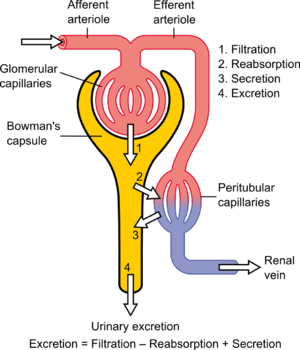
The Cardiorenal Syndrome in Heart Failure: Cardiac? Renal? syndrome?
Writer and Curator: Larry H. Bernstein, MD, FCAP
and
Curator: Aviva Lev-Ari, PhD, RN
Triposkiadis F, Starling RC, Boudoulas H, Giamouzis G, Butler J.
Heart Fail Rev. 2012 May;17(3):355-66. http://dx.doi.org/10.1007/s10741-011-9291-x Review
There has been increasing interest on the so-called cardiorenal syndrome (CRS), defined as
- a complex pathophysiological disorder of the heart and kidneys where by acute or chronic dysfunction in one organ may induce acute or chronic dysfunction in the other.
In this review, we contend that there is lack of evidence warranting the adoption of a specific clinical construct such as the CRS within the heart failure (HF) syndrome by demonstrating that:
(a) the approaches and tools regarding the definition of kidney involvement in HF are suboptimal;
(b) development of renal failure in HF is often confounded by age, hypertension, and diabetes;
(c) worsening of renal function (WRF) in HF may be largely independent of alterations in cardiac function;
(d) the bidirectional association between HF and renal failure is not unique and represents one of the several such associations encountered in HF; and
(e) inflammation is a common denominator for HF and associated noncardiac morbidities.
Based on these arguments, we believe that
- dissecting one of the multiple bidirectional associations in HF and
- constructing the so-called cardiorenal syndrome is not justified pathophysiologically.
Fully understanding of all morbid associations and not only the cardiorenal, that is of great significance for the clinician who is caring for the patient with HF.
Ultrafiltration in Heart Failure with Cardiorenal Syndrome
N Engl J Med 2013; 368:1157-1160 http://dx.doi.org/10.1056/NEJMc1300456
Bart et al. (Dec. 13 issue)1 report the results of the Cardiorenal Rescue Study in Acute Decompensated Heart Failure (CARRESS-HF). They state that ultrafiltration was inferior to a strategy of stepped pharmacologic therapy with respect to the
- bivariate primary end point of the change in the serum creatinine level and body weight in patients with acute decompensated heart failure.
It is unclear at first sight why renal function should be different at 96 hours only when serum creatinine concentrations are used as a marker of renal function,
- but not when the level of cystatin C or the glomerular filtration rate are used.
How can this discrepancy be explained?
According to the study Ultrafiltration in Decompensated Heart Failure with Cardiorenal Syndrome
Bart BA., Goldsmith SR., Lee KL, Givertz MM, et al.
N Engl J Med 2012; 367:2296-2304 http://dx.doi.org/10.1056/NEJMoa1210357
Ultrafiltration is an alternative strategy to diuretic therapy for the treatment of patients with acute decompensated heart failure.
Little is known about the efficacy and safety of ultrafiltration in patients with acute decompensated heart failure
- complicated by persistent congestion and worsened renal function.
Ultrafiltration was inferior to pharmacologic therapy with respect to the bivariate end point of
- the change in the serum creatinine level and body weight 96 hours after enrollment (P=0.003),
- owing primarily to an increase in the creatinine level in the ultrafiltration group.
- At 96 hours, the mean change in the creatinine level was −0.04±0.53 mg per deciliter (−3.5±46.9 μmol per liter) in the pharmacologic-therapy group,
- as compared with +0.23±0.70 mg per deciliter (20.3±61.9 μmol per liter) in the ultrafiltration group (P=0.003).
A higher percentage of patients in the ultrafiltration group than in the pharmacologic-therapy group had a serious adverse event (72% vs. 57%, P=0.03).
In a randomized trial involving patients hospitalized for acute decompensated heart failure,
- worsened renal function, and
- persistent congestion,
the use of a stepped pharmacologic-therapy algorithm was superior to a strategy of ultrafiltration for
- the preservation of renal function at 96 hours,
- with a similar amount of weight loss with the two approaches.
