Augmentation of the ONTOLOGY of the 3D Printing Research
Curator: Larry H. Bernstein, MD, FCAP
Encore: Research Allows for 3D Printed Augmentation of Everyday Objects
BY BRIAN KRASSENSTEIN
http://3dprint.com/87595/encore-3d-print-augment/
If you have a 3D printer then you are likely overwhelmed by the sheer number of possible objects you can find online to print out. At the same time, the technology is somewhat limited, unless you have professional CAD skills or are incredibly creative. What, for instance, would you do if you wanted to take an everyday item such as a hot glue gun and print an attached stand for it, or turn your child’s favorite action figure into a magnet?
Four researchers at Carnegie Mellon University, Xiang ‘Anthony’ Chen, Stelian Coros, Jennifer Mankoff and Scott E. Hudson, believe that they can change this via a new WebGL-based tool under development called Encore.
Funded by the National Science Foundation under grant NSF IIS 1217929, Encore is a multifaceted tool which enables three different techniques to augment already existing objects. The researchers call these three techniques Print-Over, Print-to-Affix and Print-Through, all of which allow for the adherence of newly 3D printed attachments to other objects. Before we get into what each of these three techniques involves, one first should understand the computational pipeline involved in designing these attachments.
First a user is required to design a basic attachment, or perhaps use a design that they’d like to iterate upon. Once they have a basic model of their desired attachment the Encore system will geometrically analyze the model along with a 3D scan of a target object, that the attachment will adhere to, in order to determine its printability while also deciding if it will be durable enough and usable once attached to the target object. Next comes the interactive exploration phase, where the tool will visualize and explore various areas where the attachment can be affixed to the target object. The tool will also adjust the design of the attachment, if required, to better fit the target object. Once this is all settled, it’s now time for Encore to generate a model which not only will include the attachment itself, but also any connecting structures and even supports to hold the target item or attachment in place. Below you will find the three techniques that the Encore tool can use to attach 3D printed items to a target object:
Print-Over
This technique, in my opinion, is one of the coolest, as it allows for the printing of an attachment directly on a given object. Once Encore establishes the parameters required and sizes up the model to fit the target object, it will automatically tell the printer to print supports to hold the target item in place. Once the target item is in place, Encore will then tell the printer to begin printing the attachment in a particular spot on the target item, based on its geometric analysis of that object.
“It is also important to ensure that the existing object will not impede the motion of the print head while the attachment is being printed,” warn the researchers.
An example used by the researchers for this technique was a magnet holder which they directly attached to a teddy bear figurine. They also printed an LED light onto a 9V battery by first placing a small amount of glue on the battery prior to beginning to print on top.
Print-to-Affix
This approach is similar to the Print-Over technique, only that instead of printing an attachment directly onto the target object, Encore will analyze the geometry of the target object prior to printing in order to create an attachment which will fit perfectly on that object via glue, straps (zip-ties) or even snaps. Once the attachment is printed the user can then use hot-glue or another adhesive or attachment mechanism to affix the printed object onto the target.
Print-Through
This technique is perfect for items which you don’t want to physically attach, but instead can connect to an object. For instance a tag on the loop of a pair of scissors or a charm onto a bracelet. This process requires that the printer be paused while a user manually places the target object within the print field.
“Print-through has aesthetic qualities that distinguish it from print-to-affix and print-over – it typically creates a loose but permanent connection between two objects,” explained the researchers.
While the new Encore tool is still under development as researchers improve upon its analytical capabilities, it certainly seems to show promise to those of us wishing to do more than just fabricate new items. In fact, the researchers were able to show that via all three techniques they could save a substantial amount of time and material over printing an object in one single piece. As an example, they used Slic3r to estimate the print time and total material required for printing a typical Utah teapot with a torus-shaped handle, as well as just printing the handle onto an already fabricated Utah teapot. Their estimate showed that the time of fabricating the item could be cut by more than 80% and material use reduced by as much as 85% by using their techniques and the Encore tool.
There are many variables going into the tool’s decision making algorithms, such as determining where to place attachments for the best balance when holding an object, what placement of an attachment will result in the best adhesion, etc. More research is still required as the team continues to develop the tool, as well as new techniques to attach multiple parts to one object, but it certainly seems like something which could have a sizable impact on the industry in general. Let us know your thoughts on the Encore tool in the Augmenting 3D Prints forum thread on 3DPB.com.
