Controlling focused-treatment of Prostate cancer with MRI
Writer and reporter: Dror Nir, PhD.
In recent years there is a growing trend of treating prostate cancer in a way that will preserve, at least partially, the functionality of this organ. When patients are presenting at biopsy a low-grade localized disease, they might be offered focused treatment of the cancer lesion. One of the option is treatment by high-intensity focused ultrasound (HIFU).
The offering of such treatments created the need of controlling their outcome while the prostate is still inside the patient’s body. The most commonly used protocol is following up the patient’s PSA levels and performing “control” biopsies. The biopsies part is at best case; extremely unpleasant. It also bears some risk for complications.
Therefore, urologists are constantly seeking an imaging based protocol that will enable them to assess the treatment outcome without the need for biopsy. The publication I bring below presents the possibility of using MRI for this task. Although it is not recent, it contains many images that makes the story very clear for the reader. The main weakness of the study is the small number of patients – only 15.
Alexander P. S. Kirkham, FRCR, Mark Emberton, FRCS, Ivan M. Hoh, MRCS, Rowland O. Illing, MRCS, A. Alex Freeman, FRCP and Clare Allen, FRCR
From the Department of Imaging, University College London Hospitals NHS Foundation Trust, England (A.P.S.K., C.A.); Institute of Urology (M.E., I.M.H., R.O.I.) and Department of Histopathology (A.A.F.), University College London, England.
Address correspondence to A.P.S.K., Imaging Department, University College Hospital, 235 Euston Road, London, England NW1 2BU (e-mail: alexkirkham@yahoo.com).
Radiology March 2008; 246 (3) – 833-844.
Abstract
Purpose: To prospectively evaluate magnetic resonance (MR) imaging findings after high-intensity focused ultrasound (HIFU) treatment of the prostate and to correlate them with clinical and histologic findings.
Materials and Methods: Local ethics committee approval and informed consent were obtained. Fifteen consecutive men aged 46–70 years with organ-confined prostate cancer underwent ultrasonographically guided ablation of the whole prostate. Postoperative MR images were obtained within 1 month (12 patients), at 1–3 months (five patients), and in all patients at 6 months. Prostate volume was measured on T2-weighted images, and enhancing tissue was measured on dynamic images after intravenous administration of gadopentetate dimeglumine. Prostate-specific antigen (PSA) level was measured at regular intervals, and transrectal biopsy was performed in each patient at 6 months after treatment.
Results: Initial post-HIFU images showed a central nonenhancing area, surrounded by an enhancing rim. At 6 months, the prostate was small (median volume reduction, 61%) and was of predominantly low signal intensity on T2-weighted images. The volume of prostate enhancing on the initial posttreatment image correlated well with serum PSA level nadir (Spearman r = 0.90, P < .001) and with volume at 6 months (Pearsonr = 0.80, P = .001). The three patients with the highest volume of enhancing prostate at the initial posttreatment acquisition had persistent cancer at 6-month biopsy.
Conclusion: MR imaging results of the prostate show a consistent sequence of changes after treatment with HIFU and can provide information to the operator about completeness of treatment.
There is currently little to offer men with localized prostate cancer between the two extremes of watchful waiting and radical treatment—most commonly prostatectomy or radiation therapy (1). Ablation of the gland has been proposed as an alternative that has the potential to completely treat the tumor while minimizing the sexual and urinary morbidity that still accompany established radical therapies (2). Several techniques have been used in the prostate—including microwave (3) and radiofrequency (4) ablation, cryotherapy (5), photodynamic therapy (6), and high-intensity focused ultrasound (HIFU) treatment (7).
HIFU is, in several respects, ideally suited to the prostate. In contrast to extracorporeal devices for the liver and kidney (8), with the transrectal approach, there is little movement of the target because of respiration or reflection by overlying bone. A focal distance of 3 or 4 cm allows the generation of coagulative necrosis in treatment voxels less than 0.2 mL and allows a treatment volume that conforms to the shape of the prostate (9)—a degree of precision that may be beyond that of other techniques. Even so, complete ablation is likely to affect periprostatic tissues, including the neurovascular bundles containing the cavernosal nerves (10) and the external urethral sphincter. Preservation of these structures—and the patient’s erectile and urinary function—must be balanced against full treatment of the gland.
Although impotence rates after HIFU treatment approach 50% (11), it is likely that in its current clinical implementation, the prostate is not being fully ablated: In published series, the recurrence rates for cancer range between 25% and 38% (7,11,12). To our knowledge, no groups have reported mean reductions in prostate volume of more than 50% (12,13), and several groups have found it difficult to treat the anterior gland (14).
If we are to improve outcomes, a fundamental requirement for HIFU treatment (and ablative technologies in general) is a method that provides anatomic information to the operator about areas that have been over- or undertreated. This might lead to modifications in future technique, and if obtained soon after treatment, might indicate the need for further ablation. Such a method might also help predict outcome earlier than established measures, such as prostate-specific antigen (PSA) measurement and biopsy.
Magnetic resonance (MR) imaging has great potential in this setting, and Rouviere et al (14) have described the appearance of the prostate on contrast material–enhanced MR images obtained up to 5 months after HIFU treatment. Rouviere et al found a good correlation between the theoretical treatment volume and the volume of nonenhancing prostate on a subsequent acquisition. The aim of our study was to prospectively evaluate MR imaging findings after HIFU treatment of the prostate and to correlate them with clinical and histologic findings.
MATERIALS AND METHODS
Misonix (the European distributors of the Sonablate device) funded the phase-II European study and provided equipment and reimbursed the hospital for costs. The company has funded two authors (I.M.H. and R.O.I.) through educational awards. One author (M.E.) has acted as a paid consultant to Misonix and also received honoraria for training and teaching. Authors other than I.M.H., R.O.I., and M.E. had control of the information and data submitted for publication. Misonix was not involved in the analysis of data or the writing of this article.
Patients
We included the first 15 men at University College Hospital (age range, 46–70 years; mean age, 59 years) who were taking part in a registered phase-II multicenter European study of HIFU therapy for organ-confined prostate cancer (Table 1). The study was approved by the local ethics committee, and full written consent was obtained from each patient. The patients understood that HIFU is an experimental treatment whose long-term outcome is unknown and were offered full conventional treatment as an alternative. The study was limited to men with a serum PSA level 15 μg/L or less, Gleason score less than 8, prostate volume less than 40 mL, life expectancy more than 5 years, and age less than 80 years. There was no limit to the number of biopsy cores that had a positive finding or the amount of cancer in each core removed. Patients with a history of previous prostate surgery were excluded, as were men who had undergone androgen deprivation therapy in the 6 months prior to recruitment or had intragland prostatic calcification more than 1 cm in diameter.
Table 1. Patients and Demographics