Related articles
- Advanced heart failure (pharmaceuticalintelligence.com)
- Aggressive Fluid and Sodium Restriction in Acute Decompensated Heart Failure. (zedie.wordpress.com)
- First drug to improve heart failure mortality in over a decade – HealthCanal.com (pharmaceuticalintelligence.com)
- Beta-Blocker Response (23andme.com)
- Phrenic Nerve Stimulation in Patients with Cheyne-Stokes Respiration and Congestive Heart Failure (pharmaceuticalintelligence.com)
- Heart-failure admissions and mortality highest in winter and on weekends – TheHeart.Org (pharmaceuticalintelligence.com)
- Acute Dialysis Quality Initiative (ADQI) (scicombinator.com)
- FDA grants Breakthrough Therapy designation to Novartis’ serelaxin (RLX030) for acute heart failure. (zedie.wordpress.com)
Other related articles published on this Open Access Online Scientific Journal
Acute and Chronic Myocardial Infarction: Quantification of Myocardial Perfusion Viability – FDG-PET/MRI vs. MRI or PET alone (Justin Pearlman, (Aviva Lev-Ari)
http://pharmaceuticalintelligence.com/2013/05/22/acute-and-chronic-myocardial-infarction-quantification-of-myocardial-viability-fdg-petmri-vs-mri-or-pet-alone/
Accurate Identification and Treatment of Emergent Cardiac Events (larryhbern)
http://pharmaceuticalintelligence.com/2013/03/15/accurate-identification-and-treatment-of-emergent-cardiac-events/
Nitric Oxide and it’s Impact on Cardiothoracic Surgery (tildabarliya)
http://pharmaceuticalintelligence.com/2012/12/15/nitric-oxide-and-its-impact-on-cardiothoracic-surgery/
CABG or PCI: Patients with Diabetes – CABG Rein Supreme (Aviva Lev-Ari)
http://pharmaceuticalintelligence.com/2012/11/05/cabg-or-pci-patients-with-diabetes-cabg-rein-supreme/
Endothelin Receptors in Cardiovascular Diseases: The Role of eNOS Stimulation (Aviva Lev-Ari)
http://pharmaceuticalintelligence.com/2012/10/04/endothelin-receptors-in-cardiovascular-diseases-the-role-of-enos-stimulation/
Critical Care | Abstract | Cardiac ischemia in patients with septic … (Aviva Lev-Ari)
http://pharmaceuticalintelligence.com/2013/06/26/critical-care-abstract-cardiac-ischemia-in-patients-with-septic/
Dealing with the Use of the High Sensitivity Troponin (hs cTn) Assays (larryhbern)
http://pharmaceuticalintelligence.com/2013/05/18/dealing-with-the-use-of-the-hs-ctn-assays/
Acute Chest Pain/ER Admission: Three Emerging Alternatives to Angiography and PCI – Corus CAD, hs cTn, CCTA (Aviva Lev-ARi)
http://pharmaceuticalintelligence.com/2013/03/10/acute-chest-painer-admission-three-emerging-alternatives-to-angiography-and-pci/
Assessing Cardiovascular Disease with Biomarkers (larryhbern)
http://pharmaceuticalintelligence.com/2012/12/25/assessing-cardiovascular-disease-with-biomarkers/
Heart Failure Treatment Improves, But Death Rate Remains High : NPR (Aviva Lev-Ari)
http://pharmaceuticalintelligence.com/2013/05/29/heart-failure-treatment-improves-but-death-rate-remains-high-npr/
Economic Toll of Heart Failure in the US: Forecasting the Impact of Heart Failure in the United States – A Policy Statement From the American Heart Association (Aviva Lev-Ari)
http://pharmaceuticalintelligence.com/2013/04/25/economic-toll-of-heart-failure-in-the-us-forecasting-the-impact-of-heart-failure-in-the-united-states-a-policy-statement-from-the-american-heart-association/
Stenosis, ischemia and heart failure (Aviva Lev-Ari)
http://pharmaceuticalintelligence.com/2013/05/16/stenosis-ischemia-and-heart-failure/
Congestive Heart Failure & Personalized Medicine: Two-gene Test predicts response to Beta Blocker Bucindolol (Aviva Lev-Ari)
http://pharmaceuticalintelligence.com/2012/10/17/chronic-heart-failure-personalized-medicine-two-gene-test-predicts-response-to-beta-blocker-bucindolol/
Heart Remodeling by Design – Implantable Synchronized Cardiac Assist Device: Abiomed’s Symphony (Aviva lev-Ari)
http://pharmaceuticalintelligence.com/2012/07/23/heart-remodeling-by-design-implantable-synchronized-cardiac-assist-device-abiomeds-symphony/
Long-Term Mortality in Treated Hypertensive Patients: Serum Uric Acid Level, Longitudinal Blood Pressure and Renal Function
Aviva Lev-Ari, PhD, RN
Renal Sympathetic Denervation: Updates on the State of Medicine
Aviva Lev-Ari, PhD,RN
Chapter 8: Nitric Oxide and Kidney Dysfunction
8.1 Part I: The Amazing Structure and Adaptive Functioning of the Kidneys: Nitric Oxide
Larry H. Bernstein, MD, FCAP
8.2 Part II: Nitric Oxide and iNOS have Key Roles in Kidney Diseases
Larry H. Bernstein, MD, FCAP
8.3 Part III: The Molecular Biology of Renal Disorders: Nitric Oxide
Larry H. Bernstein, MD, FCAP
8.4 Part IV: New Insights on Nitric Oxide Donors
Larry H. Bernstein, MD, FCAP
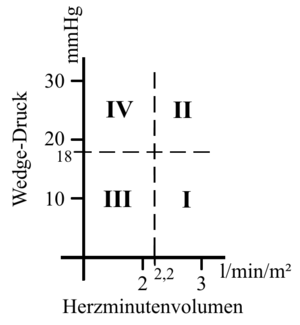
Forrester-classification for classification of Congestive heart failure ; Forrester-Klassifikation zur Einteilung einer akuten Herzinsuffizienz (Photo credit: Wikipedia)