Curator: Aviva Lev-Ari, PhD, RN
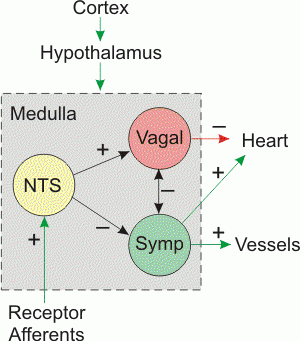
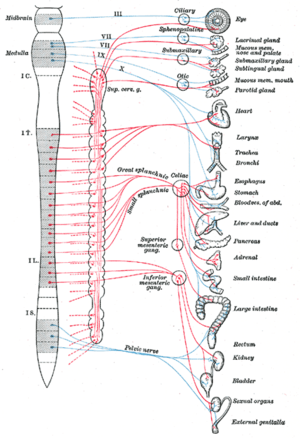
Intravascular Stimulation/Ablation of Autonomics are procedures, aka Renal Arterial Denervation, that since 1999 has greater promise for treatment of a diseases assumed to be related to an imbalance of autonomic tone. Dr. Scherlag prefers the name “cardiac sympathetic denervation”. As he has outlined, the ablation of autonomics in the renal artery has more of an effect on the heart than it does on the kidneys. I do agree that the procedure has a cardiac effect obtained by autonomic modulation in essence.
http://pharmaceuticalintelligence.com/2012/09/02/intravascular-stimulation-of-autonomics-a-letter-from-dr-michael-scherlag/
Pioneering work in this field, the first to stimulate/ablate autonomic nerves effecting the heart from the intravascular space was accomplished by Dr. Scherlag and others. The greatest accomplishment was “Method and apparatus for transvascular treatment of tachycardia and fibrillation. US Patent 6,292,695. Filed June 17, 1999.
Dr. Scherlag writes, [T]he possibility that parasympathetic or sympathetic nerves running on blood vessels could be stimulated or ablated from inside the vasculature was initially demonstrated using basket electrode catheters in a series of experimental studies (1-6) and formally patented in 1999 (7).
Treatment of Hypertension by renal arterial denervation from inside the vasculature using a basket electrode catheters by ultra sound or by RF energy is fully documented in my post Treatment of Refractory Hypertension via Percutaneous Renal Denervation
http://pharmaceuticalintelligence.com/2012/06/13/treatment-of-refractory-hypertension-via-percutaneous-renal-denervation/
Clinical Trials and the Ecosystem of all leading manufacturers of Medical Devices for Renal Arterial Denervation, including videos for demonstration of the procedure are presented in my post, link above.
Benefits and New Indications for Usage of Intravascular Stimulation/Ablation of Autonomics
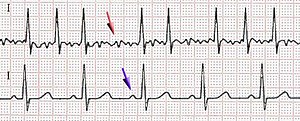
1. Reduction in Heart Rate and Heart Rate Variability
Dr. Scherlag experiments noted changes in heart rate which have also been reported in SYMPLICITY HTN-1 and SYMPLICITY HTN-2 (8-9). The SYMPLICITY HTN-2 study demonstrated profound bradycardia in 13% of patients that was treated with atropine.
The intra-procedure effect on heart rate during renal artery denervation documented in the SYMPLICITY trials is also manifest long term by measuring heart rate variability (10). Indeed, cardiac effects would be expected with autonomic modulation. Besides the two example above showing that cardiac sympathetic denervation effects heart rate, there are many more that are just beginning to be reported in the literature.
