to offer any protection.
In a study to determine if renal replacement therapy in concert with contrast administration helps, Frank et al found that although the overall clearance of contrast was significantly increased by dialysis, the peak plasma concentration of iomeprol 15 minutes after contrast administration was not significantly changed by simultaneous dialysis. In their report, the investigators prospectively studied 17 patients with chronic renal insufficiency (SCr >3 mg/dL), dialysis independent, who were then randomized to receive high-flux HD over 6 hours simultaneously with contrast administration, and[35]
In the study, Frank et al also found that to be clinically effective, simultaneous dialysis should reduce the risk of developing ESRD by 50%. If type 1 and type 2 errors are set at 0.01, the result could be accepted only if none of the 48 sequential patients with simultaneous dialysis required dialysis during the 8 weeks after contrast exposure. To reject the hypothesis, 239 sequential patients with simultaneous dialysis would have to be included. Therefore, most CIN studies, are seriously underpowered.
Studies of HD for CIN vary with respect to the definition of CIN used, the patient population, the type and volume of CM, how long after CM administration HD is started, and, finally, the dialysis treatment modality itself. While existing studies do not show HD to be superior to hydration alone for CIN prevention, if HD is used in conjunction with hydration and CIN protective therapy, such as NAC and bicarbonate, it might prove to be efficacious in some high-risk patients. However, most studies have had only an 8-week follow-up period. While the initiation of long-term dialysis was 5-15%, the progression to uremia over a long-term follow-up period is still unanswered.[16]
Other Therapies
Ascorbic acid, which has antioxidant properties, was studied for its ability to counter the effect of free radicals and reactive oxygen species. One study found that oral ascorbic acid administered in a 3-g dose preprocedure and two 2-g doses postprocedure was associated with a 62% risk reduction in CIN incidence.[36]
Theophylline and aminophylline are adenosine antagonists that counteract the intrarenal vasoconstrictor and tubuloglomerular feedback effects of adenosine. They have been found to have a statistically significant effect in preventing CIN in high-risk patients. However, their use is limited by their narrow therapeutic window and adverse effects profile.
Vasodilators, such as calcium channel blockers, dopamine/fenoldopam, atrial natriuretic peptide, and L-arginine, all with different mechanisms of action, have a favorable effect on renal hemodynamics. However, their use for CIN prevention has not been borne out by most controlled trials, and they are not routinely recommended at this point.
Forced diuresis with furosemide and mannitol was studied in the hope that this procedure would dilute CM within the tubular lumen and enhance their excretion. Furosemide and mannitol in fact worsen CIN by causing dehydration in patients who may already have intravascular volume depletion. Their use at this time is discouraged.
Deterrence and Prevention
The best therapy for CIN is prevention. Physicians need to be increasingly aware that CIN is a common and potentially serious complication. Patients at risk should be identified early, especially those with CKD (ie, eGFR < 60 mL/min/1.73 m2). A detailed history inquiring for risk factors, especially diabetes mellitus, should be ascertained.
In patients with risk factors for CIN, the possibility of alternative imaging studies that do not need contrast should be explored. MRI with gadolinium is no longer considered a safe alternative to contrast because of the risk of nephrogenic systemic fibrosis, an irreversible, debilitating condition seen mostly in patients with an eGFR of less than 30 mL/min/1.73 m2.
In patients with a moderate to severe risk of CIN, creatinine clearance rates or eGFR should be estimated by either the MDRD formula or the Cockroft-Gault formula and then measured again 24-48 hours after contrast administration.
In the emergency setting, where the benefit of very early imaging studies outweighs that of waiting, the imaging procedure can be carried out without an initial estimation of SCr or eGFR.
Intra-arterial administration of iodinated CM poses a greater risk for CIN than does the intravenous approach. For patients at an increased risk for CIN receiving intra-arterial contrast, nonionic iso-osmolar agents (iodixanol) are associated with the lowest risk of CIN.