The Limits of 3D Printing
Matthias Holweg
Harvard Business Review Feb 2015 https://hbr.org/2015/06/the-limits-of-3d-printing
Contrary to what some say, 3D printing is not going to revolutionize the manufacturing sector, rendering traditional factories obsolete. The simple fact of the matter is the economics of 3D printing now and for the foreseeable future make it an unfeasible way to produce the vast majority of parts manufactured today. So instead of looking at it as a substitute for existing manufacturing, we should look to new areas where it can exploit its unique capabilities to complement traditional manufacturing processes.
Additive manufacturing, or “3D printing” as it is commonly known, has understandably captured the popular imagination: New materials that can be “printed” are announced virtually every day, and the most recent generation of printers can even print several materials at the same time, opening up new opportunities. Exciting applications have already been demonstrated across all sectors — from aerospace and medical applications to biotechnology and food production.
Some predict that a day is coming when we’ll be able to make any part at the push of a button at a local printer, which might even render the global supply lines that dominate today’s world of manufacturing a thing of the past. Unfortunately, this vision does not stack up to economic reality. Early findings from a research project being conducted by the Additive Manufacturing and 3D Printing Research Group at the University of Nottingham and Saïd Business School at the University of Oxford show that there are both significant scale and learning effects inherent in the 3D printing process. (The project, in which I am a principal investigator, is focusing on industrial selective laser sintering (or more accurately, melting) processes and not fused deposition modelling or stereolithography processes that are more suited to rapid prototyping and home applications.)
Furthermore, the pre- and post-printing cost amount to a significant proportion of total cost per printed part. So even when the cost for printers materials come down, the labor-cost penalty will remain.
3D printing simply works best in areas where customization is key — from printing hearing aids and dental implants to printing a miniature of the happy couple for their wedding cake. Using a combination of 3D scanning and printing, implants can be customized to specific anatomic circumstances in a way that was simply not feasible beforehand. However, we also know that 99% of all manufactured parts are standard and do not require customization. In these cases, 3D printing has to compete with scale-driven manufacturing processes and rather efficient logistics operations. A good example is the wrench that NASA printed on the International Space Station last year. The cost of shipping it to the space station would have been at least $400 (assuming the unpackaged weight of 18 grams per wrench and using the most recent cost data given by NASA for transporting goods into lower-earth orbit); in comparison, shipping it from China to the United States would only cost $0.002 per unit. Thus, while it makes a lot of sense to print the wrench on the space station, printing it for local consumption in the United States wouldn’t.
The simple fact is that when customization isn’t important, 3D printing is not competitive. For one, printing costs per part are highly sensitive to the utilization of the “build room,” the three-dimensional area inside the 3D printer where the laser fuses the metal or plastic powder. Therefore, contract manufacturers that perform 3D printing such as Shapeways generally wait to fill a batch that uses the entire build room. Printing just one part raises unit cost considerably; so economies of scale do matter. Interestingly, the economic case for the most-cited standard part in 3D volume production today, the GE fuel nozzle for the CFM LEAP engine, is it is lighter and more fuel efficient, not a lower manufacturing cost per se.
A second point often overlooked is that the labor cost that remains. Counter to common perception, 3D printing does not happen “at the touch of a button”; it involves considerable pre- and post-processing, which incur non-trivial labor costs. The starting point for any 3D printing process is a 3D file that can be “printed.” Just having an electronic CAD drawing is not sufficient; currently, there is no way to automatically convert the CAD drawing into a 3D file.
Creating printable files involves two steps: creating a three-dimensional volume model that can be printed, and “slicing” that volume model in the best possible way to avoid material wastage and prevent printing errors. Both steps require tacit knowledge. Following the printing, the parts produced have to be recovered, cleaned, washed (or sanded and polished, in the case of metal prints), and inspected. This, in turn, means that using 3D printing for the aftermarket services — an application where it makes a lot of sense — requires making a significant upfront investment in generating the printable files of the spare parts that would likely be needed. This investment would have to outweigh the cost of keeping a lifetime supply of spare parts in inventory, which is a tough call for small bolts, brackets, and connectors that make up the bulk of aftermarket demand.
