Curated by: Dr. Venkat S. Karra, Ph.D.
 Systemic lupus erythematosus (SLE) is a systemic autoimmune disease resulting in chronic activation of self-reactive lymphocytes and pro-inflammatory myeloid cells. SLE may also be caused by certain drugs called drug-induced lupus erythematosus. People with SLE have abnormal deposits in the kidney cells. This leads to a condition called lupus nephritis. Patients with this condition may eventually develop kidney failure and need dialysis or a kidney transplant. The underlying cause of autoimmune diseases is not fully known and so far there is no cure for SLE.
Systemic lupus erythematosus (SLE) is a systemic autoimmune disease resulting in chronic activation of self-reactive lymphocytes and pro-inflammatory myeloid cells. SLE may also be caused by certain drugs called drug-induced lupus erythematosus. People with SLE have abnormal deposits in the kidney cells. This leads to a condition called lupus nephritis. Patients with this condition may eventually develop kidney failure and need dialysis or a kidney transplant. The underlying cause of autoimmune diseases is not fully known and so far there is no cure for SLE.
SLE effects multiple end organs including the kidneys, brain, joints and skin and causes damage to many different parts of the body, including:
1. Blood clots in the legs (deep vein thrombosis) or lungs (pulmonary embolism)
2. Destruction of red blood cells (hemolytic anemia) or anemia of chronic disease
3. Fluid around the heart, pericarditis, endocarditis or inflammation of the heart (myocarditis)
4. Fluid around the lungs (pleural effusions) and damage to lung tissue
5. Pregnancy complications, including miscarriage
6. Stroke
7. Severely low blood platelets (thrombocytopenia)
8. Inflammation of the blood vessels
The molecular basis for the various manifestations of this autoimmune disease and the impact of the systemic autoimmune process on basic metabolic processes in the body are currently obscure.
However, recently a metabolomic study was executed first to understand the metabolic disturbances that underlie systemic lupus erythematosus (SLE). The study compared the sera of 20 SLE patients against that of healthy controls, using LC/MS and GC/MS platforms. Validation of key differences was performed using an independent cohort of 38 SLE patients and orthogonal assays.
The SLE metabolome exhibited profound lipid peroxidation, reflective of oxidative damage. Deficiencies were noted in the cellular anti-oxidant, glutathione, and all methyl group donors, including cysteine, methionine, and choline, as well as phosphocholines.
SLE sera showed evidence of profoundly dampened glycolysis, Krebs cycle, fatty acid β oxidation and amino acid metabolism, alluding to reduced energy biogenesis from all sources.
Whereas long-chain fatty acids, including the n3 and n6 essential fatty acids, were significantly reduced, medium chain fatty acids and serum free fatty acids were elevated.
The best discriminators of SLE included elevated lipid peroxidation products, MDA, gamma-glutamyl peptides, GGT, leukotriene B4 and 5-HETE.
Comprehensive profiling of the SLE metabolome reveals evidence of heightened oxidative stress, inflammation, reduced energy generation, altered lipid profiles and a pro-thrombotic state.
From this study it is evident that first supplementing the diet with essential fatty acids, vitamins and methyl group donors offers novel opportunities for disease modulation in this disabling systemic autoimmune ailment.
Second quickly identifying selected molecules/ therapies is another opportunity to resetting the SLE metabolome. One such opportunity is to use adrenocorticotropic hormone (ACTH) analogue.
With Prednisone, up to 90% of adults with minimal change disease (MCD) will respond to initial therapy and may require further immunosuppression. But with diseases such as idiopathic membranous nephropathy (iMN) and focal segmental glomerulosclerosis (FSGS), for which first-line therapies produce substantially lower response rates than for MCD and physicians are often compelled to use second-, third-, and even fourth-line therapies to achieve remission.
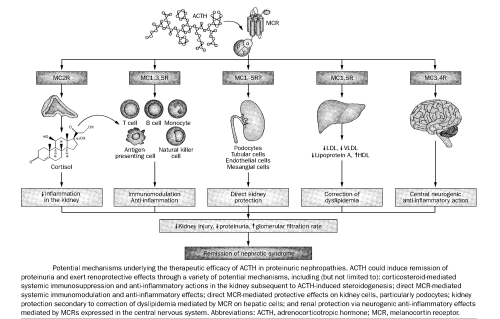
ACTH usage is not new, it was widely used way back in 1950s for the treatment of childhood nephrotic syndrome. Now there is a renewed interest in using ACTH as treatment for nephrotic syndrome as a second, third or even fourth line treatment, particularly in patients who are resistant to conventional therapies.