* Ratio of cores with a positive finding to cores obtained.
† Image not available for analysis; volume was calculated by using US measurements.
The Sonablate 500 (Focus Surgery, Indianapolis, Ind) consists of a power generator, water cooling system (the Sonachill), a treatment probe, and a positioning system. The probe contains two curved rectangular piezoceramic transducers with a driving frequency of 4 MHz and focal lengths of 30 and 40 mm. During treatment, these may be driven at low energy to provide real-time diagnostic imaging or at high energy for therapeutic ablation (in situ intensity, 1300–2200 W/cm2). The probe is covered with a condom, under which cold (17°–18°C) degassed water is circulated to help protect the rectum from thermal injury.
Patients were prepared before the procedure with two phosphate enemas to empty the rectum. Oral bowel preparation was used in some patients. Treatment was performed with general anesthesia in the lithotomy position and was performed or closely supervised in every case by an author (M.E., 2 years of experience in HIFU treatment). After gentle dilation of the anal sphincter, the treatment probe was introduced with a covering of ultrasonographic (US) gel to couple it to the rectal mucosa and was held in position with an articulated arm attached to the operating table. A 16-F Foley urethral catheter was inserted using sterile technique, and a 10-mL balloon was inflated to allow the bladder neck and median sagittal plane to be seen accurately. It was removed before treatment began.
Treatment was planned by using US-acquired volumes consisting of stacks of both sagittal and transverse sections (voxel size, 2 × 3 × 30 mm) and was applied in rows that extended in the craniocaudal axis, interleaved to avoid interference from adjacent, recently treated areas. After each 3-second period of ablation, diagnostic transverse and sagittal images in the plane of treatment were obtained to permit tailoring of the energy delivery in the next voxel according to visible changes on the gray-scale image. This is an important difference from the device used by Rouviere’s group (14), in which power is planned before the treatment begins. We aimed to set the power for each voxel at a level that produced hyperechoic change due to cavitation (as described by Illing et al [15]), and we invariably treated the whole anterior prostate. Neurovascular bundles were not identified at treatment (the Sonablate device does not yet have color Doppler capability); rather, we aimed to avoid treating outside the capsule where they lie posterolaterally (10). The time between the first ablation and the point at which treatment was considered complete was 3.0–4.4 hours (mean, 3.6 hours). A 16-F urethral catheter was placed immediately after the treatment and was left in place for 2 weeks.
MR Imaging
For most preoperative examinations and for all post-HIFU imaging, we used an MR machine (Symphony or Avanto; Siemens, Erlangen, Germany) with 1.5-T magnet and a pelvic-phased array coil. Except where stated, a full protocol of T1- and T2-weighted turbo spin-echo (Siemens) images and a dynamic fat-saturated postcontrast volume acquisition were used for both preoperative diagnostic and planning imaging and for postoperative assessment of HIFU treatment (Table 2). The contrast material used was 20 mL of gadopentetate dimeglumine (Magnevist; Schering, Berlin, Germany) given intravenously at 3 mL/sec.
Table 2. MR Sequences Used at Prostate Imaging