So while I, like many others, have fallen in love with the notion of the “ultimate lean supply chain” of having 3D printers at every other corner table to print single parts just in time where they are needed, I am afraid that this vision does not stack up against reality. 3D printing technology undoubtedly has great potential. However, it is unlikely to replace traditional manufacturing. Instead, we should see it as a complement, a new tool in the box, and exploit its unique capabilities — both in making existing products better as well as being able to manufacture entirely new ones that we previously could not make.
Medical implants and printable body parts to drive 3D printer growth
With 3D bio-printing in the pipeline, dental and medical applications could be worth $6bn by 2025
http://www.theguardian.com/business/2014/aug/24/medical-implants-drive-3d-printer-growth
False teeth, hip joints and replacement knees – and potentially printable skin and organs – will drive growth in the burgeoning market for 3D printers over the next decade, according to new research.
A report suggests that dentistry and medicine will increasingly harness one of the 21st century’s most exciting technological breakthroughs.
The technology is better known to British households for its ability to replace broken crockery or produce awkward figurine “selfies.”
But a report by Cambridge-based market research firm IDTechEx says ceramic jaw or teeth implants and metal hip replacements will become increasingly common 3D fare.
The parts are created by nozzles laying down fine sedimentary layers of material that build a product indistinguishable from an item that has rolled off a factory conveyor belt.
The dental and medical market for 3D printers is expected to expand by 365% to $867m (£523m) by 2025, according to IDTechEx analysts, even before bio-printing technology is taken into account. If bio-printing becomes suitable for commercial use – which scientists hope will allow the printing of pieces of skin, liver or kidney using live cells – analysts estimate the medical market could reach a value of $6bn or more within 10 years.
While printing of complete organs for transplants may be decades away, the use of pieces of tissue for laboratory toxicology tests for cosmetics or drugs could be ready within five years, helping the medical market for 3D printers overtake all other sectors.
Dr Jon Harrop, a director of IDTechEx, said: “Bio printing is a bit unsure as it doesn’t exist commercially at the moment but all the medical professionals we interviewed thought it was highly likely to be commercial within 10 years.”
In the US, dental labs have invested in technology that can scan a patient’s teeth so new teeth can be produced by pressing the print button.
Harrop said there are a number of stumbling blocks in the way of the commercial application of bio printing, but even in the past year, scientists have been able to extend the life of a piece of skin tissue created in the lab from just a few hours to 40 days, taking it closer to the three months required for toxicology tests.
At present, 3D printers are most widely used in the automotive industry where they help produce prototypes for new cars or car parts. The next biggest market is aerospace, where manufacturers are using the technology to make lighter versions of complex parts for aeroplanes.
Already, 3D printers have been used by the medical industry to create a jaw, a pelvis and several customised hip replacements from metal. This year, surgeons in Newcastle upon Tyne created a titanium pelvis for a man who lost half his original one to a rare bone cancer, while in May doctors in Southampton completed Britain’s first hip replacement made using a 3D printer. Professor Richard Oreffo at the University of Southampton, who helped develop the hip replacement technique, said at the time: “The 3D printing of the implant in titanium, from CT scans of the patient and stem cell graft, is cutting edge and offers the possibility of improved outcomes for patients.”
Dentists have been using 3D printers to create exact replicas of jaws or teeth in order to aid complex procedures for a few years, but increasingly they are creating implants made of durable plastic or medical ceramics.
The Future of 3D Printing
By Stephen F. DeAngelis
http://www.enterrasolutions.com/2013/03/the-future-of-3d-printing.html
In his most recent State of the Union address, President Barack Obama stated, “Last year, we created our first manufacturing innovation institute in Youngstown, Ohio. A once-shuttered warehouse is now a state-of-the art lab where new workers are mastering the 3D printing that has the potential to revolutionize the way we make almost everything. There’s no reason this can’t happen in other towns. So tonight, I’m announcing the launch of three more of these manufacturing hubs, where businesses will partner with the Department of Defense and Energy to turn regions left behind by globalization into global centers of high-tech jobs. And I ask this Congress to help create a network of 15 of these hubs and guarantee that the next revolution in manufacturing is made right here in America.” [“Remarks by the President in the State of the Union Address,” White House, 12 February 2013]
Official White House Photo by Chuck Kennedy (not shown)
Clearly, the President believes that 3D printing marks a new era in manufacturing that could change the entire business landscape. Kinaxis analyst Andrew Bell agrees that “change is inevitable.” “The question that we need to answer,” he writes, “is how will it change?” [“What Could 3D Printing Mean for the Supply Chain? The 21st Century Supply Chain, 9 January 2013] Bell offers a few thoughts on the subject; he writes:
“One thing is for sure, the supply chain isn’t going away. As usual, it will likely just get more complicated. Here are some of the areas that I propose will influence the supply chain as 3D printing becomes more and more mainstream, and I’m sure there are many more.