Subsequent clinical studies demonstrated that ACTH has prominent antiproteinuric and renoprotective effects that are not entirely explained by steroidogenic actions.
Adrenocorticotropic hormone (ACTH), also known as corticotropin, is a polypeptide tropic hormone produced and secreted by the anterior pituitry gland. It is an important component of the hypothalamic-pituitary-adrenal axis (HPA) and is often produced in response to biological stress. Its principal effects are increased production and release of corticosteriods. HPA is a complex set of direct influences and feedbackk interactions among the hypothalamus, the pituitary gland and the adrenal glands.
A deficiency of ACTH is a cause of secondary adrenal insufficiency and an excess of it is a cause of Cushing’s syndrome.
Steroid hormones ( steriod that acts as a hormone) can be grouped into five groups by the receptors to which they bind: glycocorticoids, mineralcarticoids, androgens, estrogens, and progestrogens.
Steroid hormones help control metabolism, inflammation, immune functions, salt and water balance, development of sexual characteristics, and the ability to withstand illness and injury.
As a potent physiological agonist of melanocortin system that could directly target renal parenchymal cells, such as podocytes, ACTH might serve as a promising therapy for nephrotic glomerulopathies (a disease affecting the renal glomeruli – inflammatory or non-inflammatory).
Mineralocorticoids are hormones that were involved in the retention of sodium. The primary endogenous mineralocorticoid is aldosterone. Aldosterone acts on the kidneys to provide active reabsorption of sodium and an associated passive reabsorption of water, as well as the active secretion of potassium in the principal cells of the cortical collecting tubule and active secretion of protons via proton ATPases in the lumenal membrane of the intercalated cells of the collecting tubule. This in turn results in an increase of blood pressure and blood volume.
Aldosterone is produced in the cortex of the adrenal gland and its secretion is mediated principally by angiotensin II but also by adrenocorticotropic hormone (ACTH) and local potassium levels.
Aldosterone and cortisol (a glucosteroid) have similar affinity for the mineralocorticoid receptor; however, glucocorticoids circulate at roughly 100 times the level of mineralocorticoids. Glucocorticoid concentrations are a balance between production under the negative feedback control and diurnal rhythm of the HPA axis, and peripheral metabolism, for example by the enzyme 11beta-hydroxysteroid dehydrogenase type1 (11B-HSD1), which catalyses the reduction of inactive cortisone (11-DHC in mice) to cortisol (corticosterone in mice). Reductase activity is conferred upon 11B-HSD1 by hexose-6-phosphate dehydrogenase (H6PDH). 11B-HSD1 is implicated in the development of obesity.
Knock out of H6PDH resulted in a substantial increase in urinary DHC metabolites in males (65%) and females (61%). Knock out of 11B-HSD1 alone or in combination with H6PDH led to a significant increase (36% and 42% respectively) in urinary DHC metabolites in females only. Intermediate 11B-HSD1/H6PDH heterozygotes maintained a normal HPA axis.
Urinary steroid metabolite profile by GC/MS as a biomarker assay may be beneficial in assaying HPA axis status clinically in cases of congenital and acquired 11B-HSD1/H6PDH deficiency
ACTH acts through the stimulation of cell surface ACTH receptors, which are located primarily on adrenocortical cells of the adrenal cortex. This results in the synthesis and secretion of gluco- and mineralo-corticosteriods and androgenic steroids.
An enzyme exists in mineralocorticoid target tissues to prevent overstimulation by glucocorticoids. This enzyme, 11-beta hydroxysteriod dehydrogenase type II (protein: HSD11B2), catalyzes the deactivation of glucocorticoids to 11-dehydro metabolites.
ACTH acts at several key steps to influence the steroidogenic pathway in the adrenal cortex:
ACTH stimulates lipoprotein uptake into cortical cells. This increases the bio-availability of cholestrol in the cells of the adrenal cortex.
ACTH increases the transport of cholesterol into the mitochondria and activates its hydrolysis.