We aimed to image patients less than 1 month after treatment and did so in 12 patients. The remaining three patients were imaged between 1 and 3 months after treatment. Two patients were imaged in both time periods. Every patient underwent a 6-month MR examination.
Image Analysis
All volume measurements (except where stated) were acquired by using planimetry of contiguous 3-mm sections (16). T2-weighted images were used for measurement of prostatic volume both before and after treatment. The amount of intermediate- or high-signal-intensity material (ie, higher than muscle) remaining within the prostate was also measured on the 6-month posttreatment T2-weighted image.
The volume of nonenhancing prostate tissue at the post-HIFU acquisition was measured by using the final dynamic postcontrast image. On the initial posttreatment image, we also measured the volume of extraprostatic tissue that was both of low signal intensity on the T1-weighted image and nonenhancing. The distance between this tissue and the rectal mucosa was measured at its narrowest point. The mean thickness of the enhancing rim surrounding the treatment volume was measured on transverse postcontrast T1-weighted spin-echo images and was calculated by dividing the area of the rim by its circumference.
The volume of persistently enhancing prostate tissue on the initial image was calculated by subtracting the nonenhancing volume from the total volume of prostate on the T2-weighted image. This could be calculated in 13 patients; one patient did not receive contrast material at the post-HIFU MR acquisition, and the other was imaged more than 2 months after treatment.
All measurements were performed by a first-year radiology fellow (A.P.S.K.) without knowledge of PSA and histologic results. Two other observers independently measured the three key parameters that were used for correlation calculations for each patient: (a) the volume of nonenhancing prostate on the initial image, (b) the total volume of the prostate on the initial image, and (c) the final prostate volume at 6 months. One was a consultant uroradiologist with more than 10 years of experience in the interpretation of prostate MR images (C.A.); the other was a third-year urology research fellow with an interest in prostate imaging (R.O.I.). For each parameter, the mean of the three observers’ measurements was calculated and used for further analysis.
PSA Measurement and Prostate Biopsy
Serum PSA level was measured before and at 1.5, 3, and 6 months after HIFU treatment. The nadir was defined as the lowest of the three values.
Biopsies were performed by an author (A.P.S.K., with 4 years of experience in prostate biopsy) by using a transrectal approach with US guidance and an 18-gauge needle with a 2-cm throw soon after the 6-month MR examination. The number of cores obtained depended on the amount of residual prostate and varied between two and 10 (median, eight cores).
Erectile Function and Continence
The International Index of Erectile Function was used to assess erectile function both before and 3 months after HIFU treatment in each patient (17). The most important question was, “How often were your erections hard enough for penetration [with or without phosphodiesterase type 5 inhibitors]?” A score of 2 (a few times in 4 weeks) to 5 (always) was, for the purposes of this article, considered evidence of intact erectile function.
Men were asked to complete the International Continence Society–validated continence function questionnaire at baseline and at 3 and 6 months after therapy. The question deemed to be most informative was how often the patient required the use of pads or adult diapers. Responses could include “never,” “not more than one per day,” “1–2 per day,” or “more than 3 per day.”
Statistical Analysis
To assess the variance of results between observers, we used the intraclass correlation coefficient (18) applied to measurements obtained by three observers of the calculated volume of enhancing prostate on the initial post-HIFU image and the 6-month prostate volume.
The Spearman rank test was used to assess the correlation between enhancing prostate volume and serum PSA level nadir, and the Pearson test was used to examine the correlation between initial enhancing prostate volume and final prostate volume. Only the patients who were imaged less than 1 month after treatment were included in the analysis. These tests were performed by using software (GraphPad Prism for Mac, version 3; http://www.graphpad.com).
Because some of the covariance of volumes measured after treatment was likely to be due to their correlation with pretreatment prostate volume, we also applied a correction: The values were expressed as a proportion of the pretreatment volume, and a further correlation measurement was performed by using the Pearson test. In each case, a P value of less than .02 was considered to indicate a significant difference.
RESULTS
Up to 1 Month After Treatment
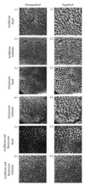
T2-weighted images.—Compared with that on the preoperative image, the prostate volume increased in every case (Table 1 and Table E1, Fig 1). The signal intensity from the prostate on T2-weighted images within the first month was always heterogeneous and variable. It was impossible to predict from the findings on T2-weighted images which areas of the prostate would enhance after intravenous contrast material administration. The periprostatic fat was also heterogeneous in signal intensity, which was consistent with edema (Fig 2).
Figure 1: Graph of change in prostate volume after HIFU treatment. Volume rises initially (less than 1 month after treatment) and is reduced in all cases at 6 months. Numbers = patient numbers.