The amount of contrast used during the procedure should be limited to as little as possible and kept under 100 mL. Most investigators have found this to be the cut-off value below which no patient needed dialysis. The risk of CIN increases by 12% for each 100 mL of contrast used beyond the first 100 mL. Most angiographic diagnostic studies usually require 100 mL of contrast, compared with 200-250 mL for angioplasty. The maximum amount of contrast that can be used safely should be individualized, taking into account the preexisting renal function.
Various formulas for calculating the maximal safe CM dose have been suggested. Two most often cited are those suggested by Cigarroa et al and the European Society of Urogenital Radiology (ESUR).[37, 38] Cigarroa et al, in a retrospective study of 115 patients undergoing cardiac catheterization and angiography, using the HOCM diatrizoate, suggested that the dose of CM should not exceed 5 mL/kg of body weight (maximum 300 mL divided by SCr [mg/dL]). The ESUR, in turn, has published maximal LOCM volumes for various SCr cut-off values.
While the formulas from Cigarroa and the ESUR take into account the SCr, it has been suggested that the eGFR (a more accurate predictor of renal function) and the iodine dose of CM should be reflected in any estimates or predictions of safe CM dosages. There exists, however, no unimpeachably safe CM dose algorithm for CIN prevention.
The length of time between 2 contrast procedures should be at least 48-72 hours. Rapid repetition of contrast administration has been found to be a univariate risk factor for CIN.
Potentially nephrotoxic drugs (eg, NSAIDs, aminoglycosides, amphotericin B, cyclosporin, tacrolimus) should be withdrawn at least 24 hours prior, in patients at risk (eGFR < 60 mL/min).
Metformin, though not nephrotoxic, should be used prudently, because if renal failure does occur, there is risk of concomitant lactic acidosis. Therefore, metformin should be stopped at the time of the procedure and resumed 48 hours later if renal function remains normal.
Angiotensin-converting enzyme (ACE) inhibitors and angiotensin receptor blockers (ARBs) cause a 10-15% rise in SCr by reducing intraglomerular pressure. While they should not be started at this time, whether they should be discontinued remains a matter of debate. Much of the literature in this area is unclear and controversial.
Minimizing contrast administration
The amount of contrast used during the procedure should be limited to as little as possible and kept under 100 mL. Most investigators have found this to be the cut-off value below which no patient needed dialysis. The risk of CIN increases by 12% for each 100 mL of contrast used beyond the first 100 mL. Most angiographic diagnostic studies usually require 100 mL of contrast, compared with 200-250 mL for angioplasty. The maximum amount of contrast that can be used safely should be individualized, taking into account the preexisting renal function.
Various formulas for calculating the maximal safe CM dose have been suggested. Two most often cited are those suggested by Cigarroa et al and the European Society of Urogenital Radiology (ESUR).[37, 38] Cigarroa et al, in a retrospective study of 115 patients undergoing cardiac catheterization and angiography, using the HOCM diatrizoate, suggested that the dose of CM should not exceed 5 mL/kg of body weight (maximum 300 mL divided by SCr [mg/dL]). The ESUR, in turn, has published maximal LOCM volumes for various SCr cut-off values.
While the formulas from Cigarroa and the ESUR take into account the SCr, it has been suggested that the eGFR (a more accurate predictor of renal function) and the iodine dose of CM should be reflected in any estimates or predictions of safe CM dosages. There exists, however, no unimpeachably safe CM dose algorithm for CIN prevention.
RAAS blockade
A prospective, 50-month Mayo study found renin-angiotensin-aldosterone system (RAAS) blockade, particularly in older patients with CHD, exacerbates CIN (43% incidence of dialysis and 29% progression to ESRD).[39] The marker used for renal function was eGFR, as calculated by the MDRD formula. The study recommended that RAAS blockade be withheld 48 hours prior to contrast exposure.
