Summary of Lipid Metabolism
Author: Larry H. Bernstein, MD, FCAP
Lipid Classification System
The LIPID MAPS Lipid Classification System is comprised of eight lipid categories, each with its own sublassification hierarchy.
http://www.lipidmaps.org/resources/tutorials/lipid_cns.html
Each LMSD record contains an image of the
- molecular structure,
- common and systematic names,
- links to external databases,
- Wikipedia pages (where available),
- other annotations and links to structure viewing tools.
All lipids in the LIPID MAPS Structure Database (LMSD) have been classified using this system and have been assigned LIPID MAPS ID’s (LM_ID) which reflects their position in the classification hierarchy.
The LIPID MAPS Structure Database (LMSD) is a relational database encompassing structures and annotations of biologically relevant lipids. As of May 3, 2013, LMSD contains over 37,500 unique lipid structures, making it the largest public lipid-only database in the world. Structures of lipids in the database come from several sources:
- LIPID MAPS Consortium’s core laboratories and partners;
- lipids identified by LIPID MAPS experiments;
- biologically relevant lipids manually curated from LIPID BANK, LIPIDAT, Lipid Library, Cyberlipids, ChEBI and other public sources;
- novel lipids submitted to peer-reviewed journals;
- computationally generated structures for appropriate classes.
All the lipid structures in LMSD adhere to the structure drawing rules proposed by the LIPID MAPS consortium. A number of structure viewing options are offered: gif image (default), Chemdraw (requires Chemdraw ActiveX/Plugin), MarvinView (Java applet) and JMol (Java applet).
(as of 10/8/14)
Number of lipids per category
Fatty acyls 5869
Glycerolipids 7541
Glycerophospholipids 8002
Sphingolipids 4338
Sterol lipids 2715
Prenol lipids 1259
Sacccharolipids 1293
Polyketides 6742
TOTAL 37,759 structures
References
Sud M, Fahy E, Cotter D, Brown A, Dennis EA, Glass CK, Merrill AH Jr, Murphy RC, Raetz CR, Russell DW, Subramaniam S. LMSD: LIPID MAPS structure database Nucleic Acids Research 35: p. D527-32. PMID:17098933 [doi:10.1093/nar/gkl838] PMID: 17098933
Fahy E, Sud M, Cotter D & Subramaniam S. LIPID MAPS online tools for lipid research Nucleic Acids Research (2007) 35: p. W606-12.PMID:17584797 [doi:10.1093/nar/gkm324] PMID: 17584797
The Recognition of Essential Fatty Acids
Dietary fat has long been recognized as an important source of energy for mammals, but in the late 1920s, researchers demonstrated the dietary requirement for particular fatty acids, which came to be called essential fatty acids. It was not until the advent of intravenous feeding, however, that the importance of essential fatty acids was widely accepted: Clinical signs of essential fatty acid deficiency are generally observed only in patients on total parenteral nutrition who received mixtures devoid of essential fatty acids or in those with malabsorption syndromes.
These signs include dermatitis and changes in visual and neural function. Over the past 40 years, an increasing number of physiological functions, such as immunomodulation, have been attributed to the essential fatty acids and their metabolites, and this area of research remains quite active.1, 2
Fatty Acid Nomenclature
The fat found in foods consists largely of a heterogeneous mixture of triacylglycerols (triglycerides)–glycerol molecules that are each combined with three fatty acids. The fatty acids can be divided into two categories, based on chemical properties: saturated fatty acids, which are usually solid at room temperature, and unsaturated fatty acids, which are liquid at room temperature. The term “saturation” refers to a chemical structure in which each carbon atom in the fatty acyl chain is bound to (saturated with) four other atoms, these carbons are linked by single bonds, and no other atoms or molecules can attach; unsaturated fatty acids contain at least one pair of carbon atoms linked by a double bond, which allows the attachment of additional atoms to those carbons (resulting in saturation). Despite their differences in structure, all fats contain approximately the same amount of energy (37 kilojoules/gram, or 9 kilocalories/gram).