- Local Manufacturing – More things will be made closer to their final destination. This will have definite impact on the logistics industry, and will change the way business try and schedule their operations.
- Customizability – It will be easier, faster, and more efficient for companies to provide made-to-order products to their end users.
- Distribution of raw materials – There will need to be a dramatic shift in the way raw materials are distributed since these printers will require raw materials in order to produce the final product.
- New replacement parts model – Business will be able to provide replacement parts as required instead of trying to predict the need and manufacture the stock well in advance (as they do today)
- Blurred boundaries within businesses – A closer integration of the various departments of an organization will be mandatory. A siloed manufacturing department will no longer allow for a competitive business.
“My predictions may be right, or they may be wrong, but one thing I think all will agree on is that 3D printing will make the supply chain more complex and more difficult to manage.”
Jim Stockton notes that 3D printing (or additive manufacturing) has been around since the 1980s. It has only broken into the mainstream because of recent breakthroughs that now have everyone talking. [“Top Innovations in the World of 3D Printing,” BestDesignTuts, 12 February 2013] He writes:
“Some products produced with this technology that might already be a part of your daily life include shoes, jewellery, clothing accessories, and educational products. The field of engineering is also benefitting from the development of 3D printing, in terms of boosting geographic information systems, aerospace engineering, engineering projects, and construction processes. Furthermore, humans are already benefitting in the field of healthcare with dental and medical instruments and products being formulated with 3D printing. The automotive industry is able to make more reliable products at a quicker speed, and the industrial design and architecture fields are able to develop new products that were inconceivable only a decade ago.”
What is really exciting people, however, is not so much what is currently being manufactured but the potential of what could be manufactured using 3D printers. Stockton writes:
“Engineers and scientists around the world are looking forward to the near future, in which printers will be able to create equipment, tools and devices via open-source models. This kind of advancement will drastically change the ways in which research and practical medicine are performed. Chemists are even attempting to build chemical compounds using 3D printers, and scientists have already started to replicate fossils and other ancient materials to better understand their compositions and functions. Architects wonder if in the future, buildings themselves can be printed from the ground up. The future of 3D printing is very bright – both in small-scale products for individual users and for large-scale projects, such as printing meters of building materials in the course of an hour and making intricate parts of automobiles and planes.”
http://www.enterrasolutions.com/media/images/2013/03/6a00d8341c4ebd53ef017c36e043b0970b-600wi.jpg
Vivek Srinivasan and Jarrod Bassan offer ten trends that will likely define the direction that will be taken by additive manufacturing in the years ahead. [“Manufacturing The Future: 10 Trends To Come In 3D Printing,” Forbes, 7 December 2012] They are:
- 3D printing becomes industrial strength. Once reserved for prototypes and toys, 3D printing will become industrial strength. You will take a flight on an airliner that includes 3D-printed components, making it lighter and more fuel efficient. In fact, there are aircrafts that already contain some 3D-printed components. The technology will also start to be adopted for the direct manufacture of specialist components in industries like defense and automotive. Overall, the number of 3D printed parts in planes, cars and even appliances will increase without you knowing.
- 3D printing starts saving lives. 3D-printed medical implants will improve the quality of life of someone close to you. Because 3D printing allows products to be custom-matched to an exact body shape, it is being used today for making better titanium bone implants, prosthetic limbs and orthodontic devices. Experiments in printing soft tissue are underway, and may soon allow printed veins and arteries to be used in operations. Today’s research into medical applications of 3D printing covers nano-medicine, pharmaceuticals and even printing of organs. Taken to the extreme, 3D printing could one day enable custom medicines and reduce if not eliminate the organ donor shortage.
