Failed pain relief drug candidate clinical trial
Larry H. Bernstein, MD, FCAP, Curator
LPBI
What was the drug in Clinical Trial Tragedy In France Jan 2016 |

BIA 10-2474
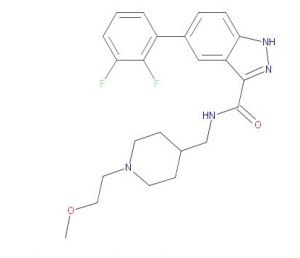
3-(1-(cyclohexyl(methyl)carbamoyl)-1H-imidazol-4-yl)pyridine 1-oxide
BIA 10-2474 is an experimental fatty acid amide hydrolase inhibitor[1] developed by the Portuguese pharmaceutical company Bial-Portela & Ca. SA. The drug was developed to relieve pain,[2][3] to ease mood and anxiety problems, and to improve movement coordination linked to neurodegenerative illnesses.[4] It interacts with the humanendocannabinoid system.[5][6] It has been linked to severe adverse events affecting 5 patients in a drug trial in Rennes, France, and at least one death, in January 2016.[7]
French newspaper Le Figaro has obtained Bial study protocol documents listing the the chemical name of BIA-10-2474 as 3-(1-(cyclohexyl(methyl)carbamoyl)-1H-imidazol-4-yl)pyridine 1-oxide.[8] A Bial news release described BIA-10-2474 as “a long-acting inhibitor of FAAH”.[9]
Fatty acid amide hydrolase (FAAH) is an enzyme which degrades endocannabinoid neurotransmitters like anandamide,[10] which relieves pain and can affect eating and sleep patterns.[11][12] FAAH inhibitors have been proposed for a range of nervous-system disorders including anxiety, alcoholism, pain and nausea.
The Portuguese pharmaceutical company Bial holds several patents on FAAH enzyme inhibitors.[12][13][14][15]
No target organ was identified during toxicology studies and few adverse clinical findings were observed at the highest dose tested. For the single ascending dose part [of the clinical trial], a starting dose of 0.25 mg was judged to be safe for a first-in-human administration.[8]
The protocol defines no starting dose for the multi-dose treatment groups, noting that this will be based on the outcome of the single dose portion of the trial (an approach known as adaptive trial design). The authors note that nonetheless, the starting dose will not exceed 33% of the maximum tolerated dose (MTD) identified in the single dose groups (or 33% of the maximum administered dose if the MTD is not reached).[8]
In July 2015 Biotrial, a contract research organization, began testing the drug in a human phase one clinical trial for the manufacturer. The study was approved by French regulatory authority, the Agence Nationale de Sécurité du Médicament (ANSM), on June 26, 2015, and by the Brest regional ethics committee on July 3, 2015.[20] The trial commenced on July 9, 2015,[21] in the city of Rennes, and recruited 128 healthy volunteers, both men and women aged 18 to 55. According to French authorities, the study employed a three-stage design with 90 of the volunteers having received the drug during the first two stages of the trial, with no serious adverse events being reported .[17][20] Participants of the study were to receive €1,900 and, in turn, asked to stay at Biotrial’s facility for two weeks during which time they would take the drug for ten days and undergo tests.[22]
In the third stage of the trial evaluating multiple doses, six male volunteers received doses by mouth, starting on 7 January 2016. The first volunteer was hospitalized at theRennes University Hospital on January 10, became brain dead,[17][23][24][25] and died on January 17.[26] The other five men in the same dosage group were also hospitalized, in the period of January 10 through January 13[27] four of them suffering injuries including deep hemorrhagic and necrotic lesions seen on brain MRI.[7] The six men who were hospitalised were the group which received the highest dose.[26] A neurologist at the University of Rennes Hospital Center, Professor Pierre-Gilles Edan, stated in a press conference with the French Minister for Health, that 3 of the 4 men who were displaying neurological symptoms “already have a severe enough clinical picture to fear that even in the best situation there will be an irreversible handicap” and were being given corticosteroids to control the inflammation.[27] The sixth man from the group was not showing adverse effects but had been hospitalized for observation.[25][28][29] Biotrial stopped the experiment on January 11, 2016.[4]
Le Figaro posted a 96-page clinical study protocol for BIA 10-2474 that the French newspaper procured from an unnamed source.
