Prostacyclin and Nitric Oxide: Adventures in Vascular Biology – A Tale of Two Mediators
Reporter: Aviva Lev-Ari, PhD, RN
WordCloud Image Produced by Adam Tubman
Prostacyclin and Nitric Oxide: Adventures in vascular biology – a tale of two mediators
The e-Readers are encouraged to review two additional Sources on this topic on this Open Access Online Scientific Journal
Perspectives on Nitric Oxide in Disease Mechanisms
Interaction of Nitric Oxide and Prostacyclin in Vascular Endothelium
S Moncada*
Prof. Moncada:
I would like to thank the Royal Society for inviting me to deliver the Croonian Lecture. In so doing, the Society is adding my name to a list of very distinguished scientists who, since 1738, have preceded me in this task. This is, indeed, a great honour.
For most of my research career my main interest has been the understanding of the normal functioning of the blood vessel wall and the way this is affected in pathology. During this time, our knowledge of these subjects has grown to such an extent that many people now believe that the conquering of vascular disease is a real possibility in the foreseeable future.
My lecture concerns the discovery of two substances, prostacyclin and nitric oxide. I would like to describe the moments of insight and some of the critical experiments that contributed significantly to the uncovering of their roles in vascular biology. The process was often adventurous, hence the title of this lecture. It is the excitement of the adventure that I would like to convey in the text that follows.
9. THE TWO STORIES CONVERGE
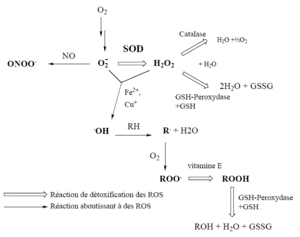
Although the research fields of prostacyclin/thromboxane and NO are now mature, they have developed mostly as parallel research activities with few points of contact between them. Thus, our understanding of how both might operate in relation to each other in physiology and pathophysiology remains to be developed. Table 2 shows some of the similarities between prostacyclin and NO. Both mediators, from very different biochemical pathways, play a variety of roles in the modulation and protection of the vascular wall. The release of both mediators is dependent on constitutive enzymes, the activity of which seems to be regulated locally, predominantly by the shear stress caused by the blood passing over the endothelial surface (Grabowski et al. 1985; Frangos et al. 1985; for review see Boo & Jo 2003). However, while the constitutive eNOS—localized only in the vascular endothelium—is the enzyme that responds to shear stress, the generation of prostacyclin is dependent on the activity of two enzymes, COX-1 and COX-2, in relation to which several questions remain unanswered. These include whether COX-2 is a constitutive as well as an inducible enzyme, and whether COX-1 or COX-2, or both, respond to shear stress by increases in their mRNA, their activity, or both (Topper et al. 1996; Okahara et al. 1998;McCormick et al. 2000; Garcia-Cardena et al. 2001). Prostacyclin, unlike NO, is constitutively generated throughout the vessel wall (Moncada et al. 1977c) and at this stage we also do not know whether the ratio between COX-1 and COX-2 changes in the different layers. In addition, the similarities and differences between regulation of NO and prostacyclin by shear stress are only now being investigated (Osanai et al. 2000; McAllister et al. 2000; Walshe et al. 2005).
Comparison of the properties of nitric oxide and prostacyclin.
A clear synergism between NO and prostacyclin has been demonstrated in regard to inhibition of platelet aggregation; however, only one of them (NO) plays a role in inhibiting platelet adhesion. The significance of this difference remains to be understood. Many years ago a physiological role for platelets in repairing the vessel wall was investigated (for discussion see Higgs et al. 1978). This subject has not been re-evaluated in the light of all this new knowledge about the roles of NO and prostacyclin in platelet/vessel wall interactions. Both mediators also regulate vascular smooth muscle proliferation and white cell vessel wall interactions through similar mechanisms which include, at least in part, the activation of adenylate cyclase and the soluble guanylate cyclase. The interactions between NO and prostacyclin in the control of these functions are not fully understood.
Both mediators are further increased by inflammatory stimuli; however, while in the case of prostacyclin the same COX-2 which responds to shear stress responds to such stimuli by a further increase in its expression, NO is generated during inflammation by a specific ‘inducible’ NO synthase which is not normally present physiologically in the vessel wall. The induction of both is inhibited by anti-inflammatory glucocorticoids (Axelrod 1983; Knowles et al. 1990). It is remarkable that both compounds possess antioxidant properties (Wink et al. 1995; Egan et al. 2004) but are themselves affected by oxidative stress, which inhibits the synthesis of prostacyclin and decreases the bioavailability of NO. This mechanism might be relevant to the ‘malfunctioning’ of the constitutive generation of both mediators and therefore to the genesis of endothelial dysfunction. This, however, is an early phenomenon. In advanced disease the situation is far more complex, akin to chronic inflammation in other parts of the body and, as such, probably varies significantly in the different stages of the disease. A simple hypothesis would suggest that any amount of prostacyclin which is bioavailable, although pro-inflammatory, will provide anti-thrombotic protection, while in the case of NO the balance will vary between bioavailable NO which is protective and cytotoxic peroxynitrite formed from the interaction of NO with O2−. Currently, however, the results are not clear and on the crucial question of the role of both mediators in the progression of atherosclerosis, the information in relation to prostacyclin is contradictory (Burleigh et al. 2002; Olesen et al. 2002; Rott et al. 2003). The evidence in relation to NO, on the other hand, seems to suggest that, while constitutive NO generated by eNOS is protective (e.g. Kawashima & Yokoyama 2004), NO generated by the inducible enzyme favours the development of atherosclerosis (Chyu et al. 1999). Studies of genetically manipulated animals are providing some important clues. For example, knockout of the prostacyclin receptor (IP) leads to mice with normal blood pressure but an increased tendency to thrombosis when the endothelium is damaged (Murata et al. 1997) These animals also exhibit an increased platelet activation and proliferative response to injury that can be prevented by deletion or antagonism of the TXA2 receptor (Cheng et al. 2002). Furthermore, deletion of the IP receptor in animals prone to spontaneous atherosclerosis accelerates the development of the disease (Egan et al. 2004;Kobayashi et al. 2004). On the other hand, knocking out the thromboxane receptor or the thromboxane synthase gives rise to a mild bleeding tendency and a resistance to platelet aggregation and sudden death induced by arachidonic acid infusion (Thomas et al. 1998; Yu et al. 2004). Deletion of the thromboxane receptor also seems to retard atherogenesis in murine models of atherosclerosis (Cayatte et al. 2000;Egan et al. 2005).