- Customization becomes the norm. You will buy a product, customized to your exact specifications, which is 3D-printed and delivered to your doorstep. Innovative companies will use 3D printing technologies to give themselves a competitive advantage by offering customization at the same price as their competitor’s standard products. At first this may range from novelty items like custom smartphone cases or ergonomic improvements to standard tools, but it will rapidly expand to new markets. The leaders will adjust their sales, distribution and marketing channels to take advantage of their capability to provide customization direct to the customer. Customization will also play a big role in healthcare devices such as 3D-printed hearing aids and artificial limbs.
- Product innovation is faster. Everything from new car models to better home appliances will be designed more rapidly, bringing innovation to you faster. Because rapid prototyping using 3D printers reduces the time to turn a concept into a production-ready design, it allows designers to focus on the function of products. Although the use of 3D printing for rapid prototyping is not new, the rapidly decreasing cost, improved design software and increasing range of printable materials means designers will have more access to printers, allowing them to innovate faster by 3D printing an object early in the design phase, modifying it, re-printing it, and so on. The result will be better products, designed faster.
- New companies develop innovative business models built on 3D printing. You will invest in a 3D printing company’s IPO. Start-up companies will flourish as a generation of innovators, hackers and “makers” take advantage of the capabilities of 3D printing to create new products or deliver services to the burgeoning 3D printer market. Some enterprises will fail, and there may be a boom-bust cycle, but 3D printing will spawn new and creative business models.
- 3D print shops open at the mall. 3D print shops will begin to appear, at first servicing local markets with high-quality 3D printing services. Initially designed to service rapid-prototyping and other niche capabilities, these shops will branch into the consumer marketplace. As retailers begin to “ship the design, not the product,” the local 3D print shop will one day be where you pick up your customized, locally manufactured products, just like you pick up your printed photos from the local Walmart today.
- Heated debates on who owns the rights emerge. As manufacturers and designers start to grapple with the prospect of their copyrighted designs being replicated easily on 3D printers, there will be high-profile test cases over the intellectual property of physical object designs. Just like file-sharing sites shook the music industry because they made it easy to copy and share music, the ability to easily copy, share, modify and print 3D objects will ignite a new wave of intellectual property issues.
- New products with magical properties will tantalize us. New products – that can only be created on 3D printers – will combine new materials, nano scale and printed electronics to exhibit features that seem magical compared to today’s manufactured products. These printed products will be desirable and have distinct competitive advantage. The secret sauce is that 3D printing can control material as it is printed, right down to the molecules and atoms. As today’s research is perfected into tomorrow’s commercially available printers, expect exciting and desirable new products with amazing capabilities. The question is: What are these products and who will be selling them?
- New machines grace the factory floor. Expect to see 3D printing machines appearing in factories. Already some niche components are produced more economically on 3D printers, but this is only on a small scale. Many manufacturers will begin experimenting with 3D printing for applications outside of prototyping. As the capabilities of 3D printers develop and manufacturers gain experience in integrating them into production lines and supply chains, expect hybrid manufacturing processes that incorporate some 3D-printed components. This will be further fueled by consumers desiring products that require 3D printers for their manufacture.
- “Look what I made!” Your children will bring home 3D printed projects from school. Digital literacy – including Web and app development, electronics, collaboration and 3D design – will be supported by 3D printers in schools. A number of middle schools and high schools already have 3D printers. As 3D printing costs continue to fall, more schools will sign on. Digital literacy will be about things as well as bits.
If, as President Obama believes, a manufacturing revolution, led by 3D printing, is coming, it behooves business leaders in every field to ask themselves how it could affect their business model and assumptions about the future.
Research Leads to the 3D Printing of Pure Graphene Nanostructures
http://www.33rdsquare.com/2014/12/research-leads-to-3d-printing-of-pure.html
Graphene
Researchers in Korea have successfully 3D printed graphene nano-structures without the use of any other material. With the entire printed structure being composed of graphene, the strength, as well as full conductivity of the material can be taken advantage of.

3d printing graphene
https://pharmaceuticalintelligence.com/wp-content/uploads/2015/09/8d75d-3d2bprinting2bgraphene.jpg
There is no question that graphene, has enormous potential, from solar cell technology, to electronics to medicine. A key factor in developing practical and commercial applications of the one-atom thick carbon sheets is in aligning the material in the desired form depending on the application.
