Heart and Aging Research in Genomic Epidemiology: 1700 MIs and 2300 coronary heart disease events among about 29 000 eligible patients: Design of Prospective Meta-Analyses of Genome-Wide Association Studies From 5 Cohorts
Reporter: Aviva Lev-Ari, PhD, RN
Cohorts for Heart and Aging Research in Genomic Epidemiology (CHARGE) Consortium
Heart and Aging Research in Genomic Epidemiology: 1700 MIs and 2300 coronary heart disease events among about 29 000 eligible patients: Design of Prospective Meta-Analyses of Genome-Wide Association Studies From 5 Cohorts
Bruce M. Psaty, MD, PhD, Christopher J. O’Donnell, MD, MPH, Vilmundur Gudnason, MD, PhD, Kathryn L. Lunetta, PhD, Aaron R. Folsom, MD, Jerome I. Rotter, MD,André G. Uitterlinden, PhD, Tamara B. Harris, MD, Jacqueline C.M. Witteman, PhD,Eric Boerwinkle, PhD and on Behalf of the CHARGE Consortium
Author Affiliations
From the Cardiovascular Health Research Unit, Departments of Medicine, Epidemiology, and Health Services (B.M.P.), University of Wash; Center for Health Studies, Group Health (B.M.P.), Seattle, Wash; the National Heart, Lung and Blood Institute and the Framingham Heart Study (C.J.O.D.), Framingham, Mass; Icelandic Heart Association and the Department of Cardiovascular Genetics (Y.G.), University of Iceland, Reykjavik, Iceland; Department of Biostatistics (K.L.), Boston University School of Public Health, Mass; Division of Epidemiology and Community Health (A.R.F.), University of Minnesota, Minneapolis; Medical Genetics Institute (J.I.R.), Cedars-Sinai Medical Center, Los Angeles, Calif; Departments of Internal Medicine (A.G.U.) and Epidemiology (A.G.U., J.C.M.W.), Erasmus Medical Center, Rotterdam, The Netherlands; Laboratory of Epidemiology, Demography, and Biometry (T.B.H.), Intramural Research Program, National Institute on Aging, Bethesda, Md; and Human Genetics Center and Division of Epidemiology (E.B.), University of Texas, Houston.
Guest editor for this article was Elizabeth R. Hauser, PhD.
Abstract
Background— The primary aim of genome-wide association studies is to identify novel genetic loci associated with interindividual variation in the levels of risk factors, the degree of subclinical disease, or the risk of clinical disease. The requirement for large sample sizes and the importance of replication have served as powerful incentives for scientific collaboration.
Methods— The Cohorts for Heart and Aging Research in Genomic Epidemiology Consortium was formed to facilitate genome-wide association studies meta-analyses and replication opportunities among multiple large population-based cohort studies, which collect data in a standardized fashion and represent the preferred method for estimating disease incidence. The design of the Cohorts for Heart and Aging Research in Genomic Epidemiology Consortium includes 5 prospective cohort studies from the United States and Europe: the Age, Gene/Environment Susceptibility—Reykjavik Study, the Atherosclerosis Risk in Communities Study, the Cardiovascular Health Study, the Framingham Heart Study, and the Rotterdam Study. With genome-wide data on a total of about 38 000 individuals, these cohort studies have a large number of health-related phenotypes measured in similar ways. For each harmonized trait, within-cohort genome-wide association study analyses are combined by meta-analysis. A prospective meta-analysis of data from all 5 cohorts, with a properly selected level of genome-wide statistical significance, is a powerful approach to finding genuine phenotypic associations with novel genetic loci.
Conclusions— The Cohorts for Heart and Aging Research in Genomic Epidemiology Consortium and collaborating non-member studies or consortia provide an excellent framework for the identification of the genetic determinants of risk factors, subclinical-disease measures, and clinical events.
Example of Coronary Heart Disease
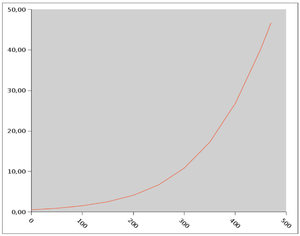
The cohort-study methods papers provide detail about many of the phenotypes listed in Table 2. For coronary heart disease, investigators knowledgeable about the phenotype in each study decided to focus on fatal and nonfatal myocardial infarction (MI) as the primary outcome because the MI criteria differed in only trivial ways among the studies. There were some minor differences in the definition of the composite outcome of MI, fatal coronary heart disease, and sudden death, which became the secondary outcome. Only subjects at risk for an incident event were included in the analysis. MI survivors whose DNA was drawn after the event were not eligible. The primary analysis was restricted to Europeans or European Americans. Patients entered the analysis at the time of the DNA blood draw, and were followed until an event, death, loss to follow up, or the last visit. The main recommendations of the Analysis Committee were adopted, and a threshold of 5×10−8 was selected for genome-wide statistical significance. Analyses in progress include about 1700 MIs and 2300 coronary heart disease events among about 29 000 eligible patients. Each cohort conducted its own analysis, and results were uploaded to a secure share site for the fixed-effects meta-analysis. Even with this number of events (Supplemental Figure 2), power is good for only for relatively high minor allele frequencies (>0.25) and large relative risks (>1.3).
