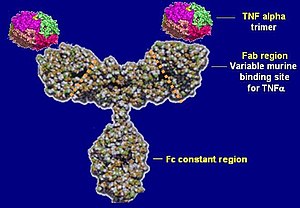
struktura infliximabu (Photo credit: Wikipedia)
Larry H Bernstein, MD, FCAP, Reporter
GI Disease, inflammation, elastase-inhibitor, membrane junctions and fatty acids
Sci Transl Med 2012; 4(158): 158ra144
Sci. Transl. Med. DOI: 10.1126/scitranslmed.3004212
RESEARCH ARTICLE
INFLAMMATORY BOWEL DISEASES
Food-Grade Bacteria Expressing Elafin Protect Against Inflammation and Restore Colon Homeostasis
Jean-Paul Motta1,2,3,*, Luis G. Bermúdez-Humarán4,*, Céline Deraison1,2,3, Laurence Martin1,2,3, Corinne Rolland1,2,3, Perrine Rousset1,2,3, Jérôme Boue1,2,3, Gilles Dietrich1,2,3, Kevin Chapman5, Pascale Kharrat4, Jean-Pierre Vinel3,6, Laurent Alric3,6, Emmanuel Mas1,2,3,7, Jean-Michel Sallenave8,9,10, Philippe Langella4,* and Nathalie Vergnolle1,2,3,5,†
1INSERM, U1043, Centre de Physiopathologie de Toulouse Purpan (CPTP), Toulouse F-31300, France.
2CNRS, U5282, Toulouse F-31300, France.
3CPTP, Université de Toulouse, Université Paul Sabatier (UPS), Toulouse F-31300, France.
4Institut National de la Recherche Agronomique (INRA), UMR 1319 Micalis, Commensal and Probiotics-Host Interactions Laboratory, Domaine de Vilvert, 78352 Jouy-en-Josas Cedex, France.
5Department of Physiology and Pharmacology, Faculty of Medicine, University of Calgary, Calgary, Alberta T2N 4N1, Canada.
6Pôle Digestif, CHU Purpan, Toulouse F-31059, France.
7Division of Gastroenterology, Hepatology and Nutrition, Children’s Hospital, Toulouse F-31059, France.
8Institut Pasteur, Unité de Défense Innée et Inflammation, Paris F-75015, France.
9INSERM U874, Paris F-75724, France.
10Universite Paris Diderot, Sorbonne Paris Cite, Cellule Pasteur F-75013, France.
ABSTRACT
Elafin, a natural protease inhibitor expressed in healthy intestinal mucosa, has pleiotropic anti-inflammatory properties in vitro and in animal models. We found that mucosal expression of Elafin is diminished in patients with inflammatory bowel disease (IBD). This defect is associated with increased elastolytic activity (elastase-like proteolysis) in colon tissue. We engineered two food-grade strains of lactic acid bacteria (LAB) to express and deliver Elafin to the site of inflammation in the colon to assess the potential therapeutic benefits of the Elafin-expressing LAB. In mouse models of acute and chronic colitis, oral administration of Elafin-expressing LAB decreased elastolytic activity and inflammation and restored intestinal homeostasis. Furthermore, when cultures of human intestinal epithelial cells were treated with LAB secreting Elafin, the inflamed epithelium was protected from increased intestinal permeability and from the release of cytokines and chemokines, both of which are characteristic of intestinal dysfunction associated with IBD. Together, these results suggest that oral delivery of LAB secreting Elafin may be useful for treating IBD in humans.
Copyright © 2012, American Association for the Advancement of Science
Citation: J.-P. Motta, L. G. Bermúdez-Humarán, C. Deraison, L. Martin, C. Rolland, P. Rousset, J. Boue, G. Dietrich, K. Chapman, P. Kharrat, J.-P. Vinel, L. Alric, E. Mas, J.-M. Sallenave, P. Langella, N. Vergnolle, Food-Grade Bacteria Expressing Elafin Protect Against Inflammation and Restore Colon Homeostasis. Sci. Transl. Med. 4, 158ra144 (2012).

Cytokines involved in IBD (Photo credit: Wikipedia)
Metabolism
Front. Physio., 10 October 2012 | doi: 10.3389/fphys.2012.00401
Outlook: membrane junctions enable the metabolic trapping of fatty acids by intracellular acyl-CoA synthetases
Joachim Füllekrug*, Robert Ehehalt and Margarete Poppelreuther
Molecular Cell Biology Laboratory, Internal Medicine IV, University of Heidelberg, Heidelberg, Germany
The mechanism of fatty acid uptake is of high interest for basic research and clinical interventions. Recently, we showed that mammalian long chain fatty acyl-CoA synthetases (ACS) are not only essential enzymes for lipid metabolism but are also involved in cellular fatty acid uptake. Overexpression, RNAi depletion or hormonal stimulation of ACS enzymes lead to corresponding changes of fatty acid uptake. Remarkably, ACS are not localized to the plasma membrane where fatty acids are entering the cell, but are found instead at the endoplasmic reticulum (ER) or other intracellular organelles like mitochondria and lipid droplets. This is in contrast to current models suggesting that ACS enzymes function in complex with transporters at the cell surface. Drawing on recent insights into non-vesicular lipid transport, we suggest a revised model for the cellular fatty acid uptake of mammalian cells which incorporates trafficking of fatty acids across membrane junctions. Intracellular ACS enzymes are then metabolically trapping fatty acids as acyl-CoA derivatives. These local decreases in fatty acid concentration will unbalance the equilibrium of fatty acids across the plasma membrane, and thus provide a driving force for fatty acid uptake.
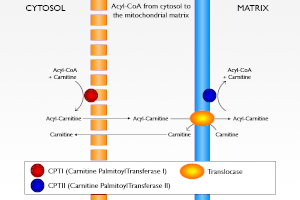
English: Acyl-CoA from the cytosol to the mitochondrial matrix. Français : Transport de l’Acyl-CoA du Cytosol jusqu’à la matrice mitochondriale. (Photo credit: Wikipedia)
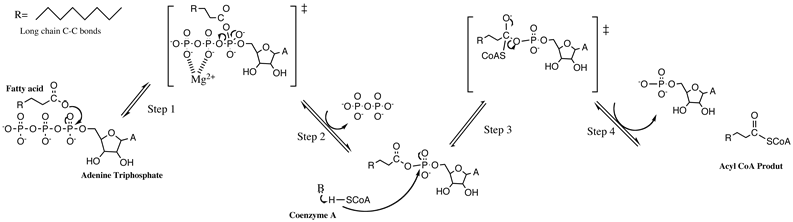
English: The mechanism for Long Chain Fatty Acyl-CoA Synthetase (Photo credit: Wikipedia)
Like this:
Like Loading...
Read Full Post »






