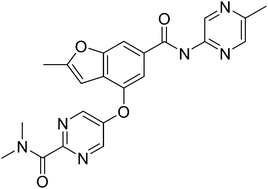
novel Schiff base metal complexes
Larry H. Bernstein, MD, FCAP, Curator
LPBI
Synthesis and Characterisation of some novel Schiff base metal complexes: Spectral, XRD, Photoluminescence and Antimicrobial Studies
Rajendran Jayalakshmi and Rangappan Rajavel* – Department of chemistry, Periyar University, Salem-636 011, Tamil Nadu, India
Chem Sci Rev Lett 2015; 4(15); 851-859 Article CS072046073 http://chesci.com/articles/csrl/v4i15/11_CS072046073.pdf
Schiff base ligands are potentially capable of forming stable complexes with most transition metal ions, which act as model compounds for biologically important species. Cu (II), Co (II), Ni (II) and Mn (II) Schiff base metal complexes was prepared by the condensation of benzil and 2,6-diamino pyridine in 1:1 molar ratio. The chemical structures of the Schiff base ligand and its metal complexes were confirmed by various spectroscopic studies like IR, UV-VIS, 1H NMR, TGA/DTA, Powder XRD, Molar conductance, and Photo luminescence. The free Schiff base ligand and its complexes have been tested for their antibacterial activity using disc diffusion method. From the biological studies, the complexes exhibit more activity than the ligand.
Metal-chelate Schiff base complexes have continued to play the role of one of the most important stereo chemical models in main group and transition metal coordination chemistry due to their preparative accessibility, diversity and structural variability(Garnovskii.,1993). Schiff bases have gained importance because of physiological and pharmacological activities associated with them. Schiff base metal complexes have ability to reversibly bind oxygen in epoxidation reactions [1], biological activity [2-4], catalytic activity in hydrogenation of olefins [5, 6] non-linear optical materials and [7] and photochromic properties [8]. The Schiff base complexes were also used as drugs and they possess a wide variety of antimicrobial activity against bacteria, fungi and it also inhibits the growth of certain type of tumours [9, 10]. The symmetric nature of a number of homo dinuclear transition metal derived metallobiosites and of the ability of the individual metal ions to have quite distinct roles in the functioning of the metalloenzyme concerned has led to a search for symmetrical dinucleating ligands which will give binuclear complexes capable of acting as models for the metallobiosites [11, 12]. 2, 6-Diamino-pyridine is a mediumproduction-volume chemical used as a pharmaceutical intermediate and a hair dye coupler in oxidation/permanent formulations. Although mutagenic activity has been reported, here in the present work we report the formation of Schiff base ligand from the condensation of 2, 6-diamino pyridine with benzil and the complexation with metal ions to form potentially active macrocyclic binuclear Schiff base metal complexes.
Synthesis of Ligand ((6E)-N2-((E)-2-(6-aminopyridin-2-ylimino)-1, 2-diphenylethyidine) pyridine-2, 6-diamine) The Schiff base ligand ((6E)-N2-((E)-2-(6-aminopyridin-2-ylimino)-1,2-diphenylethylidine)pyridine-2,6-diamine) were prepared by the drop wise addition of a solution of 2,6-diaminopyridine (0.22 g, 2 mmol) in ethanol (20 ml) to a stirred solution of benzil (0. 21 g, 1 mmol) in ethanol (20 ml). After the addition was completed, the mixture was condensed for 3 h at 900C. A brown precipitate solution was formed. The solution was kept for slow evaporation. The formed brown precipitate was filtered and washed with ethanol and then dried in air. Yield: 0.44 g (53%). Anal Calcd. For C24H20N6: C-73.45, H-5.14, and N-21.41. Found: C-73.38, H- 5.09, and N-21.40.
Synthesis of Schiff base metal complexes ([M2 (L) 2].4(OAc)) The macrocyclic binuclear Schiff base metal complexes (Cu(II), Ni(II), Co(II) and Mn(II)) were prepared by the condensation of 20 ml DMF solution of synthesized ligand (2mmol) adding to the constant stirring of 20 ml of ethanolic solution of metal salt (2 mmol M (where M = Cu2+, Ni2+, Co2+, Mn2+)) which was boiled for 3 hour under reflux. The coloured solution was formed. It was kept for slow evaporation and then collected the precipitate. It was filtered and washed with ethanol and then dried in vacuum.