Now 3D printing of graphene is nearing a feasible stage and companies such as Graphene 3D Lab, are at the forefront of the technology.
However, there is a difference between 3D printing pure graphene, and 3D printing a graphene/thermoplastic composites like Graphene 3D has been doing.
While printing with composite materials, using a typical FDM/FFF or powder based laser sintering process, will keep some of graphene’s superior properties intact, most will be lost. The plastic will eventually break down leaving any prints weak, and not much different from a typical object you’d print with a MakerBot Replicator.
Now, researchers, led by Professor Seung Kwon Seol from Korea Electrotechnology Research Institute (KERI), recentlypublished a paper in Advanced Materials where they describe a new process of directly 3D printing pure graphene.
Their techniques mean that graphene nano-structures can be fabricated without the use of any other material. With the entire printed structure being composed of graphene, the strength, as well as full conductivity of the material can be realized.
| “We are convinced that this approach will present a new paradigm for implementing 3D patterns in printed electronics.” |
“We developed a nanoscale 3D printing approach that exploits a size-controllable liquid meniscus to fabricate 3D reduced graphene oxide (rGO) nanowires,” Seol told Nanowerk. “Different from typical 3D printing approaches which use filaments or powders as printing materials, our method uses the stretched liquid meniscus of ink. This enables us to realize finer printed structures than a nozzle aperture, resulting in the manufacturing of nanostructures.”
“So far, to the best of our knowledge, nobody has reported 3D printed nanostructures composed entirely of graphene,” says Seol. “Several results reported the 3D printing (millimeter- or centimeter-scale) of graphene or carbon nanotube/plastic composite materials by using a conventional 3D printer. In such composite system, the graphene (or CNT) plays an important role for improving the properties of plastic materials currently used in 3D printers. However, the plastic materials used for producing the composite structures deteriorate the intrinsic properties of graphene (or CNT).”
“We are convinced that this approach will present a new paradigm for implementing 3D patterns in printed electronics,” says Seol.
For their technique, the team grew graphene oxide (GO) wires at room temperature using the meniscus formed at the tip of a micropipette filled with a colloidal dispersion of GO sheets, then reduced it by thermal or chemical treatment (with hydrazine).
The deposition of GO was obtained by pulling the micropipette as the solvent rapidly evaporated, thus enabling the growth of GO wires. The researchers were able to accurately control the radius of the rGO wires by tuning the pulling rate of the pipette; they managed to reach a minimum value of ca. 150 nm.
Using this technique, they were able to produce arrays of different freestanding rGO architectures, grown directly at chosen sites and in different directions: straight wires, bridges, suspended junctions, and woven structures.
Seol points out that this 3D nanoprinting approach can be used for manufacturing 2D patterns and 3D geometry in diverse devices such as printed circuit boards, transistors, light emitting devices, solar cells, sensors and so on.
A lot of work remains to reduce the 3D printable size to below 10 nm and increase the production yield. A short video of Seol’s process is below:
https://youtu.be/cVdFzQu0uMc
Nanoparticle Solar Cells May Drive Down Price of Solar Cells
http://www.33rdsquare.com/2013/09/nanoparticle-solar-cells-may-drive-down.html
University of Alberta researchers have found that abundant materials in the Earth’s crust can be used to make inexpensive and easily manufactured nanoparticle-based solar cells.
The research, which was supported by the Natural Sciences and Engineering Research Council of Canada, is published in the latest issue of ACS Nano.
The discovery, several years in the making, is an important step forward in making solar power more accessible to parts of the world that are off the traditional electricity grid or face high power costs, such as the Canadian North, said researcher Jillian Buriak, a chemistry professor and senior research officer of the National Institute for Nanotechnology based on the U of A campus.
Buriak and her team have designed nanoparticles that absorb light and conduct electricity from two very common elements: phosphorus and zinc. Both materials are more plentiful than scarce materials such as cadmium and are free from manufacturing restrictions imposed on lead-based nanoparticles.
“Half the world already lives off the grid, and with demand for electrical power expected to double by the year 2050, it is important that renewable energy sources like solar power are made more affordable by lowering the costs of manufacturing,” Buriak said.