The class of unsaturated fatty acids can be further divided into monounsaturated and polyunsaturated fatty acids. Monounsaturated fatty acids (the primary constituents of olive and canola oils) contain only one double bond. Polyunsaturated fatty acids (PUFAs) (the primary constituents of corn, sunflower, flax seed and many other vegetable oils) contain more than one double bond. Fatty acids are often referred to using the number of carbon atoms in the acyl chain, followed by a colon, followed by the number of double bonds in the chain (e.g., 18:1 refers to the 18-carbon monounsaturated fatty acid, oleic acid; 18:3 refers to any 18-carbon PUFA with three double bonds).
PUFAs are further categorized on the basis of the location of their double bonds. An omega or n notation indicates the number of carbon atoms from the methyl end of the acyl chain to the first double bond. Thus, for example, in the omega-3 (n-3) family of PUFAs, the first double bond is 3 carbons from the methyl end of the molecule. Finally, PUFAs can be categorized according to their chain length. The 18-carbon n-3 and n-6 short-chain PUFAs are precursors to the longer 20- and 22-carbon PUFAs, called long-chain PUFAs (LCPUFAs).
Fatty Acid Metabolism
Mammalian cells can introduce double bonds into all positions on the fatty acid chain except the n-3 and n-6 position. Thus, the short-chain alpha- linolenic acid (ALA, chemical abbreviation: 18:3n-3) and linoleic acid (LA, chemical abbreviation: 18:2n-6) are essential fatty acids.
No other fatty acids found in food are considered ‘essential’ for humans, because they can all be synthesized from the short chain fatty acids.
Following ingestion, ALA and LA can be converted in the liver to the long chain, more unsaturated n-3 and n-6 LCPUFAs by a complex set of synthetic pathways that share several enzymes (Figure 1). LC PUFAs retain the original sites of desaturation (including n-3 or n-6). The omega-6 fatty acid LA is converted to gamma-linolenic acid (GLA, 18:3n-6), an omega- 6 fatty acid that is a positional isomer of ALA. GLA, in turn, can be converted to the longerchain omega-6 fatty acid, arachidonic acid (AA, 20:4n-6). AA is the precursor for certain classes of an important family of hormone- like substances called the eicosanoids (see below).
The omega-3 fatty acid ALA (18:3n-3) can be converted to the long-chain omega-3 fatty acid, eicosapentaenoic acid (EPA; 20:5n-3). EPA can be elongated to docosapentaenoic acid (DPA 22:5n-3), which is further desaturated to docosahexaenoic acid (DHA; 22:6n-3). EPA and DHA are also precursors of several classes of eicosanoids and are known to play several other critical roles, some of which are discussed further below.
The conversion from parent fatty acids into the LC PUFAs – EPA, DHA, and AA – appears to occur slowly in humans. In addition, the regulation of conversion is not well understood, although it is known that ALA and LA compete for entry into the metabolic pathways.
Physiological Functions of EPA and AA
As stated earlier, fatty acids play a variety of physiological roles. The specific biological functions of a fatty acid are determined by the number and position of double bonds and the length of the acyl chain.
Both EPA (20:5n-3) and AA (20:4n-6) are precursors for the formation of a family of hormone- like agents called eicosanoids. Eicosanoids are rudimentary hormones or regulating – molecules that appear to occur in most forms of life. However, unlike endocrine hormones, which travel in the blood stream to exert their effects at distant sites, the eicosanoids are autocrine or paracrine factors, which exert their effects locally – in the cells that synthesize them or adjacent cells. Processes affected include the movement of calcium and other substances into and out of cells, relaxation and contraction of muscles, inhibition and promotion of clotting, regulation of secretions including digestive juices and hormones, and control of fertility, cell division, and growth.3
The eicosanoid family includes subgroups of substances known as prostaglandins, leukotrienes, and thromboxanes, among others. As shown in Figure 1.1, the long-chain omega-6 fatty acid, AA (20:4n-6), is the precursor of a group of eicosanoids that include series-2 prostaglandins and series-4 leukotrienes. The omega-3 fatty acid, EPA (20:5n-3), is the precursor to a group of eicosanoids that includes series-3 prostaglandins and series-5 leukotrienes. The AA-derived series-2 prostaglandins and series-4 leukotrienes are often synthesized in response to some emergency such as injury or stress, whereas the EPA-derived series-3 prostaglandins and series-5 leukotrienes appear to modulate the effects of the series-2 prostaglandins and series-4 leukotrienes (usually on the same target cells). More specifically, the series-3 prostaglandins are formed at a slower rate and work to attenuate the effects of excessive levels of series-2 prostaglandins. Thus, adequate production of the series-3 prostaglandins seems to protect against heart attack and stroke as well as certain inflammatory diseases like arthritis, lupus, and asthma.3.