Figure 2: MR images in patient 1 (a–d) and (e–h) patient 8 show low volume of enhancing prostate at initial imaging and small residual prostate at 6 months. Posttreatment serum PSA level was less than 0.05 μg/L in both cases.
Figure 2a:

Figure 2b:

Figure 2c:

Figure 2d:

Figure 2e:

Figure 2f:

Figure 2g:

Figure 2h:

T1-weighted images.—The prostate was of predominantly low signal intensity, although patchy areas of intermediate or high signal intensity, likely to represent hemorrhage, were a constant finding within the gland and in all but one of 28 seminal vesicles.
Postcontrast images.—In each patient, the postcontrast images showed a central area of nonenhancing tissue. This conformed to the treatment volume and was surrounded by an enhancing rim of mean thickness of 2–8 mm (median, 4 mm) that was continuous around the prostate in most patients (Fig 2; Table E1,).
The enhancing prostate varied in size and position. Part of the enhancing rim usually lay within the prostatic capsule and continued to the prostatic apex where there was almost always some enhancing tissue between the nonenhancing prostate and the external urethral sphincter. In many patients, more central areas of enhancement were seen: at the apex or base, either posteriorly or anteriorly (Table E1), and were almost always in continuity with the rim.
In every patient, the nonenhancing, low signal intensity within the prostate extended outside the gland and involved the periprostatic fat and the levator ani muscle, particularly anterolaterally (Table E1, Figs 2, 3). This varied considerably and tended to be most prominent in those who had no residual gland enhancement and had an undetectable serum PSA level after HIFU treatment (Table E1). In several patients, the nonenhancing area extended to involve the Denonvilliers fascia. (The distance between its margin and the rectal muscle is listed in Table E1.) In one patient, a proportion of the rectal wall enhanced avidly, but in no patient was there loss of rectal wall enhancement to suggest necrosis.
Figure 3: MR images obtained near the prostate apex show incomplete treatment and persisting high signal intensity in prostate. Serum PSA level nadir = 0.61 μg/L.
Figure 3a: Patient 4:

Figure 3b: Patient 4:

Figure 3c: Patient 4:

Figure 3d: Patient 4:

At 1–3 Months
In three patients, there was a “double rim” (Fig 4) on postcontrast images obtained at 36 and 56 days after HIFU treatment. The inner component lay within the prostate and the outer at the prostatic capsule; the intervening part was of low signal intensity on both T1- and T2-weighted images.
Figure 4: MR images of “double rim” at 56 days after HIFU treatment.
Figure 4a: Patient 3:

Figure 4b: Patient 3:

Figure 4c: Patient 3:

Six-month Appearance
T2-weighted images.—In every patient, the volume of the prostate was reduced by more than 45% (median, 61% reduction) (Table E1). On T2-weighted images, the majority of the persisting prostate was of low signal intensity, with poor definition to the capsule and with persisting heterogeneous signal intensity to the surrounding fat. However, in 12 of 15 patients, there was persisting high or intermediate signal intensity of the prostate—up to 5.34 mL in volume and most often seen posteriorly and at the apex (Table E1, Figs 3 and 5). In many patients (for example, those in Fig 2), low-signal-intensity prostate of reduced volume surrounded a capacious prostatic cavity continuous with the urethra, which is similar to the cavity seen after transurethral resection (19).
Figure 5: MR images of incomplete treatment of tumor and positive biopsy findings in three of 10 cores at 6 months (in right lateral midzone, right lateral base, and right parasagittal base samples). Serum PSA level nadir = 1.19 μg/L.
Figure 5a: Patient 13:

Figure 5b: Patient 13:

Figure 5c: Patient 13:

Figure 5d: Patient 13:

Figure 5e: Patient 13:

Postcontrast images.—Some small areas of nonenhancing tissue persisted in eight of 14 patients, but this was less than 1 mL in all but one (patient 13, in whom 4 mL of the gland volume of 18.7 mL was nonenhancing). The levator muscle showed a normal signal intensity.
Correlation Between Initial Imaging and Later Findings
In the 12 patients who underwent the initial acquisition within 1 month of HIFU treatment, the volume of enhancing tissue on the initial posttreatment image was positively correlated with the serum PSA level nadir (Fig 6) (Spearman r = 0.90, P < .001) and with the amount of residual tissue at 6 months (including all low-signal-intensity material that was likely to represent fibrosis or necrosis) (Fig 7) (Pearson r = 0.80, P = .001).
Figure 6: Graph of relationship between the proportion of the prostate still enhancing on initial image and serum PSA level nadir. There is a significant positive correlation (Spearman r = 0.90, P < .001). * = patient 13, who was included in graph but not in analysis (imaged 56 days after HIFU treatment). Patients 14 and 15 are not included because they did not undergo contrast-enhanced acquisition within 2 months of HIFU treatment. μgl−1 = μg/L.

Figure 7: Graph of relationship between the proportion of the prostate still enhancing on initial image and final volume of prostate. There is a significant positive correlation between the variables (Pearson r = 0.80, P = .001). * = patient 13, who was included in graph but not in analysis (imaged 56 days after HIFU treatment). Patients 14 and 15 are not included because they did not undergo contrast-enhanced acquisition within 2 months of HIFU treatment.