These articles shows the effects of renal denervation on heart rate.
http://www.ncbi.nlm.nih.gov/pubmed/1735574
http://www.ncbi.nlm.nih.gov/pubmed/8777835
A Cleveland Clinic review article states: “Additionally, the resting heart rate was lower and heart rate recovery after exercise improved after the procedure, particularly in patients without diabetes.”
http://www.ccjm.org/content/79/7/501.full
2. Renal Sympathetic Denervation lowers Atrial Fibrillation
This article discusses the effect of renal sympathetic denervation on atrial fibrillation.
http://www.ncbi.nlm.nih.gov/pubmed/22585944
3. Regression of Left Ventricular Hypertrophy, Increase in Ejection Fraction (EF) and improved Diastolic Dysfunction
“Brandt reported regression of left ventricular hypertrophy and significantly improved cardiac functional parameters, including increase in ejection fraction and improved diastolic dysfunction, in a study of 46 patients who underwent renal denervation. This findings suggests a potential beneficial effect on cardiac remodeling.” (Brandt MC, Mahfoud F, Reda S, et al. Renal sympathetic denervation reduces left ventricular hypertrophy and improves cardiac function in patients with resistant hypertension. J Am Coll Cardiol 2012; 59:901–909)
4. Reduction in Ventricular Tachyarrhythmias (VT)
“Ukena reported reduction in ventricular tachyarrhythmias in two patients with congestive heart failure who had therapy-resistant electrical storm.” (Ukena C, Bauer A, Mahfoud F, et al. Renal sympathetic denervation for treatment of electrical storm: first-inman experience. Clin Res Cardiol 2012; 101:63–67)
5. Intravascular Stimulation of Autonomics Effects on Heart Failure
The most recent data from Europe shows the following effects on heart failure:
http://www.eurekalert.org/pub_releases/2012-08/esoc-rdg082712.php
http://www.theheart.org/article/1364267.do
Dr. Scherlag, writes, [N]early ten examples of the effects of “CARDIAC SYMPATHETIC DENERVATION” and what are the effects on the kidney?
No change in GFR. No change in creatinine.
Medical Debate on the Procedure – The candidates are hypertensive patients receiving blood-pressure-lowering medication that are truly “resistant.”
The Symplicity system (Medtronic) is the far-and-away front runner, having demonstrated average office-based BP drops of 32/12 mm Hg at six months in the SYMPLICITY HTN 2 trial, as reported by heartwire, with 84% of patients having had a >10-mm-Hg drop in systolic blood pressure from baseline.
Upwards of 20 other companies, according to Dr Ron Waksman (Washington Hospital, DC), are busy developing competing systems, some of which were featured in a EuroPCR session devoted to emerging technologies in May 2012 in Paris.
Leading this pack is St Jude’s EnligHTN system, which received CE Mark on the opening day of the meeting. Dr Stephen Worthley (Royal Adelaide Hospital, Australia) presented 30-day results in 47 resistant-hypertension patients treated with the multielectrode, RF-ablation-based system. Mean office BP changes at one month in EnligHTN 1 were -28 systolic and -10 diastolic (p<0.0001 from baseline), with 78% of patients having systolic BP drops of >10 mm Hg.
https://www.massdevice.com/news/europcr-st-judes-enlightn-lowers-blood-pressure-faster-rival-systems
In terms of safety, no serious complications were seen in the renal artery or at the access site in the EnligHTN study; minor procedure-related events included four hematomas, three vasovagal responses to sheath removal, and two postprocedure transient bradycardias.
Other devices featured in the session included a second RF-energy system and two ultrasound systems, see below technology description by supplier.
The risk of cardiovascular death doubles with every 20 point increase in systolic blood pressure, so an average blood pressure reduction of 28 points is quite significant and demonstrates just how effective the technology is. Principal investigator Prof. Stephen Worthley said in prepared remarks. “From other clinical trials studying the impact of renal denervation we have learned that blood pressure continues to be reduced over time, so I would not be surprised to see this trend continue and see an even greater benefit for patients.” St. Jude’s study included 47 patients with high blood pressure that wasn’t managed with drug therapy. Participants had an average of 176/96 mmHg baseline blood pressure, despite taking multiple medications, before the denervation procedure and an average of 148/87 mmHg after. More than 40% had systolic rates below 140 mmHg.
http://investors.sjm.com/phoenix.zhtml?c=73836&p=irol-newsArticle&ID=1695802
Interventionalists who spoke with heartwire were unvaryingly excited about the potential of renal denervation, with some caveats.