“My goal is that a store like Ikea could sell rolls of these things with simple instructions and baggies of screws and do-dads and you could install them yourself,” said Buriak
Her team’s research supports a promising approach of making solar cells cheaply using mass manufacturing methods like roll-to-roll printing (as with newspaper presses) or spray-coating (similar to automotive painting). “Nanoparticle-based ‘inks’ could be used to literally paint or print solar cells or precise compositions,” Buriak said.
Buriak collaborated with U of A post-doctoral fellows Erik Luber of the U of A Faculty of Engineering and Hosnay Mobarok of the Faculty of Science to create the nanoparticles. The team was able to develop a synthetic method to make zinc phosphide nanoparticles, and demonstrated that the particles can be dissolved to form an ink and processed to make thin films that are responsive to light.
Buriak and her team are now experimenting with the nanoparticles, spray-coating them onto large solar cells to test their efficiency. The team has applied for a provisional patent and has secured funding to enable the next step to scale up for manufacturing.
Graphene-Based Solar Cells Get Major Boost
http://www.33rdsquare.com/2013/10/graphene-based-solar-cells-get-major.html

graphene on silicon
https://pharmaceuticalintelligence.com/wp-content/uploads/2015/09/25063-grapheneonsilicon.jpg
raphene has extreme conductivity and is completely transparent while being inexpensive and nontoxic. This makes it a perfect candidate material for transparent contact layers for use in solar cells to conduct electricity without reducing the amount of incoming light – at least in theory. Whether or not this holds true in a real world setting is questionable as there is no such thing as “ideal” graphene – a free floating, flat honeycomb structure consisting of a single layer of carbon atoms: interactions with adjacent layers can change graphene’s properties dramatically.
The research recently appeared in the journal Applied Physics Letters.
“We examined how graphene’s conductive properties change if it is incorporated into a stack of layers similar to a silicon based thin film solar cell and were surprised to find that these properties actually change very little,” Marc Gluba explains.
To this end, they grew graphene on a thin copper sheet, next transferred it to a glass substrate, and finally coated it with a thin film of silicon. They examined two different versions that are commonly used in conventional silicon thin-film technologies: onesample contained an amorphous silicon layer, in which the silicon atoms are in a disordered state similar to a hardened molten glass; the other sample contained poly-crystalline silicon to help them observe the effects of a standard crystallization process on graphene‘s properties.
Even though the morphology of the top layer changed completely as a result of being heated to a temperature of several hundred degrees Celcius, the graphene is still detectable. “That’s something we didn’t expect to find, but our results demonstrate that graphene remains graphene even if it is coated with silicon,” says Norbert Nickel.
Their measurements of carrier mobility using the Hall-effect showed that the mobility of charge carriers within the embedded graphene layer is roughly 30 times greater than that of conventional zinc oxide based contact layers.
Says Gluba: “Admittedly, it’s been a real challenge connecting this thin contact layer, which is but one atomic layer thick, to external contacts. We’re still having to work on that.” Adds Nickel: “Our thin film technology colleagues are already pricking up their ears and wanting to incorporate it.” The researchers obtained their measurements on one square centimeter samples, although in practice it is feasible to coat much larger areas than that with graphene.
SOURCE Helmholtz Zentrum Berlin
Fabricated data bodies: Reflections on 3D printed digital body objects in medical and health domains
Deborah Lupton
Social Theory & Health 13, 99-115 (May 2015) | http://dx.doi.org:/10.1057/sth.2015.3
The advent of 3D printing technologies has generated new ways of representing and conceptualizing health and illness, medical practice and the body. There are many social, cultural and political implications of 3D printing, but a critical sociology of 3D printing is only beginning to emerge. In this article I seek to contribute to this nascent literature by addressing some of the ways in which 3D printing technologies are being used to convert digital data collected on human bodies and fabricate them into tangible forms that can be touched and held. I focus in particular on the use of 3D printing to manufacture non-organic replicas of individuals’ bodies, body parts or bodily functions and activities. The article is also a reflection on a specific set of digital data practices and the meaning of such data to individuals. In analyzing these new forms of human bodies, I draw on sociomaterialist perspectives as well as the recent work of scholars who have sought to theorize selfhood, embodiment, place and space in digital society and the nature of people’s interactions with digital data. I argue that these objects incite intriguing ways of thinking about the ways in digital data on embodiment, health and illnesses are interpreted and used across a range of contexts. The article ends with some speculations about where these technologies may be headed and outlining future research directions.