When posttreatment volumes are expressed as a proportion of pretreatment prostate volume, the correlation between enhancing tissue volume on the initial posttreatment image and the 6-month prostate volume persists (Pearson r = 0.70, P = .001).
Interobserver Correlation
The interobserver variation was excellent for the calculated volume of prostate enhancing on the initial post-HIFU image, with an intraclass correlation coefficient of 0.92, and was good for final prostate volume (intraclass correlation coefficient = 0.73).
Clinical Findings
In five patients (patients 1, 3, 8, 11, and 13), there was imaging evidence (at MR imaging or retrograde urethrography) of a stricture in the mid- or distal prostatic urethra, which was confirmed by using flow rate studies and treated by using self-catheterization or with graded urethral dilators. None have required formal urethrotomy. Patient 14 developed a bladder neck stricture, which was treated successfully by incision.
Before treatment, no men required pads or adult diapers for incontinence. At 6 months after the treatment, four men still required not more than one pad per day. In two cases, this was for reassurance rather than actual leakage.
In the 14 patients in whom there was intact erectile function (score 2–5 for the question, “How often were your erections hard enough for penetration?”) before HIFU treatment, it was intact in nine patients after the procedure. One patient had stopped trying to achieve erections, and four could not achieve penetration.
Histologic Findings
In the three patients in whom there was no high-signal-intensity peripheral zone at 6 months and with serum PSA level less than 0.05 μg/L, there was either no prostatic tissue or only a small group of acini in one core. The remaining patients had a variable amount of residual prostate at core biopsy.
Five patients had residual tumor. In three patients, it was seen in at least two cores (Table E1), and these three patients also had the largest volume of enhancing prostate on the initial post-HIFU MR image (Figs 6 and 7) and more than 2 mL of intermediate- or high-signal-intensity gland on T2-weighted images at 6 months.
In four of five patients with residual cancer, it could not be identified on either contrast-enhanced or T2-weighted images. In one patient (Fig 4), the early dynamic images showed prominent enhancement in the anterior gland, which was consistent with residual cancer found at the distal (ie, nonrectal) end of three right-sided biopsy cores. Such enhancement was not seen in patients with no cancer found at core biopsy.
DISCUSSION
We found a consistent sequence of changes at MR imaging after HIFU treatment of the whole prostate. The proportion of enhancing tissue on the initial posttreatment MR image was predictive of gland volume at 6 months and serum PSA level nadir. A strong statistical relationship between the latter and outcome has recently been demonstrated (20).
Most patients with residual cancer had evidence of incomplete ablation early (a large volume of enhancing prostate on the initial image) and late (a large volume of high-signal-intensity residual prostate on T2-weighted images at 6 months).
In some patients it was possible to achieve an undetectable serum PSA level at 6 months and entirely low signal intensity on T2-weighted images in the region of the prostate. These patients had either no or a small amount of viable prostate in one core at biopsy.
Conversely, in spite of reductions in prostate volume of more than 45% at 6 months, the majority of patients had histologic evidence of persisting viable prostate, and in a group of patients with organ-confined disease but no limit to the volume of cancer pretreatment, one-third had evidence of residual tumor.
Persisting enhancing prostatic tissue usually occurred at the periphery (or extended toward the center of the gland from it) and was particularly common at the apex and near the rectum.
Results of one previously published study (14) of post-HIFU appearances with MR imaging show a similar sequence of acute changes, although there was no attempt to quantify prostate volume at 6 months. There is also a large body of work on the MR imaging appearances with thermotherapy (whether laser [21,22] or radiofrequency [23]) and cryotherapy (24) within the prostate and other organs. The hyperenhancing rim of tissue is a constant finding in several tissues, including the liver (25), the kidney (26), and the brain (27). In the liver and the kidney, it is thin (1 mm or less) and, in most cases, has disappeared by 2 months after ablation (28). Within the prostate, the hyperenhancing rim has been shown to occur after laser ablation of benign prostatic hyperplasia (21,22) and after HIFU treatment (14).
Histologic evidence in animal models—including rabbit and porcine liver (29)—suggests that the enhancing rim corresponds to an area of inflammation and then fibrosis, with a variable amount of residual, viable tissue. How much of the rim will be viable after ablation of the prostate in humans remains uncertain. On the one hand, after HIFU treatment, core biopsy results show “partial or complete necrosis” in the rim (14). On the other, after laser ablation of benign prostatic hyperplasia, the volume of coagulative necrosis at histologic examination correlates very well with the central nonenhancing region at MR imaging, not including the rim (22). The answer is likely to be that a variable amount of the rim contains viable tissue (depending on the organ being imaged [30], the nature of the treatment, and the interval before the acquisition), and the implication is that the only reliably necrotic area at MR imaging is that which does not enhance. We have avoided the term necrosis for the nonenhancing areas of prostate seen in our current study, but from these data it is likely that the areas of prostate without enhancement are truly necrotic.