“You need enthusiasm to develop new things, and in hypertension we haven’t seen an innovation in decades,” Dr Thomas Lüscher (University Hospital Zürich, Switzerland) told heartwire. “So just the possibility that you would be able to have a persistent treatment effect by a procedure that helps severe hypertension patients and maybe in the future even the option to cure hypertension is very exciting indeed. But I agree it’s a dream at this point. I think we need the SYMPLICITY HTN 3 trial, which hopefully will confirm what the other studies have shown.”
Now enrolling at as many as 90 US centers, SYMPLICITY HTN 3, Lüscher pointed out, has design characteristics addressing two concerns with the earlier trials, namely a sham procedure for the control group and ambulatory blood-pressure monitoring in all patients.
During the same emerging-technologies session, Lüscher explored the albeit-scant data supporting a role for renal denervation in other conditions: everything from metabolic syndrome and obstructive sleep apnea to heart failure, atrial fibrillation, and polycystic-ovary syndrome.
But his counterpoint, Dr Jean Renkin (UCL St Luc University Hospital, Brussels, Belgium), was skeptical, pointing to the myriad unanswered questions with the technology.
“Currently, reasonably solid data are available only for patients with hypertension resistant to pharmacotherapy, which cannot necessarily be extrapolated to other forms of hypertension or conditions referred to [by Dr Lüscher]. However, at this point in time, no clouds have appeared in the sky, so let us dream on.”
Dr Renkin had one staggering number for the audience to consider: of 5000 patients who have undergone renal denervation, only 250 were actually treated as part of clinical studies. While no device has US approval, five denervation systems already hold CE Mark in Europe and are being used with increasing frequency.
Treating the Truly Medication Treatment “Resistant”
For a comprehensive presentation of Triple Antihypertensive Combination Therapy Significantly Lowers Blood Pressure in Hard-to-Treat Patients with Hypertension and Diabetes, refer to
http://pharmaceuticalintelligence.com/2012/05/29/445/
Another talking point is the proportion of patients who are truly “resistant.” The number agreed on by Lüscher, Waksman, and session comoderator Dr Robert Whitbourn (St Vincent’s Hospital, Fitzroy, Australia) was that just 3% of all hypertensive patients receiving blood-pressure-lowering medication are truly “resistant.” Numbers as high as 30% have been suggested in other reports, he noted.
“Interestingly, when we’ve been involved in various trials, every cardiologist says they have hundreds of these patients, but when we actually go to get them, no one actually has any,” Whitbourn quipped. “I think it should be a sobering thought—the numbers are actually quite small.”
Dr William Wijns (Cardiovascular Center Aalst, Belgium), also speaking with heartwire, agreed that the subset was “small” but argued it was “still big numbers, millions of people,” and “a massive unmet need.”
Waksman, insisting he was “excited” by what he called “robust reductions in blood pressure,” nevertheless urged eager interventionalists to work with hypertension experts and resist the urge “to jump on patients before we truly verify that they are resistant to medical treatment.”
In the vast majority of people even for whom renal denervation is appropriate, it “won’t be a cure,” Waksman said. “Most of these patients will have to continue on medical treatment—this is not replacing medical treatment, it is just getting [patients] more in control.”
http://www.theheart.org/article/1402321/print.do
REFERENCES
1. Schauerte P, Scherlag BJ, Scherlag MA, Goli S, Jackman WM, Lazzara R. Transvenous parasympathetic cardiac nerve stimulation: an approach for stable sinus rate control. J Electrophysiol. 1999 Nov;10(11):1517-24.
2. Schauerte P, Scherlag BJ, Scherlag MA, Goli S, Jackman WM, Lazzara R. Ventricular rate control during atrial fibrillation by cardiac parasympathetic nerve stimulation: a transvenous approach. J Am Coll Cardiol. 1999 Dec;34(7):2043-50.