ACTH Stimulates cholesterol side-chain cleavage enzyme, which makes the rate-limiting step in steroidogenesis. This results in the production of pregnenolone.
Receptor-binding studies have revealed that mineralcorticoids show a strong affinity for ACTH thereby establishing the potential for this hormone to activate mineralocorticoid receptors (MCRs). There are five MCRs and all of them show affinity for ACTH.
MCRs are expressed in kidney cells and that indicates that kidney is a target organ for the affects of ACTH.
Functions include:
1. Steroidogenic and adrenotropic activity
2. A multifaceted extra adrenal action that is mediated by the different MCRs present in the peripheral tissues and CNS
3. Has a lipostatic effect and stimulates lipolysis – (thus ACTH deficiency leads to obesity)
4. Its administration lowers levels of plasma lipids including Triglycerides, Total cholestrol, LDL-cholestrol and phospholipids
5. Its administration (complete ACTH molecule) rapidly increases the plasma insulin
Other activities include:
1. regulation of skin and hair pigmentation,
2. modulation of sebacious gland function and
3. anti-inflammatory and immunomodulatory functions
The total adrenocorticotropic hormone (ACTH) analogue is available as H.P. Acthar Gel (repository corticotropin injection) and is used for:
1. Monotherapy treatment of infantile spasms (IS) in infants and children under 2 years of age.
2. The treatment of exacerbations of multiple sclerosis in adults.
3. For inducing a diuresis or a remission of proteinuria in the nephrotic syndrome without uremia of the idiopathic type or that due to lupus erythematosus.
4. Also:: rheumatic disorders; collagen diseases; dermatologic diseases; allergic states; ophthalmic diseases and respiratory diseases.
FDA approved indications for the above prodcut are available at the following URL:
http://www.ncbi.nlm.nih.gov/pmc/articles/PMC2697107/table/t1-ptj34_5p250/
Disclaimer: This is for information purpose only, not a medical advise.
For a full list of warnings, precautions, and adverse events related to Acthar, please refer to the full Prescribing Information including the Medication Guide for the treatment of Infantile Spasms and discuss this information with your healthcare provider.
Literature:
The renaissance of corticotropin therapy in proteinuric nephropathies
Metabolic Disturbances Associated with Systemic Lupus Erythematosus
H.P. Acthar Gel and Cosyntropin Review
Childhood nephrotic syndrome—current and future therapies



Dr. Kara,
Thank you for an excellent review and presentation of the topic. The mechanism of action of the drug ‘Acthar’ for Systemic Lupus Erythematosus (SLE) is a very important topic of research and this drug is used for patients that have exhausted the benefits of prednisone which sustaining the side efects of this drug which is a results of dosage and duration of use. Any pharmacotherapy that provides an alternative regimen is of very great clinical importance. Thus, bringing this post to the online reader in the context of SLE represents a contribution.
[…] http://pharmaceuticalintelligence.com/2012/07/08/the-mechanism-of-action-of-the-drug-acthar-for-syst… […]
I actually consider this amazing blog , âSAME SCIENTIFIC IMPACT: Scientific Publishing –
Open Journals vs. Subscription-based « Pharmaceutical Intelligenceâ, very compelling plus the blog post ended up being a good read.
Many thanks,Annette
I actually consider this amazing blog , âSAME SCIENTIFIC IMPACT: Scientific Publishing –
Open Journals vs. Subscription-based « Pharmaceutical Intelligenceâ, very compelling plus the blog post ended up being a good read.
Many thanks,Annette
I actually consider this amazing blog , âSAME SCIENTIFIC IMPACT: Scientific Publishing –
Open Journals vs. Subscription-based « Pharmaceutical Intelligenceâ, very compelling plus the blog post ended up being a good read.
Many thanks,Annette
I actually consider this amazing blog , âSAME SCIENTIFIC IMPACT: Scientific Publishing –
Open Journals vs. Subscription-based « Pharmaceutical Intelligenceâ, very compelling plus the blog post ended up being a good read.
Many thanks,Annette
I actually consider this amazing blog , âSAME SCIENTIFIC IMPACT: Scientific Publishing –
Open Journals vs. Subscription-based « Pharmaceutical Intelligenceâ, very compelling plus the blog post ended up being a good read.
Many thanks,Annette