EPA (22:6 n-3) also affects lipoprotein metabolism and decreases the production of substances – including cytokines, interleukin 1ß (IL-1ß), and tumor necrosis factor a (TNF-a) – that have pro-inflammatory effects (such as stimulation of collagenase synthesis and the expression of adhesion molecules necessary for leukocyte extravasation [movement from the circulatory system into tissues]).2 The mechanism responsible for the suppression of cytokine production by omega-3 LC PUFAs remains unknown, although suppression of omega-6-derived eicosanoid production by omega-3 fatty acids may be involved, because the omega-3 and omega-6 fatty acids compete for a common enzyme in the eicosanoid synthetic pathway, delta-6 desaturase.
DPA (22:5n-3) (the elongation product of EPA) and its metabolite DHA (22:6n-3) are frequently referred to as very long chain n-3 fatty acids (VLCFA). Along with AA, DHA is the major PUFA found in the brain and is thought to be important for brain development and function. Recent research has focused on this role and the effect of supplementing infant formula with DHA (since DHA is naturally present in breast milk but not in formula).
Overview of Lipid Catabolism:
http://www.elmhurst.edu/~chm/vchembook/622overview.html
The major aspects of lipid metabolism are involved with
- Fatty Acid Oxidation to produce energy or
- the synthesis of lipids which is called Lipogenesis.
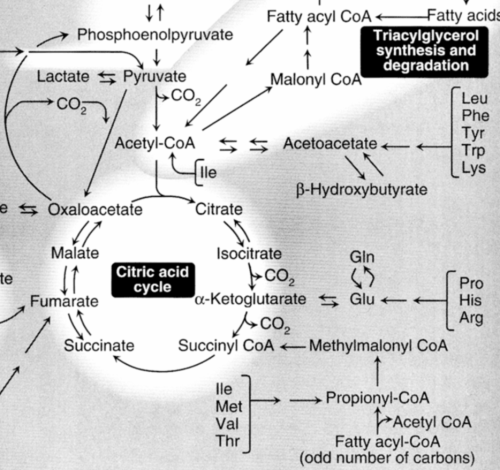
The metabolism of lipids and carbohydrates are related by the conversion of lipids from carbohydrates. This can be seen in the diagram. Notice the link through actyl-CoA, the seminal discovery of Fritz Lipmann. The metabolism of both is upset by diabetes mellitus, which results in the release of ketones (2/3 betahydroxybutyric acid) into the circulation.
http://www.elmhurst.edu/~chm/vchembook/images/590metabolism.gif
The first step in lipid metabolism is the hydrolysis of the lipid in the cytoplasm to produce glycerol and fatty acids.
Since glycerol is a three carbon alcohol, it is metabolized quite readily into an intermediate in glycolysis, dihydroxyacetone phosphate. The last reaction is readily reversible if glycerol is needed for the synthesis of a lipid.
The hydroxyacetone, obtained from glycerol is metabolized into one of two possible compounds. Dihydroxyacetone may be converted into pyruvic acid, a 3-C intermediate at the last step of glycolysis to make energy.