According to the document, BIA 10-2474 is 3-(1-(cyclohexyl(methyl)carbamoyl)-1H-imidazol-4-yl)pyridine 1-oxide.
BIA 10-2474 “is designed to act as a long-active and reversible inhibitor of brain and peripheral FAAH,” notes the protocol. The compound “increases anandamide levels in the central nervous system and in peripheral tissues.”
The clinical trial protocol also notes that the company tested BIA 10-2474 on mice, rats, dogs, and monkeys for effects on the heart, kidneys, and gastrointestinal tract, among other pharmacological and toxicological evaluations.
The clinical trial, conducted by the company Biotrial on behalf of the Portuguese pharmaceutical firm Bial, was evaluating a pain relief drug candidate called BIA 10-2474 that inhibits fatty acid amide hydrolase (FAAH) enzymes. Blocking these enzymes prevents them from breaking down cannabinoids in the brain, a family of compounds that includes the euphoria-inducing neurotransmitter anandamide and Δ9-tetrahydrocannabinol, the major psychoactive component of marijuana.
Phase I clinical trials are conducted to check a drug candidate’s safety profile in healthy, paid volunteers. In this case, the drug caused hemorrhagic and necrotic brain lesions in five out of six men in a group who received the highest doses of the drug, said Gilles Edan, a neurologist at the University Hospital Center of Rennes.
The French health minister has stated the drug acted on natural receptors found in the body known as endocannibinoids, which regulate mood and appetite. It did not contain cannabis or anything derived from it, as was originally reported. All six trial participants were administered the doses simultaneously.
The trial was being performed at Biotrial, a French-based firm that was formed in 1989 and has conducted thousands of trials. A message on the company’s website stated that they are working with health authorities to understand the cause of the accident, while extending thoughts to the patients and their families. Bial has disclosed the drug was a FAAH (fatty acid amide hydrolase) inhibitor, which is an enzyme produced in the brain and elsewhere that breaks down neurotransmitters called endocannabinoids. Two scientists from the Nottingham Medical School who have worked with FAAH tried over the weekend to try and identify the drug by examining a list of drugs Bial currently has in its pipeline. They believe the culprit is one identified by the codename BIA 10-2474.
While safety issues like this are rare, they are not unheard of. In 2006, a clinical trial in London left six men ill. All were taking part in a study testing a drug designed to fight auto-immune disease and leukemia. Within hours of taking the drug TGN1412, all experienced a serious reaction, were admitted to intensive care, and had to be treated for organ failure.
The Duff Report, written in response to the TGN1412 trial, noted the medicine should have been tested in one person at a time. It also helped to put additional safety measures in place. The Medicines and Health Products Regulatory Agency (MHRA) now requires committees to look at pre-clinical data to determine the proper initial dose, and rules are in place to stop the trial if unintended reactions occur.
Other pharmaceutical companies, including Merck, Pfizer, Johnson & Johnson, Sanofiand Vernalis, have previously taken other FAAH inhibitors into clinical trials without experiencing such adverse events (e.g. respectively, MK-4409,[35][36] PF-04457845,JNJ-42165279,[37] SSR411298 and V158866.[38][39] Related enzyme inhibitor compounds such as URB-597 and LY-2183240 have been sold illicitly as designer drugs,[40][41] all without reports of this type of toxicity emerging, so the mechanism of the toxicity observed with BIA 10-2474 remains poorly understood.
Clinical Trial Tragedy, France, Jan 2016, PHASE 1 | Categories: Uncategorized | URL:http://wp.me/p38LX5-4ut