The distribution of enhancing prostate on posttreatment MR images fits with histologic evidence that “ventral, lateral and dorsal sides of the prostate” have residual viable prostatic tissue at histologic examination after HIFU treatment (31). What all of these areas have in common is proximity to the more richly vascular prostatic capsule. Is it possible that increased vascularity here results in reduced efficacy? This is another area that has been addressed by Rouviere’s group (32), who did not find a correlation between successful ablation and prostate vascularity by using power Doppler US; they conclude, as others have (33,34), that short (3-second) high-intensity bursts of focused ultrasound are unlikely to be markedly affected by blood flow. An alternate explanation is a geometric one: Centrally lying voxels are easier to treat because they may be rendered necrotic either by direct treatment or by damage to supplying vessels in the periphery.
An implication of these results is that the best strategies for minimizing complications while ensuring destruction of the cancer are likely to involve a degree of targeting: If the tumor can be imaged with MR imaging, the patient might be treated with higher power and wider margins (including periprostatic fat, muscle, or even neurovascular bundles) at the site of the cancer and with a standard intensity to the rest of the gland. An analogous approach is the wide excision, including a unilateral neurovascular bundle, of bulky tumors at radical prostatectomy (35). Such an approach may well have benefited our patients 7 and 13.
One methodologic issue that is currently unresolved relates to the timing of MR imaging. A detailed within-patient study of MR imaging changes after HIFU treatment is needed to properly describe the longitudinal changes in the appearance of the prostate. Rouviere et al (14) found that the area of nonenhancing tissue decreases by 50% at 1 month compared with that at an immediate (<1 week) post-HIFU acquisition, which suggests that for an accurate assessment of necrosis volume, the prostate should be imaged as soon as possible after treatment. Of course, perfusion would ideally be assessed during HIFU treatment so that undertreated areas could be further ablated. There is some evidence that Doppler or contrast-enhanced US (36) could play this role, but, to our knowledge, there are no studies on the correlation of immediate findings with later clinical data, such as serum PSA level or histologic examination.
We used fast low-angle shot sequences to assess enhancement because we found that the subjective assessment (together with objective measurements of signal intensity) of the dynamic series helped us identify truly nonenhancing tissue. However, the T1-weighted spin-echo postcontrast sequence would have been adequate, and we consider, as others do (22), dynamic contrast-enhanced sequences not to be an essential part of the protocol for postablation assessment. What is certain is that unenhanced T2-weighted sequences are inadequate for assessing necrosis (14,22).
Our results differ from those of other published series of HIFU treatment in the marked reduction in gland volume and absence of zonal anatomy in many patients observed at 6 months. In contrast to the study of post-HIFU MR imaging by Rouviere et al (14) who used a different device, we did not find that “HIFU-induced abnormalities seem to disappear within 3–5 months.” Rather, in several patients, it was difficult to discern any residual prostate at all at both MR and US studies. The difference probably lies in the power used for treatment and the completeness of gland coverage. The stricture rate of six of 15 is high when compared with that in published series (7,37,38) and may be related to the power used, the degree of fibrosis occurring in the prostate, and the strategy for catheterization. The latter is considered likely to be important, and we have recently changed to using a suprapubic catheter (rather than urethral) after treatment. The rate of impotence after treatment is similar to that in published series (11), as is grade I incontinence.
Our work has implications for the conduct of HIFU. The finding that the volume of enhancing prostate on the initial posttreatment image correlates well with intermediate measures, such as serum PSA level nadir and biopsy evidence of residual cancer, suggests that MR imaging can provide the operator with feedback on the effectiveness of the intervention. This information might enable modification of the technique to treat areas that have been incompletely ablated in previous patients—in our series, those areas encompassed the apex and posterior gland and rarely anterior tissue (in contrast to other study results [14]). Conversely, we might have reduced power or treatment volume at the anterolateral aspect of the gland adjacent to the levator muscle. Such feedback has been cited as a desirable attribute for ablation technology (39) and up to now has been missing.