In addition, the dihydroxyacetone may also be used in gluconeogenesis (usually dependent on conversion of gluconeogenic amino acids) to make glucose-6-phosphate for glucose to the blood or glycogen depending upon what is required at that time.
Fatty acids are oxidized to acetyl CoA in the mitochondria using the fatty acid spiral. The acetyl CoA is then ultimately converted into ATP, CO2, and H2O using the citric acid cycle and the electron transport chain.
There are two major types of fatty acids – ω-3 and ω-6. There are also saturated and unsaturated with respect to the existence of double bonds, and monounsaturated and polyunsatured. Polyunsaturated fatty acids (PUFAs) are important in long term health, and it will be seen that high cardiovascular risk is most associated with a low ratio of ω-3/ω-6, the denominator being from animal fat. Ω-3 fatty acids are readily available from fish, seaweed, and flax seed. More can be said of this later.
Fatty acids are synthesized from carbohydrates and occasionally from proteins. Actually, the carbohydrates and proteins have first been catabolized into acetyl CoA. Depending upon the energy requirements, the acetyl CoA enters the citric acid cycle or is used to synthesize fatty acids in a process known as LIPOGENESIS.
The relationships between lipid and carbohydrate metabolism are
summarized in Figure 2.
Energy Production Fatty Acid Oxidation:
“Visible” ATP:
In the fatty acid spiral, there is only one reaction which directly uses ATP and that is in the initiating step. So this is a loss of ATP and must be subtracted later.
A large amount of energy is released and restored as ATP during the oxidation of fatty acids. The ATP is formed from both the fatty acid spiral and the citric acid cycle.
Connections to Electron Transport and ATP:
One turn of the fatty acid spiral produces ATP from the interaction of the coenzymes FAD (step 1) and NAD+ (step 3) with the electron transport chain. Total ATP per turn of the fatty acid spiral is:
Electron Transport Diagram – (e.t.c.)
Step 1 – FAD into e.t.c. = 2 ATP
Step 3 – NAD+ into e.t.c. = 3 ATP
Total ATP per turn of spiral = 5 ATP
In order to calculate total ATP from the fatty acid spiral, you must calculate the number of turns that the spiral makes. Remember that the number of turns is found by subtracting one from the number of acetyl CoA produced. See the graphic on the left bottom.
Example with Palmitic Acid = 16 carbons = 8 acetyl groups
Number of turns of fatty acid spiral = 8-1 = 7 turns
ATP from fatty acid spiral = 7 turns and 5 per turn = 35 ATP.
This would be a good time to remember that single ATP that was needed to get the fatty acid spiral started. Therefore subtract it now.
NET ATP from Fatty Acid Spiral = 35 – 1 = 34 ATP
SREBPs: activators of the complete program of cholesterol and fatty acid synthesis in the liver
Jay D. Horton1,2, Joseph L. Goldstein1 and Michael S. Brown1
1Department of Molecular Genetics, and
2Department of Internal Medicine, University of Texas Southwestern Medical Center, Dallas, Texas, USA
J Clin Invest. 2002;109(9):1125–1131.
http://dx.doi.org:/10.1172/JCI15593
Lipid homeostasis in vertebrate cells is regulated by a family of membrane-bound transcription factors designated sterol regulatory element–binding proteins (SREBPs). SREBPs directly activate the expression of more than 30 genes dedicated to the synthesis and uptake of cholesterol, fatty acids, triglycerides, and phospholipids, as well as the NADPH cofactor required to synthesize these molecules (1–4). In the liver, three SREBPs regulate the production of lipids for export into the plasma as lipoproteins and into the bile as micelles. The complex, interdigitated roles of these three SREBPs have been dissected through the study of ten different lines of gene-manipulated mice. These studies form the subject of this review.