Osteoconduction and osteoinduction of low-temperature 3D printed bioceramic implants.
Habibovic P, Gbureck U, Doillon CJ, Bassett DC, van Blitterswijk CA, Barralet JE
Europe Pubmed Central http://dx.doi.org:/10.1016/j.biomaterials.2007.10.023
Rapid prototyping is a valuable implant production tool that enables the investigation of individual geometric parameters, such as shape, porosity, pore size and permeability, on the biological performance of synthetic bone graft substitutes. In the present study, we have employed low-temperature direct 3D printing to produce brushite and monetite implants with different geometries. Blocks predominantly consisting of brushite with channels either open or closed to the exterior were implanted on the decorticated lumbar transverse processes of goats for 12 weeks. In addition, similar blocks with closed channel geometry, consisting of either brushite or monetite were implanted intramuscularly. The design of the channels allowed investigation of the effect of macropore geometry (open and closed pores) and osteoinduction on bone formation orthotopically. Intramuscular implantation resulted in bone formation within the channels of both monetite and brushite, indicating osteoinductivity of these resorbable materials. Inside the blocks mounted on the transverse processes, initial channel shape did not seem to significantly influence the final amount of formed bone and osteoinduction was suggested to contribute to bone formation.
Like this:
Like Loading...
Read Full Post »








 The schematic on Slide 10 can be found in
The schematic on Slide 10 can be found in 






 On slide 20 the traditional Ti casting is compared with Ti 3D printing from the powders. The advantage of 3D method is low cost and high productivity. This link
On slide 20 the traditional Ti casting is compared with Ti 3D printing from the powders. The advantage of 3D method is low cost and high productivity. This link  Slide 21 For 3D Bioploter made by EnvisionTec we notice the usage of materials such as metal followed by post-processing sintering, Hydroxylapatite, TCP, Titanium. Using a preciptation method the machine can handle Chitosan, Collagen, 2-component system of the two possible combination: Alginate, Fibrin, PU, and Silicone. More details in
Slide 21 For 3D Bioploter made by EnvisionTec we notice the usage of materials such as metal followed by post-processing sintering, Hydroxylapatite, TCP, Titanium. Using a preciptation method the machine can handle Chitosan, Collagen, 2-component system of the two possible combination: Alginate, Fibrin, PU, and Silicone. More details in 





















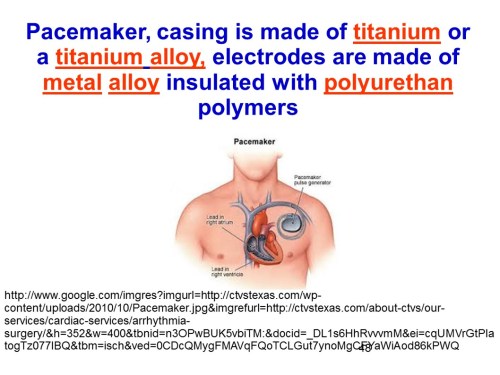

 The insulin pump shown in slide 50 is a schematic of the pump controlled electronically by a control algorithm device, a sensor, an electronic receiver that connects with an iPhone through an wireless channel.
The insulin pump shown in slide 50 is a schematic of the pump controlled electronically by a control algorithm device, a sensor, an electronic receiver that connects with an iPhone through an wireless channel.
 Slide 52 describes how easily available bio-compatible metal powders
Slide 52 describes how easily available bio-compatible metal powders
 Slide 54 shows conclusions regarding the hardware of the presentation, in which: 6) there are two types of metal 3D printing hardware for medical applications: Selective Laser Melting / Selective Laser Sintering, and 3D Bioploter (metal powder mixed with binder and further thermal treatment to remove binder and sinter the metallic matrix in a solid object that can be used as a replacement. Thank you for your attention!
Slide 54 shows conclusions regarding the hardware of the presentation, in which: 6) there are two types of metal 3D printing hardware for medical applications: Selective Laser Melting / Selective Laser Sintering, and 3D Bioploter (metal powder mixed with binder and further thermal treatment to remove binder and sinter the metallic matrix in a solid object that can be used as a replacement. Thank you for your attention!