RAAS blockage, however, can improve renal perfusion and decrease proximal tubular reabsorption, including CM absorption by the tubular cells. This effect can be documented with the increase in the fractional excretion of urea seen with low-dose RAAS therapy in patients with CHF and moderate CKD (the majority of the CIN-susceptible population).[40] In this group, reduction in intraglomerular pressure and filtration fraction from RAAS therapy might decrease tubular CM concentration and therefore lessen its adverse effects.
Medication Summary
NAC is acetylated L-cysteine, an amino acid. As previously mentioned, its sulfhydryl groups make it an excellent antioxidant and scavenger of free oxygen radicals. It also enhances the vasodilatory properties of nitric oxide. Twelve meta-analyses covering 29 randomized, controlled trials have been published on the effect of NAC therapy in CIN. They all suffer from significant heterogeneity. The standard oral NAC regimen consists of 600 mg twice daily for 24 hours before and on the day of the procedure. Higher doses of 1 g, 1200 mg, and 1500 mg twice daily have also been studied, with no significant dose-related or route-related (oral vs intravenous) difference. NAC has very low oral bioavailability; substantial interpatient variability and inconsistency between the available oral products obscure the picture further.[24, 28]
Antidote, Acetaminophen
Class Summary
Used for prevention of contrast toxicity.
N-acetylcysteine (Acetadote)
Used for prevention of contrast toxicity in susceptible individuals such as those with diabetes mellitus. May provide substrate for conjugation with toxic metabolites.
Antilipemic Agents
Class Summary
These agents are used for their favorable effects on endothelin and thrombus formation, plaque stabilization and anti-inflammatory properties by improving lipid profile.
Simvastatin (Zocor)
Indicated for hyperlipoproteinemia (Type III). Inhibit 3-hydroxy-3-methylglutaryl coenzyme A (HMG-CoA reductase), which in turn inhibit cholesterol synthesis, and increases cholesterol metabolism. Increase HDL cholesterol and decrease LDL-C, total-C, apolipoprotein B, VLDL cholesterol, and plasma triglycerides.
Atorvastatin (Lipitor)
The most efficacious of the statins at high doses. Inhibits 3-hydroxy-3-methylglutaryl coenzyme A (HMG-CoA reductase), which in turn inhibits cholesterol synthesis and increases cholesterol metabolism. Reports have shown as much as a 60% reduction in LDL-C. The Atorvastatin versus Revascularization Treatment study (AVERT) compared 80 mg atorvastatin daily to standard therapy and angioplasty in patients with CHD. While events at 18 mo were the same between both groups, the length of time until the first CHD event occurred was longer with aggressive LDL-C lowering. The half-life of atorvastatin and its active metabolites is longer than that of all the other statins (ie, approximately 48 h compared to 3-4 h).
May modestly elevate HDL-C levels. Clinically, reduced levels of circulating total cholesterol, LDL-C, and serum TGs are observed.
Before initiating therapy, patients should be placed on a cholesterol-lowering diet for 3-6 mo; the diet should be continued indefinitely.
Lovastatin (Mevacor, Altoprev)
Adjunct to dietary therapy in reducing serum cholesterol. Immediate-release (Mevacor) and extended-release (Altocor) are available.
Fluvastatin (Lescol, Lescol XL)
Synthetically prepared HMG-CoA reductase inhibitor with some similarities to lovastatin, simvastatin, and pravastatin. However, structurally distinct and has different biopharmaceutical profile (eg, no active metabolites, extensive protein binding, minimal CSF penetration).
Used as an adjunct to dietary therapy in decreasing cholesterol levels.
Pravastatin (Pravachol)
Effective in reducing circulating lipid levels and improving the clinical and anatomic course of atherosclerosis.
Rosuvastatin (Crestor)
HMG-CoA reductase inhibitor that in turn decreases cholesterol synthesis and increases cholesterol metabolism. Reduces total-C, LDL-C, and TG levels and increases HDL-C level. Used adjunctively with diet and exercise to treat hypercholesterolemia.
SOURCE