3. Schauerte P, Scherlag BJ, Pitha J, Scherlag MA, Reynolds D, Lazzara R, Jackman WM. Catheter ablation of cardiac autonomic nerves for prevention of vagal atrial fibrillation. Circulation. 2000 Nov 28;102(22):2774-80.
4. Scherlag MA, Scherlag BJ, Yamanashi W, Schauerte P, Goli S, Jackman WM, Reynolds D, Lazzara R. Endovascular neural stimulation via a novel basket electrode catheter: comparison of electrode configurations. J Interv Card Electrophysiol. 2000 Apr;4(1):219-24.
5. Scherlag BJ, Yamanashi WS, Schauerte P, Scherlag M, Sun YX, Hou Y, Jackman WM, Lazzara R. Endovascular stimulation within the left pulmonary artery to induce slowing of heart rate and paroxysmal atrial fibrillation. Cardiovasc Res. 2002 May; 54(2):470-5.
6. Hasdemir C, Scherlag BJ, Yamanashi WS, Lazzara R, Jackman WM. Endovascular stimulation of autonomic neural elements in the superior vena cava using a flexible loop catheter. Jpn Heart J. 2003 May;44(3):417-27.
7. Webster W Jr, Scherlag BJ, Scherlag MA, Schauerte P. Method and apparatus for transvascular treatment of tachycardia and fibrillation. US Patent 6,292,695. Filed June 17, 1999.
8. Krum H, Schlaich M, Whitbourn R, Sobotka PA, Sadowski J, Bartus K, Kapelak B, Walton A, Sievert H, Thambar S, Abraham WT, Esler M. Catheter-based renal sympathetic denervation for resistant hypertension: a multicentre safety and proof-of-principle cohort study. Lancet. 2009;373(9671):1275-1281.
9. Symplicity HTN-2 Investigators. Renal sympathetic denervation in patients with treatment-resistant hypertension (The Symplicity HTN-2 Trial): a randomised controlled trial. Lancet. 2010;376:1903-1909.
10. Frank Himmel MD, Joachim Weil MD, Michael Reppel MD, Kai Mortensen MD, Klaas Franzen, Leidinger Ansgar MD, Heribert Schunkert MD, Frank Bode MD. Improved Heart Rate Dynamics in Patients Undergoing Percutaneous Renal Denervation. Letter to the Editor. JCH. 31 MAY 2012.1751-7176.
Like this:
Like Loading...
Read Full Post »



 The MIT-BIH Polysomnographic Database is a collection of recordings of multiple physiologic signals during sleep. Subjects were monitored in Boston’s Beth Israel Hospital Sleep Laboratory for evaluation of chronic obstructive sleep apnea syndrome, and to test the effects of constant positive airway pressure (CPAP), a standard therapeutic intervention that usually prevents or substantially reduces airway obstruction in these subjects. The database contains over 80 hours’ worth of four-, six-, and seven-channel polysomnographic recordings, each with an ECG signal annotated beat-by-beat, and EEG and respiration signals annotated with respect to sleep stages and apnea. For further information, see
The MIT-BIH Polysomnographic Database is a collection of recordings of multiple physiologic signals during sleep. Subjects were monitored in Boston’s Beth Israel Hospital Sleep Laboratory for evaluation of chronic obstructive sleep apnea syndrome, and to test the effects of constant positive airway pressure (CPAP), a standard therapeutic intervention that usually prevents or substantially reduces airway obstruction in these subjects. The database contains over 80 hours’ worth of four-, six-, and seven-channel polysomnographic recordings, each with an ECG signal annotated beat-by-beat, and EEG and respiration signals annotated with respect to sleep stages and apnea. For further information, see 







 World-class rowing champion Frederick Schoch
World-class rowing champion Frederick Schoch
 Mark E. Josephson, MD
Mark E. Josephson, MD