Although the lack of either mediator has been shown to increase the risk of thrombosis and atherosclerosis, especially in animals with additional risk factors such as ApoE deficiencies (Kuhlencordtet al. 2001; Belton et al. 2003), there seems to be a certain specialization in their actions, so that NO has a more significant role in the regulation of blood pressure and blood flow, while prostacyclin has a clearer role in regulating platelet/vessel wall interactions. For example, inhibition of NO generation has an immediate and dramatic effect on blood flow and blood pressure and the eNOS−/− animal exhibits a clear hypertensive phenotype. On the other hand, inhibition of prostacyclin synthesis by the coxibs leads to a slow effect on blood pressure and apparently to a more thrombotic situation (Muscara et al. 2000;FitzGerald 2003). Similarly, COX-1−/− and COX-2−/− animals show no change in blood pressure (Norwood et al. 2000; Cheung et al. 2002) and manipulation of COX or IP results in a prothrombotic phenotype.
Protection against decreases in the generation of constitutive NO and prostacyclin in the vasculature may prevent the development of vascular disease. In relation to NO, the most often tried interventions relate to the use of antioxidants (see Carr & Frei 2000) and the manipulation of eNOS expression by genetic means (Von der Leyen & Dzau 2001). Each of these interventions has shown promise in both animal experiments and in humans. An unexpected and highly interesting development relates to the effects of statins which, in the last few years, have been shown to increase the production of endothelial NO in endothelial cell cultures and in animals (for review see Laufs 2003). Many mechanisms have been claimed for this action. However, of interest in the context of our discussion is the fact that statins have been claimed to reduce oxidative stress by increasing the synthesis of BH4 (Hattori et al. 2002), increasing the coupling of the eNOS (Brouet et al. 2001) or reducing the activation of NADPH oxidase (Wagner et al. 2000). Reduction of oxidative stress is likely to preserve the generation of prostacyclin, and to our knowledge there is at least one report suggesting that statins also increase prostacyclin in endothelial cell cultures of human coronary arteries (Mueck et al. 2001). Studies on the transfection of COX-1 or COX-2 into endothelial and other cells, on the other hand, are at an early stage and clear results are not conclusive (Murakami et al. 1999; Shyue et al. 2001). The full consequences of overexpression of both NO and prostacyclin in the vasculature remain to be investigated.
Also relevant to this discussion are studies of the role that NO and prostacyclin play in the protection of the cardiovascular system provided by oestrogens, and therefore in the difference between genders in susceptibility to cardiovascular disease. Oestrogens increase the expression and the activity of eNOS (Weiner et al. 1994; Yang et al. 2000) and the activity of the COX-2 enzyme (Akarasereenont et al. 2000;Egan et al. 2004). They could therefore reduce oxidative stress by simply increasing both mediators. Alternatively, it has been claimed that oestrogens increase the efficiency of the NO synthase, thus reducing free radical formation (Barbacanne et al. 1999).
In summary, the concept of the balance between prostacyclin and TXA2 has to be expanded to include NO. Furthermore, although not discussed in this review, the way in which these compounds interact with many other systems known to be involved in vessel wall physiology and pathophysiology requires further investigation. Both prostacyclin and NO synergize in the protection of the vessel wall. TXA2, however, lies on the negative side of this balance being responsible for, among other things, platelet aggregation and vasoconstriction. The investigation into the interplay between these three molecules is just beginning. This is a sobering thought when one is contemplating probably close to 100 000 papers and over 30 years of research! However, it is clear that the discoveries of prostacyclin and NO have transformed our comprehension of vascular physiology and opened avenues for further understanding of pathophysiological processes. This knowledge has already benefited clinical medicine and no doubt will continue providing clues that will guide future therapy and prevention of vascular disease. I have had the good fortune to be intimately involved with both discoveries. More importantly, many of the colleagues that I have interacted with in the process of doing this work have become life-long personal friends. To those with whom I have managed to combine scientific excitement with friendship I owe a double debt of gratitude.
000 papers and over 30 years of research! However, it is clear that the discoveries of prostacyclin and NO have transformed our comprehension of vascular physiology and opened avenues for further understanding of pathophysiological processes. This knowledge has already benefited clinical medicine and no doubt will continue providing clues that will guide future therapy and prevention of vascular disease. I have had the good fortune to be intimately involved with both discoveries. More importantly, many of the colleagues that I have interacted with in the process of doing this work have become life-long personal friends. To those with whom I have managed to combine scientific excitement with friendship I owe a double debt of gratitude.