The resultant macro cyclic Schiff metal complexes were colored powders, and stable for a long time in the open atmosphere. The analytical data and some of the physical properties of the Schiff base ligands and their binuclear metal complexes were summarized. All the metal complexes were sparingly soluble in general organic solvents, and soluble in DMF and DMSO, but insoluble in H2O, EtOH and MeOH. From the molar conductivity data, we clearly found the metal complexes were electrolytic in nature. The structural studies of the ligand and their complexes were done by spectroscopic methods.
Molar conductance The molar conductivity measurements commonly employed in the determination of the geometrical structure of inorganic compounds at infinite dilution. The molar conductance of binuclear Schiff base complexes was dissolved in DMSO and recorded (10-3M molar conductivity solution) at room temperature (Table 1). The complexes showed the range of molar conductance (127-134 ohm1 cm2mol1 ). From these values, we concluded that the complexes were electrolytic in nature. From the molar conductance, we concluded that the anions were outside the coordination sphere and not bonded to the metal ion therefore, these complexes may be formulated as [M2L2]4Z where, Z = acetate ion.
Table 1 Molar conductance and magnetic moment data of Schiff base binuclear metal complexes
S.No Compounds Solvent Molar conductance Type of electrolyte Magnetic moment Geometry
Λm (ohm1 cm2 mol-1 ) μ eff B.M
1. [Cu2(L)2]4(OCOCH3) DMSO 132 1 :2 electrolyte 1.74
2. [CO2(L)2]4(OCOCH3) DMSO 127 1 : 2 electrolyte 4.83
3. [Ni2(L)2]4(OCOCH3) DMSO 134 1 : 2 electrolyte 2.91
4. [Mn2(L)2]4(OCOCH3) DMSO 129 1 : 2 electrolyte 5.82 Octahedral
IR Spectra of the free ligand and their binuclear metal complexes Vibrational spectra provide valuable information regarding the nature of functional group attached to the metal ion in the complexes. The IR spectra of the complexes show very similar spectra to one another. These spectra indicates the replacement of Ѵ(NH2) and Ѵ(C=O) of the starting materials with Ѵ(C=N) which suggest the occurs of the condensation reaction between amine and carbonyl groups [13]. Selected vibration bands of ligands and their metal complexes are given in Table 2. From the IR spectral analysis, the assignment of the important bands was made and recorded. In order to give a conclusive idea about the structure of the metal complexes, the IR bands of metal complexes were compared with free Schiff base ligand. The appearance of a strong, broad band at 3177 cm−1 in the spectra of the free ligands was assigned to ν (NH2). The IR band was shifted in the region (3063–3198 cm -1 ) shows the involvement of primary amine nitrogen atom coordinate to the metal ion for all the Schiff base metal complexes (Ray et al., 2009) after the complexation. The appearance of the band at 1629 cm -1 which may be assigned to the azomethine group Ѵ(C=N) vibration, indicate the condensation of the amino group of 2,6-diamino pyridine with the carbonyl group of benzil and formation of the proposed Schiff base. The IR spectra of all metal complexes show significant changes compared to free Schiff base ligand. After Complexation, the positions of the Ѵ(C=N) were shifted in the range (1660-1667cm1 ) indicates the participation of the azomethine group in complex formation (Singh et al., 2010). The position of an N – atom of the azomethine group and group of the pyridine ring in coordination is further supported by the presence of new bands in the range from 470–495cm-1which is assignable to (M-N) vibration. From the spectroscopic behaviour of metal complexes of pyridine, after the complexation the ring deformation found at 797 cm-1 and 711 cm−1. It was clearly indicate that the free pyridine is shifted to higher frequencies [14], and the coordination takes place via the pyridine nitrogen, as previously reported for pyridine complexes [15]. Therefore, this shift is clearly indicates the participation of pyridine in complex formation. The appearance of band range from 1660 cm−1 to 1438 cm−1 were due to symmetric stretching frequency and asymmetric stretching frequency of acetate ion. This clearly indicates that the acetate ions were coordinated outside of the coordination sphere.
