Precision Medicine Market to surpass USD 96.6 Bn by 2024, study by Global Market Insights, Inc.
Reporter: Aviva Lev-Ari, PhD, RN
Precision Medicine Market to surpass USD 96.6 Bn by 2024
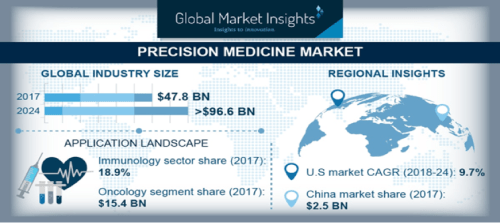
The Precision Medicine Market is set to grow from its current market value of more than $47.8 billion to over $96.6 billion by 2024; as reported in the latest study by Global Market Insights, Inc.
Advancements in cancer biology will ensure development in gene sequencing technique and other platforms available for cancer treatment. Furthermore, rising development in field of bioinformatics that support gene sequencing to initiate formulations of precision medicines will enhance the precision medicine market growth in foreseeable future.
Increase in adoption rate of gene therapy in developed economies with relatively high-income level will further enhance the precision medicine market growth. Rising incidences of genetic disorders has positively influenced the business growth. According to WHO, sickle cell anemia affects around millions of people globally every year. Along with sickle cell anemia, there are many other genetic diseases such as thalassemia, hemophilia and others that are prevalent globally. Moreover, increase in research and development activities that boosts the production and manufacturing of precision medicine in emerging countries such as Africa and Brazil will augment the industry growth in future. However, high cost of precision medicines may restrain the market growth.
Precision Medicine Market
Bioinformatics segment was valued at USD 7.4 billion in 2017 and is expected to have considerable growth during the forecast timeframe. Bioinformatics is composed of information that includes data for gene sequencing, amino acid sequences of proteins and structural classification of protein enabling efficient drug development process for treating diseases such as cancer at molecular level. Additionally, bioinformatics is also used to determine the treatment for hereditary metabolic disorders that will drive the segmental growth in forthcoming years.
Oncology segment will experience 11.1% CAGR during the forecast timeframe. High segmental growth can be attributed to the increasing prevalence of cancer globally. According to Globocan 2018, the number of new cancer cases are estimated to be 18.1 million cases. Development of target novel therapies and precision medicines has drastically reduced the mortality rates and hence, high demand for precision medicine in cancer treatment will drive the segmental growth in foreseeable future.
Pharmaceutical companies segment of precision medicine market accounted for 37.1% in 2017 owing to the increasing R&D efforts for drug development activities. Pharmaceutical companies are currently trying to develop precision medicines for rare genetic diseases. According to Global genes, there are around 7,000 rare genetic diseases discovered.
U.S. precision market will experience 9.7% CAGR during the forecast timeframe. Enormous market growth will be due to increasing cases of rare genetic diseases. Furthermore, certain initiatives undertaken by the regulatory bodies have also positively impacted availability of precision medicines.
U.S. Precision Medicine Market Size, By Technology, 2017 & 2024 (USD Million)
Germany precision medicine market was valued at USD 2.6 billion in 2017 and high segmental growth will be due to the growing pediatric as well as adult population susceptible to metabolic diseases such as diabetes. According to NCBI, prevalence of Type 2 diabetes is high in the German population. Researchers and scientists have geared up efforts to develop precision medicines that are altered according to patient’s genome ensuring efficient treatment of genetic disorders.
Prominent industry players operational in the precision medicines market include Biocrates Life Sciences AG, Eagle Genomics Ltd, Ferrer inCode, Intomics, Laboratory Corporation of America Holdings, NanoString Technologies, Novartis, Pfizer, Qiagen, Quest Diagnostics, F. Hoffmann-La Roche, Silicon Biosystems, Tepnel Pharma Services, Teva Pharmaceuticals.
Source: https://www.gminsights.com/industry-analysis/precision-medicine-market
SOURCE for Request to Publish in pharmaceuticalintelligence.com
From: Shankar Khatkale <shankar.k@gminsights.com>
Date: Tuesday, November 27, 2018 at 6:48 AM
To: Aviva Lev-Ari <AvivaLev-Ari@alum.berkeley.edu>
Subject: Article Submission Request
Dear Editor,
An industry news titled ‘Precision Medicine Market Size worth $96.6bn by 2024’ by Global Market Insights is relevant to your esteemed website https://pharmaceuticalintelligence.com. This email is a suggestion to publish this news (content attached in word format or can be picked from link mentioned below) on your website with an objective to share the information with your audiences.
News Link: https://www.gminsights.com/pressrelease/precision-medicine-market
Looking forward to hear from you.
On behalf of and as instructed by Global Market Insights, Inc.
Best Regards,
Shankar Khatkale |SEO Executive