The authors had full access to and take full responsibility for the integrity of the data. All authors have read and agree to the manuscript as written.
Discussion
In thousands of published papers, the 5 CHARGE cohort studies and many of the collaborating studies have already characterized the risk factors for and the incidence and prognosis of a variety of aging-related and cardiovascular conditions. The analysis of the incident MI, for instance, is free from the survival bias typically associated with cross-sectional or case-control studies. The methodologic advantages of the prospective population-based cohort design, the similarity of phenotypes across 5 studies, the availability of genome-wide genotyping data in each cohort, and the need for large sample sizes to provide reliable estimates of genotype-phenotype associations have served as the primary incentives for the formation of the CHARGE consortium, which includes GWAS data on about 38 000 individuals. The consortium effort relies on collaborative methods that are similar to those used by the individual contributing cohorts.
Phenotype experts who know the studies and the data well are responsible for phenotype-standardization across cohorts. The coordinated prospectively planned meta-analyses of CHARGE provide results that are virtually identical to a cohort-adjusted pooled analysis of individual level data. This approach–the within-study analysis followed by a between-study meta-analysis–avoids the human subjects issues associated with individual-level data sharing.
Editors, reviewers, and readers expect replication as the standard in science.6 The finding of a genetic association in one population with evidence for replication in multiple independent populations provides moderate assurance against false-positive reports and helps to establish the validity of the original finding. In a single experiment, the discovery-replication structure is traditionally embodied in a 2-stage design. The CHARGE consortium includes up to 5 independent replicate samples as well as additional collaborating studies for some phenotype working groups, so that it would have been possible to set up analysis plans within CHARGE to mimic the traditional 2-stage design for replication. For instance, the 2 largest cohorts could have served as the discovery set and the others as the replication set. However, attaining the extremely small probability values expected in GWAS requires large sample sizes. For any phenotype, a prospective meta-analysis of all participating cohorts, with a properly selected level of genome-wide statistical significance to minimize the chance of false-positives, is the most powerful approach to finding new genuine associations for genetic loci.25 When findings narrowly miss the prespecified significance threshold, genotyping individuals in other independent populations provides additional evidence about the association. For findings that substantially exceed pre-established significance thresholds, the results of a CHARGE meta-analysis effectively provide evidence of a multistudy replication.
The effort to assemble and manage the CHARGE consortium has provided some interesting and unanticipated challenges. Participating cohorts often had relationships with outside study groups that predated the formation of CHARGE. Timelines for genotyping and imputation have shifted. Purchases of new computer systems for the volume of work were sometimes necessary. Each cohort came to the consortium with their own traditions for methods of analysis, organization, and authorship policies that, while appropriate for their own work, were not always optimal for collaboration with multiple external groups. Within each cohort, the investigators had often formed working groups that divided up the large number of available phenotypes in ways that made sense locally but did not necessarily match the configuration that had been adopted by other cohorts. The Research Steering Committee has attempted to create a set of CHARGE working groups that accommodate the needs and the conventions of the various cohorts. Transparency, disclosure, and professional collaborative behavior by all participating investigators have been essential to the process.
Resource limitations are another challenge. Grant applications that funded the original single-study genome-wide genotyping effort typically imagined a much simpler design. The CHS whole-genome study had as its primary aim, for instance, the analysis of data on 3 endpoints, coronary disease, stroke and heart failure. With a score of active phenotype working groups, the CHARGE collaboration broadened the scope of the short-term work well beyond initial expectations for all the participating cohorts.
One of the premier challenges has been communications among scores of investigators at a dozen sites. CHS and ARIC are themselves multi-site studies. To be successful, the CHARGE collaboration has required effective communications: (1) within each cohort; (2) between cohorts; (3) within the CHARGE working groups; and (4) among the major CHARGE committees. In addition to the traditional methods of conference calls and email, the CHARGE “wiki,” set up by Dr J. Bis (Seattle, Wash), has provided a crucial and highly functional user-driven website for calendars, minutes, guidelines, working group analysis plans, manuscript proposals, and other documents. In the end, there is no substitute for face-to-face meetings, especially at the beginning of the collaboration, and this complex meta-organization has benefited from several CHARGE-wide meetings.
The major emerging opportunity is the collaboration with other studies and consortia. Many working groups have already incorporated nonmember studies into their efforts. Several working groups have coordinated submissions of initial manuscripts with the parallel submission of manuscripts from other studies or consortia. Several working groups have embarked on plans for joint meta-analyses between CHARGE and other consortia. CHARGE has tried to acknowledge and reward the efforts of champions, who assume leadership responsibility for moving these large complex projects forward and who are often hard-working young investigators, the key to the future success of population science.
The CHARGE Consortium represents an innovative model of collaborative research conducted by research teams that know well the strengths, the limitations, and the data from 5 prospective population-based cohort studies. By leveraging the dense genotyping, deep phenotyping and the diverse expertise, prospective meta-analyses are underway to identify and replicate the major common genetic determinants of risk factors, measures of subclinical disease, and clinical events for cardiovascular disease and aging.
SOURCE:
Circulation: Cardiovascular Genetics.2009; 2: 73-80
doi: 10.1161/ CIRCGENETICS.108.829747