Table 2 IR spectral data (cm-1 ) of the Schiff base (L) and their binuclear metal complexes
1H NMR spectrum of ligand and its macro cyclic binuclear metal complexes The 1H NMR data of the Schiff base (L) and the metal complexes were recorded in DMSO-d6 (Table 3). Assignment of 1H NMR signals were made according to their reported results for 2,6-diaminopyridine and its complexes [16-20]. The 1H-NMR spectra of ligand and its metal complexes show different peaks in the range 6.99-7.94 ppm corresponding to H3, H4, and H5 protons indicate unsymmetrical binding of the ligand to M (II) complexes. In the 1H NMR spectrum of M (II) complexes, singlet signal of the pyridine-NH2 (s, 3.1 to 3.7 ppm) and multiplet signals of aromatic protons (m, 6.72 to 7.94 ppm) of Schiff base (L) shifted compared to the starting material which suggests coordination through nitrogen atom of the azomethine group. For the metal complexes, a single sharp signal is appeared (region from 2.1 – 2.6 ppm) in the 1H NMR spectrum, suggest that the acetate ion is present in the outside coordination sphere of the metal complexes.
Table 3 1H NMR spectrum of ligand and its macro cyclic binuclear metal complexes
Electronic absorption spectra and magnetic moment measurements The electronic spectrum of the Schiff base ligand in DMSO (Table 4), the absorption band observed at 274 nm were assigned to π→π* transition and the band at 386 nm were assigned due to n→π* transition associated with the azomethine chromophore (-C=N).The absorption bands of the complexes are shift to longer wave numbers compared to that of the ligand [21]. For [M2 (L) 2]4(OCOCH3) complexes, the electronic absorption band occurs at 468-474 nm due to charge transfer from ligand to metal ion (LMCT). The obtained Cu (II) complexes exhibits a band at 652 nm assigned to 2Eg → 2T2g transition which is in conformity with octahedral geometry around the Cu (II) ion (Patil et al., 2010; Lever, 1968). The obtained magnetic moment value (µeff) for Cu(II)complex is 1.89 BM indicating that magnetic exchange occurs between the two copper sites and also supports octahedral geometry of Cu(II) complex [22]. The electronic absorption spectra of Co (II) complexes showed a band at 648 nm corresponding to 4T1g(F)→4A2g(F) transition and also the obtained magnetic moment value is 4.84 BM which confirm the octahedral geometry of the complex [23]. For the Ni (II) complex, it has the 3.06 BM magnetic moment value and the electronic spectrum showed a band at 645nm corresponding to 3A2g (F) →3T1g (F) transition which is consistent with the octahedral geometry of the complex. The Mn (II) binuclear complex shows bands at 633 nm corresponds to 6A1g→4T2g (4G) transitions and 5.82 BM magnetic value were compatible to an octahedral geometry of the ligand around manganese (II) ion [24].
Table 4 Electronic Spectral data of Schiff base ligand and their macro cyclic binuclear metal complexes
Compounds Electronic absorption spectra (nm) Magnetic moment Geometry of
π→π* n→π* L→M d-d value(µeff)BM the complex
(nm) (nm) (nm) (nm)
C12H12N6 (ligand) 274 342 – – –
[Cu2(L)2]4(OCOCH3) 268 340 474 652 1.89
[CO2(L)2]4(OCOCH3) 266 338 468 648 4.84
[Ni2(L)2]4(OCOCH3) 256 339 471 645 3.06 Octahedral
[Mn2(L)2]4(OCOCH3) 261 341 474 636 5.82
TGA and DTA studies By using TGA and DTA analysis the thermal stability of the complexes were explained. The observation thermogram and curves (Table 5, Figure 2) were obtained at a heating rate of 100C/min over a temperature range of 40–7300C. The complex was stable up to 1600C and its decomposition started at this temperature. In the thermal decomposition process of the Cu (II) complex proceeds two steps of the mass losses corresponded to acetate, and NH2 leaving in the first, and second stages of the decomposition. The decomposition of the Cu (II) are irreversible. The Cobalt complexes were stable up to 2000C and its decomposition started at this temperature. The Cobalt (II) complex was decomposed in two steps with the temperature ranges from 200-470˚C corresponding to the loss of acetate and NH2 respectively. The Ni (II) complexes were stable up to 180oC and its decomposition started at this temperature. In the decomposition process of the Ni(II) complex, the estimated mass loss of the first step 6.85(6.91) corresponded to the loss of four acetate group and the second stage the liberation of four NH2 unit respectively, shown in table 6. The decomposition of the Nickel complex was irreversible. The thermal decomposition of the Manganese complex was stable up to 1700C and its decomposition started at this temperature. Thereafter, they start the decomposition process of the Mn (II) complex and weight loss observed in the temperature range 170-4600C, the mass loses corresponded to four acetate and four NH2 leaving in the first and second stages of the decomposition. The decomposition of the Mn (II) complexes are irreversible. The amount of acetate and NH2 groups stoichiometrically corresponding to the weight losses are given in the proposal chemical formulas of complexes.