SREBPs: activation through proteolytic processing
SREBPs belong to the basic helix-loop-helix–leucine zipper (bHLH-Zip) family of transcription factors, but they differ from other bHLH-Zip proteins in that they are synthesized as inactive precursors bound to the endoplasmic reticulum (ER) (1, 5). Each SREBP precursor of about 1150 amino acids is organized into three domains: (a) an NH2-terminal domain of about 480 amino acids that contains the bHLH-Zip region for binding DNA; (b) two hydrophobic transmembrane–spanning segments interrupted by a short loop of about 30 amino acids that projects into the lumen of the ER; and (c) a COOH-terminal domain of about 590 amino acids that performs the essential regulatory function described below.
In order to reach the nucleus and act as a transcription factor, the NH2-terminal domain of each SREBP must be released from the membrane proteolytically (Figure1). Three proteins required for SREBP processing have been delineated in cultured cells, using the tools of somatic cell genetics (see ref. 5for review). One is an escort protein designated SREBP cleavage–activating protein (SCAP). The other two are proteases, designated Site-1 protease (S1P) and Site-2 protease (S2P). Newly synthesized SREBP is inserted into the membranes of the ER, where its COOH-terminal regulatory domain binds to the COOH-terminal domain of SCAP (Figure 1).
Figure 1
Model for the sterol-mediated proteolytic release of SREBPs from membranes. SCAP is a sensor of sterols and an escort of SREBPs. When cells are depleted of sterols, SCAP transports SREBPs from the ER to the Golgi apparatus, where two proteases, Site-1 protease (S1P) and Site-2 protease (S2P), act sequentially to release the NH2-terminal bHLH-Zip domain from the membrane. The bHLH-Zip domain enters the nucleus and binds to a sterol response element (SRE) in the enhancer/promoter region of target genes, activating their transcription.
SCAP is both an escort for SREBPs and a sensor of sterols. When cells become depleted in cholesterol, SCAP escorts the SREBP from the ER to the Golgi apparatus, where the two proteases reside. In the Golgi apparatus, S1P, a membrane-bound serine protease, cleaves the SREBP in the luminal loop between its two membrane-spanning segments, dividing the SREBP molecule in half. (Fig 1) The NH2-terminal bHLH-Zip domain is then released from the membrane via a second cleavage mediated by S2P, a membrane-bound zinc metalloproteinase. The NH2-terminal domain, designated nuclear SREBP (nSREBP), translocates to the nucleus, where it activates transcription by binding to nonpalindromic sterol response elements (SREs) in the promoter/enhancer regions of multiple target genes.
SREBPs: two genes, three proteins
The mammalian genome encodes three SREBP isoforms, designated SREBP-1a, SREBP-1c, and SREBP-2.
SREBP-1a is a potent activator of all SREBP-responsive genes, including those that mediate the synthesis of cholesterol, fatty acids, and triglycerides. High-level transcriptional activation is dependent on exon 1a, which encodes a longer acidic transactivation segment than does the first exon of SREBP-1c. The roles of SREBP-1c and SREBP-2 are more restricted than that of SREBP-1a. SREBP-1c preferentially enhances transcription of genes required for fatty acid synthesis but not cholesterol synthesis.
SREBP-1c and SREBP-2 activate three genes required to generate NADPH, which is consumed at multiple stages in these lipid biosynthetic pathways (8) (Figure 2).

major metabolic intermediates in the pathways for synthesis of cholesterol, fatty acids, and triglycerides
Steroids
A major class of lipids, steroids, have a ring structure of three cyclohexanes and one
cyclopentane in a fused ring system as shown below. There are a variety of functional
groups that may be attached. The main feature, as in all lipids, is the large number of
carbon-hydrogens which make steroids non-polar.
Steroids include such well known compounds as cholesterol, sex hormones, birth
control pills, cortisone, and anabolic steroids.
Adrenocorticoid Hormones
The adrenocorticoid hormones are products of the adrenal glands.
The most important mineralcorticoid is aldosterone, which regulates the
reabsorption of sodium and chloride ions in the kidney tubules and increases
the loss of potassium ions.Aldosterone is secreted when blood sodium ion
levels are too low to cause the kidney to retain sodium ions. If sodium
levels are elevated, aldosterone is not secreted, so that some sodium
will be lost in the urine. Aldosterone also controls swelling in the tissues.