Our study had several limitations. Although it is likely that nonenhancing areas at MR imaging represent necrosis, we do not have direct histologic evidence. Sampling error and misregistration limit the utility of core biopsies in this context. We have shown that the MR imaging appearances soon after HIFU treatment correlate with findings at 6 months, but this is not the same as outcome. A considerably longer follow-up and a larger number of patients will be necessary to determine both the ultimate efficacy of HIFU treatment and the ability of MR imaging to help predict outcome. Last, while our findings suggest that MR imaging soon after treatment may be useful to assess areas of under- and overtreatment, this is not real-time feedback and does not allow modification of the treatment as it progresses.
In summary, MR imaging results in the first 6 months after HIFU treatment show a consistent sequence of changes, and appearances in the 1st month correlate with serum PSA level nadir and imaging findings at 6 months. Such imaging results hold promise for providing feedback to the operator about the effectiveness of treatment.
ADVANCES IN KNOWLEDGE
- Treatment of prostate cancer by using ablation with high-intensity focused ultrasound (HIFU) results in a consistent series of changes within the gland during 6 months seen at contrast-enhanced MR imaging.
- Within 1 month after treatment, a central nonenhancing area is surrounded by an enhancing rim of tissue lying variably within and outside the prostate.
- At 6 months, the gland is markedly smaller and of partly or completely low signal intensity on T2-weighted images.
- The amount of enhancing prostate on the initial image correlates with several findings at 6 months, including serum prostate-specific antigen level nadir and prostate volume.
IMPLICATION FOR PATIENT CARE
- MR imaging after HIFU treatment may provide information about completeness of tumor ablation and the need for early retreatment or close monitoring in cases of incomplete coverage.
Footnotes
- Trial registration: This trial started recruiting before the trial registration requirements of the International Committee of Medical Journal Editors were formalized.
See Materials and Methods for pertinent disclosures.
Author contributions: Guarantors of integrity of entire study, A.P.S.K., I.M.H., C.A.; study concepts/study design or data acquisition or data analysis/interpretation, all authors; manuscript drafting or manuscript revision for important intellectual content, all authors; manuscript final version approval, all authors; literature research, A.P.S.K., M.E., I.M.H., R.O.I., C.A.; clinical studies, A.P.S.K., R.O.I., C.A.; statistical analysis, A.P.S.K.; and manuscript editing, all authors
Abbreviations:HIFU = high-intensity focused ultrasoundPSA = prostate-specific antigen
References
- ↵
Parker C. Active surveillance: towards a new paradigm in the management of early prostate cancer. Lancet Oncol 2004; 5: 101–106.
CrossRefMedline
- ↵
Steineck G, Helgesen F, Adolfsson J, et al. Quality of life after radical prostatectomy or watchful waiting. N Engl J Med 2002;347:790–796.
CrossRefMedline
- ↵
Huidobro C, Bolmsjo M, Larson T, et al. Evaluation of microwave thermotherapy with histopathology, magnetic resonance imaging and temperature mapping. J Urol 2004;171:672–678.
CrossRefMedline
- ↵
Shariat SF, Raptidis G, Masatoschi M, Bergamaschi F, Slawin KM. Pilot study of radiofrequency interstitial tumor ablation (RITA) for the treatment of radio-recurrent prostate cancer. Prostate 2005;65:260–267.
CrossRefMedline
- ↵
Onik G. The male lumpectomy: rationale for a cancer targeted approach for prostate cryoablation—a review. Technol Cancer Res Treat2004;3:365–370.
Medline
- ↵
Moore CM, Hoh IM, Bown SG, Emberton M. Does photodynamic therapy have the necessary attributes to become a future treatment for organ-confined prostate cancer? BJU Int 2005;96:754–758.
CrossRefMedline
- ↵
Uchida T, Ohkusa H, Nagata Y, Hyodo T, Satoh T, Irie A. Treatment of localized prostate cancer using high-intensity focused ultrasound. BJU Int2006;97:56–61.
CrossRefMedline
- ↵
Kennedy JE. High-intensity focused ultrasound in the treatment of solid tumours. Nat Rev Cancer 2005;5:321–327.
CrossRefMedline
- ↵
Uchida T, Sanghvi NT, Gardner TA, et al. Transrectal high-intensity focused ultrasound for treatment of patients with stage T1b-2n0m0 localized prostate cancer: a preliminary report. Urology 2002;59:394–398.
CrossRefMedline
- ↵
Walsh PC, Donker PJ. Impotence following radical prostatectomy: insight into etiology and prevention. J Urol 1982;128:492–497.
Medline
- ↵
Blana A, Walter B, Rogenhofer S, Wieland WF. High-intensity focused ultrasound for the treatment of localized prostate cancer: 5-year experience.Urology 2004;63:297–300.
CrossRefMedline
- ↵
Gelet A, Chapelon JY, Bouvier R, et al. Transrectal high-intensity focused ultrasound: minimally invasive therapy of localized prostate cancer. J Endourol 2000;14:519–528.
Medline
- ↵
Chaussy C, Thuroff S. The status of high-intensity focused ultrasound in the treatment of localized prostate cancer and the impact of a combined resection. Curr Urol Rep 2003;4:248–252.
CrossRefMedline
- ↵
Rouviere O, Lyonnet D, Raudrant A, et al. MRI appearance of prostate following transrectal HIFU ablation of localized cancer. Eur Urol2001;40:265–274.