Powder XRD Analysis Synthesized Schiff base metal (II) complexes were subjected to Powder X-ray diffractograms in the range (2ɵ = 10– 600 ) were shown in (Figure 3). Among the metal complexes Ni (II) complex shows well defined crystalline sharp peak which indicate the sample were crystalline nature. The appearance of crystallinity in the Schiff base metal complexes is due to the inherent crystalline nature of the metallic compounds. The average grain size (dXRD) of the Ni (II) complex is 32 nm which was calculated by using Scherer’s formula (Dhanaraj and Nair, 2009a,b) suggesting that the Ni(II) complex are nanocrystalline.
Table 5 Thermo gravimetric data of metal complexes
Figure 2 DTA/TGA Curve for metal complexes
Figure 3 Powder X-ray diffractogram for Ni complex
Fluorescence spectra The Schiff base and its binuclear metal complexes were analysed by the photoluminescence emission spectra (Figure 4) and recorded in DMSO at room temperature. Comparing with Schiff base ligand and its macro cyclic binuclear metal complexes, the metal complexes have strong fluorescence intensity than Schiff base. Among the metal complexes the Co (II) complex exhibited a strong fluorescence emission at 400 nm (Flourescence intensity 713) with excitation at 269 nm. The quenching of metal (II) complexes indicates that the ligand has a less potential photo active than metal (II) complexes.
Figure 4 Fluorescence spectra for all metal complexes
In vitro antimicrobial activity of Schiff base ligand and their metal complexes By using broth micro dilution procedures, the Schiff base ligand and their metal complexes were screened separately against for two Gram positive bacteria (Staphylococcus aureus and B. Subtilis), two Gram negative bacteria (E. Coli and S. typhi) and the fungi (A. fumigatus) for their antimicrobial activity. When the activity of Schiff base ligand and their metal complexes were increased by increasing the antimicrobial screening concentration (Table 6, Figure 5), because the concentration plays an important role in the zone of inhibition and the chelated metal complexes deactivate the various cellular enzyme [25]. Metal complexes show considerable antimicrobial activity even at low concentration and also more toxicity towards Gram-positive strains, Gram-negative strains and fungi compare with Schiff base ligand. The antimicrobial data shows that the copper complex noticed an excellent activity against bacteria and fungi than other metal complexes. The different antimicrobial activity of different metal complexes depends on the impermeability of the cell or the difference in ribosomes in microbial cell (Sengupta et al., 1998).
Table 6 Antimicrobial activity of Schiff base ligand and their metal complexes
Figure 5 Anti-Bacterial Activities of the Schiff Base and Its Binuclear Metal Complexes against Gram positive and negative Bacterias
Conclusion Macrocyclic binuclear metal (II) complexes was synthesized by using condensation method of a novel Schiff base ligand derived from 2, 6-diamino pyridine and benzil. The data which have been the physico chemical and spectral studies provides excellent structure and chemical composition of Schiff base and its metal complexes. The electronic absorption spectra, IR spectra and magnetic moment value reveals that the metal complexes were octahedral geometry and the Schiff base coordinated through six nitrogen atoms of azomethine group and pyridine ring. Powder XRD data reveals that the Ni(II) complex was nano crystalline structure. Based on the photo luminescence studies, we have confirmed the metal complexes were more potential photo active than Schiff base. The in vitro antimicrobial studies of metal (II) complexes showed better activity than Schiff base.
References
[1] Viswanathamurthi P, Natarajan K, Synth.React.Inorg.Met.-Org.Chem 2006; 36:415-418.
[2] Ren S, Wang R, Komastu K, Krause P.B, Zyrianov Y, Mckenna C. E, Csipke C, TokesZ. A, Lien E. J, J. Med. Chem 2002; 45: 410

 Jackie Caynon, pictured right, a lawyer with more than 18 years of health law experience, and partner and co-chair of Mirick O’Connell’s Health Law Group, told FierceHealthcare in an exclusive interview that the answer has to come from each hospital’s unique risk management assessment.
Jackie Caynon, pictured right, a lawyer with more than 18 years of health law experience, and partner and co-chair of Mirick O’Connell’s Health Law Group, told FierceHealthcare in an exclusive interview that the answer has to come from each hospital’s unique risk management assessment.