Cortisol, the most important glucocortinoid, has the function of increasing
glucose and glycogen concentrations in the body. These reactions are
completed in the liver by taking fatty acids from lipid storage cells and
amino acids from body proteins to make glucose and glycogen.
In addition, cortisol is elevated in the circulation with cytokine mediated
(IL1, IL1, TNFα) inflammatory reaction, called the systemic inflammatory
response syndrome. Its ketone derivative, cortisone, has the ability
to relieve inflammatory effects. Cortisone or similar synthetic derivatives
such as prednisolone are used to treat inflammatory diseases, rheumatoid
arthritis, and bronchial asthma. There are many side effects with the use
of cortisone drugs, such as bone resorption, so there use must be
monitored carefully.
Hormone Receptors
Steroid hormone receptors are found on the plasma membrane, in the cytosol and also in the nucleus of target cells. They are generally intracellular receptors (typically cytoplasmic) and initiate signal transduction for steroid hormones which lead to changes in gene expression over a time period of hours to days. The best studied steroid hormone receptors are members of the nuclear receptor subfamily 3 (NR3) that include receptors for estrogen (group NR3A)[1] and 3-ketosteroids (group NR3C).[2] In addition to nuclear receptors, several G protein-coupled receptors and ion channels act as cell surface receptors for certain steroid hormones.
Steroid Hormone Receptors and their Response Elements
Steroid hormone receptors are proteins that have a binding site for a particular steroid molecule. Their response elements are DNA sequences that are bound by the complex of the steroid bound to its Steroid receptor.
The response element is part of the promoter of a gene. Binding by the receptor activates or represses, as the case may be, the gene controlled by that promoter.
It is through this mechanism that steroid hormones turn genes on (or off).
http://users.rcn.com/jkimball.ma.ultranet/BiologyPages/S/Sigler.jpg
The glucocorticoid receptor, like all steroid hormone receptors, is a zinc-finger transcription factor; the zinc atoms are the four yellow spheres. Each is attached to four cysteines.
For a steroid hormone to regulate (turn on or off) gene transcription, its receptor must:
- bind to the hormone (cortisol in the case of the glucocorticoid receptor)
- bind to a second copy of itself to form a homodimer
- be in the nucleus, moving from the cytosol if necessary
- bind to its response element
- bind to other protein cofactors
Each of these functions depend upon a particular region of the protein (e.g., the zinc fingers for binding DNA).
Each of these functions depend upon a particular region of the protein (e.g., the zinc fingers for binding DNA). Mutations in any one region may upset the function of that region without necessarily interfering with other functions of the receptor.
Positive and Negative Response Elements
Some of the hundreds of glucocorticoid response elements in the human genome activate gene transcription when bound by the hormone/receptor complex. Others inhibit gene transcription when bound by the hormone/receptor complex.
Example: When the stress hormone cortisol — bound to its receptor — enters the nucleus of a liver cell, the complex binds to the positive response elements of the many genes needed for gluconeogenesis — the conversion of protein and fat into glucose resulting in a rise in the level of blood sugar.
the negative response element of the insulin receptor gene thus diminishing the ability of the cells to remove glucose from the blood. (This hyperglycemic effect is enhanced by the binding of the cortisol/receptor complex to a negative response element in the beta cells of the pancreas thus reducing the production of insulin.)
Note that every type of cell in the body contains the same response elements in its genome. What determines if a given cell responds to the arrival of a hormone depends on the presence of the hormone’s receptor in the cell.
The Nuclear Receptor Superfamily
http://users.rcn.com/jkimball.ma.ultranet/BiologyPages/R/Retinoids.png
The zinc-finger proteins that serve as receptors for glucocorticoids and progesterone are members of a large family of similar proteins that serve as receptors for a variety of small, hydrophobic molecules. These include:
- other steroid hormones like
- the mineralocorticoid aldosterone
- estrogens
- the thyroid hormone, T3
- calcitriol, the active form of vitamin D
- retinoids: vitamin A (retinol) and its relatives
- retinal
- retinoic acid (tretinoin — also available as the drug Retin-A®); and its isomer
- isotretinoin (sold as Accutane® for the treatment of acne).