CrossRefMedline
- ↵
Illing RO, Leslie TA, Kennedy JE, Calleary JG, Ogden CW, Emberton M. Visually directed high-intensity focused ultrasound for organ-confined prostate cancer: a proposed standard for the conduct of therapy. BJU Int 2006;98:1187–1192.
CrossRefMedline
- ↵
Bakker J, Olree M, Kaatee R, de Lange EE, Beek FJ. In vitro measurement of kidney size: comparison of ultrasonography and MRI. Ultrasound Med Biol 1998;24:683–688.
CrossRefMedline
- ↵
Rosen RC, Cappelleri JC, Smith MD, Lipsky J, Pena BM. Development and evaluation of an abridged, 5-item version of the International Index of Erectile Function (IIEF-5) as a diagnostic tool for erectile dysfunction. Int J Impot Res 1999;11:319–326.
CrossRefMedline
- ↵
Fleiss JL. Reliability of measurement. In: The design and analysis of clinical experiments. New York, NY: Wiley, 1986; 1–32.
- ↵
Sheu MH, Chiang H, Wang JH, Chang YH, Chang CY. Transurethral resection of the prostate-related changes in the prostate gland: correlation of MRI and histopathology. J Comput Assist Tomogr 2000;24:596–599.
CrossRefMedline
- ↵
Uchida T, Illing RO, Cathcart PJ, Emberton M. To what extent does the prostate-specific antigen nadir predict subsequent treatment failure after transrectal high-intensity focused ultrasound therapy for presumed localized adenocarcinoma of the prostate? BJU Int 2006;98:537–539.
CrossRefMedline
- ↵
Mueller-Lisse UG, Heuck AF, Schneede P, et al. Postoperative MRI in patients undergoing interstitial laser coagulation thermotherapy of benign prostatic hyperplasia. J Comput Assist Tomogr 1996;20:273–278.
CrossRefMedline
- ↵
Boni RA, Sulser T, Jochum W, Romanowski B, Debatin JF, Krestin GP. Laser ablation-induced changes in the prostate: findings at endorectal MR imaging with histologic correlation. Radiology 1997;202:232–236.
Abstract/FREE Full Text
- ↵
Zlotta AR, Djavan B, Matos C, et al. Percutaneous transperineal radiofrequency ablation of prostate tumour: safety, feasibility and pathological effects on human prostate cancer. Br J Urol 1998;81:265–275.
Medline
- ↵
Vellet AD, Saliken J, Donnelly B, et al. Prostatic cryosurgery: use of MR imaging in evaluation of success and technical modifications. Radiology1997;203:653–659.
Abstract/FREE Full Text
- ↵
Guan YS, Sun L, Zhou XP, Li X, Zheng XH. Hepatocellular carcinoma treated with interventional procedures: CT and MRI follow-up. World J Gastroenterol 2004;10:3543–3548.
Medline
- ↵
Merkle EM, Nour SG, Lewin JS. MR imaging follow-up after percutaneous radiofrequency ablation of renal cell carcinoma: findings in 18 patients during first 6 months. Radiology 2005;235:1065–1071.
Abstract/FREE Full Text
- ↵
Kahn T, Bettag M, Ulrich F, et al. MRI-guided laser-induced interstitial thermotherapy of cerebral neoplasms. J Comput Assist Tomogr1994;18:519–532.
Medline
- ↵
Dromain C, de Baere T, Elias D, et al. Hepatic tumors treated with percutaneous radiofrequency ablation: CT and MR imaging follow-up. Radiology2002;223:255–262.
Abstract/FREE Full Text
- ↵
Raman SS, Lu DS, Vodopich DJ, Sayre J, Lassman C. Creation of radiofrequency lesions in a porcine model: correlation with sonography, CT, and histopathology. AJR Am J Roentgenol 2000;175:1253–1258.
Abstract/FREE Full Text
- ↵
Illing RO, Kennedy JE, Wu F, et al. The safety and feasibility of extracorporeal high-intensity focused ultrasound (HIFU) for the treatment of liver and kidney tumours in a Western population. Br J Cancer 2005;93:890–895.
CrossRefMedline
- ↵
Van Leenders GJ, Beerlage HP, Ruijter ET, de la Rosette JJ, van de Kaa CA. Histopathological changes associated with high intensity focused ultrasound (HIFU) treatment for localised adenocarcinoma of the prostate. J Clin Pathol 2000;53:391–394.
Abstract/FREE Full Text
- ↵
Rouviere O, Curiel L, Chapelon JY, et al. Can color doppler predict the uniformity of HIFU-induced prostate tissue destruction? Prostate2004;60:289–297.
- ↵
Billard BE, Hynynen K, Roemer RB. Effects of physical parameters on high temperature ultrasound hyperthermia. Ultrasound Med Biol1990;16:409–420.
CrossRefMedline
- ↵
Chen L, ter Haar G, Hill CR, et al. Effect of blood perfusion on the ablation of liver parenchyma with high-intensity focused ultrasound. Phys Med Biol 1993;38:1661–1673.
CrossRefMedline
- ↵
Park EL, Dalkin B, Escobar C, Nagle RB. Site-specific positive margins at radical prostatectomy: assessing cancer-control benefits of wide excision of the neurovascular bundle on a side with cancer on biopsy. BJU Int 2003;91:219–222.
CrossRefMedline
- ↵
Sedelaar JP, Aarnink RG, van Leenders GJ, et al. The application of three-dimensional contrast-enhanced ultrasound to measure volume of affected tissue after HIFU treatment for localized prostate cancer. Eur Urol 2000;37:559–568.
CrossRefMedline
- ↵
Thuroff S, Chaussy C, Vallancien G, et al. High-intensity focused ultrasound and localized prostate cancer: efficacy results from the European multicentric study. J Endourol 2003;17:673–677.
CrossRefMedline
- ↵
Gelet A, Chapelon JY, Bouvier R, Rouviere O, Lyonnet D, Dubernard JM. Transrectal high intensity focused ultrasound for the treatment of localized prostate cancer: factors influencing the outcome. Eur Urol 2001;40:124–129.
CrossRefMedline
- ↵
Gillett MD, Gettman MT, Zincke H, Blute ML. Tissue ablation technologies for localized prostate cancer. Mayo Clin Proc 2004;79:1547–1555.
CrossRefMedline
Like this:
Like Loading...