- bile acids
- fatty acids.
These bind members of the superfamily called peroxisome-proliferator-activated receptors (PPARs). They got their name from their initial discovery as the receptors for
- drugs that increase the number and size of peroxisomes in cells.
In every case, the receptors consist of at least
- three functional modules or domains.
From N-terminal to C-terminal, these are:
- a domain needed
- the zinc-finger domain needed for DNA binding (to the response element)
- the domain responsible for binding the particular hormone as well as the second unit of the dimer.
- for the receptor to activate the promoters of the genes being controlled
Schematic diagram of type II zinc finger proteins characteristic of the DNA-binding domain structure of members of the steroid hormone receptor superfamily. Zinc fingers are common features of many transcription factors, allowing proteins to bind to DNA. Each circle represents one amino acid. The CI zinc finger interacts specifically with five base pairs of DNA and determines the DNA sequence recognized by the particular steroid receptor. The three shaded amino acids indicated by the arrows in the knuckle of the CI zinc finger are in the “P box” that allows HRE sequence discrimination between the GR and ERα. The vertically striped aa within the knuckle of the CII zinc finger constitutes the “D box” that is important for dimerization and contacts with the DNA phosphate backbone. Adapted from Tsai M-J, O’Malley BW. Molecular mechanisms of action of steroid/thyroid receptor superfamily members. Annu Rev Biochem 1994;63:451-483; Gronemeyer H. Transcription activation by estrogen and progesterone receptors. Annu Rev Genet 1991;25:89-123.
Cytoskeleton and Cell Membrane Physiology
http://pharmaceuticalinnovation.com/10/28/2014/larryhbern/Cytoskeleton_
and_Cell_Membrane_Physiology
Definition and Function
The cytoskeleton is a series of intercellular proteins that help a cell with
- shape,
- support, and
- movement.
Cytoskeleton has three main structural components:
- microfilaments,
- intermediate filaments, and
- movement
The cytoskeleton mediates movement by
- helping the cell move in its environment and
- mediating the movement of the cell’s components.
Thereby it provides an important structural framework for the cell –
- the framework for the movement of organelles, contiguous with the cell membrane, around the cytoplasm. By the activity of
- the network of protein microfilaments, intermediate filaments, and microtubules.
The structural framework supports cell function as follows:
Cell shape. For cells without cell walls, the cytoskeleton determines the shape of the cell. This is one of the functions of the intermediate filaments.
Cell movement. The dynamic collection of microfilaments and microtubles can be continually in the process of assembly and disassembly, resulting in forces that move the cell. There can also be sliding motions of these structures. Audesirk and Audesirk give examples of white blood cells “crawling” and the migration and shape changes of cells during the development of multicellular organisms.
Organelle movement. Microtubules and microfilaments can help move organelles from place to place in the cell. In endocytosis a vesicle formed engulfs a particle abutting the cell. Microfilaments then attach to the vesicle and pull it into the cell. Much of the complex synthesis and distribution function of the endoplasmic reticulum and the Golgi complex makes use of transport vescicles, associated with the cytoskeleton.
Cell division. During cell division, microtubules accomplish the movement of the chromosones to the daughter nucleus. Also, a ring of microfilaments helps divide two developing cells by constricting the central region between the cells (fission).
References:
Hickman, et al. Ch 4 Hickman, Cleveland P., Roberts, Larry S., and Larson, Allan, Integrated Principles of Zoology, 9th. Ed., Wm C. Brown, 1995.
Audesirk & Audesirk Ch 6 Audesirk, Teresa and Audesirk, Gerald, Biology, Life on Earth, 5th Ed., Prentice-Hall, 1999.
http://hyperphysics.phy-astr.gsu.edu/hbase/biology/bioref.html#c1
http://hyperphysics.phy-astr.gsu.edu/hbase/biology/cytoskel.html