Statins’ Nonlipid Effects on Vascular Endothelium through eNOS Activation
Curator, Author,Writer, Reporter: Larry Bernstein, MD, FACP
Categories of Research:
Disease biology, Cell Biology and Cell Signaling, Biological Networks and Gene Regulation, Pharmacotherapy of Cardiovascular Disease, Nitric Oxide, HMG Co A inhibitors, Endothelial Receptor, Hypertension, Therapeutic Targets
Introduction
Statins have an effect on the vascular endothelium, which plays an important role in the development of atherosclerosis and angiogenesis, a role independent of the lipid lowering effect. The vascular endothelium plays an important role regulating vascular wall contraction and as a mediator for the vascular wall. Endothelial dysfunction, the hallmark of which is reduced activity of endothelial cell derived nitric oxide (NO), is a key factor in developing atherosclerosis and cardiovascular disease. Vascular endothelial cells play a pivotal role in modulation of leukocyte and platelet adherence, thrombogenicity, anticoagulation, and vessel wall contraction and relaxation, so that endothelial dysfunction has become almost a synonym for vascular disease. A single layer of endothelial cells is the only constituent of capillaries, which differ from other vessels, which contain smooth muscle cells and adventitia. Capillaries directly mediate nutritional supply as well as gas exchange within all organs. The failure of the microcirculation leads to tissue apoptosis/necrosis. expanded cultured EPC transplantation and cytokine-induced EPC mobilization from bone marrow have been shown to enhance angiogenesis with significant improvement of microcirculation in ischemic tissue.
It has been generally assumed that cholesterol reduction by statins mechanism underlying their beneficial effects in cardiovascular disease. The statins — potent inhibitors of 3-hydroxy-3-methylglutaryl coenzyme A (HMG-CoA) reductase, an enzyme that plays a critical role in cholesterol metabolism — block substrate accessibility to HMG-CoA reductase , effectively subverting cholesterol metabolism. Sufficient evidence now supports a hypothesis that cholesterol-independent or “pleiotropic” effects of statins improve endothelial dysfunction, effects on angiogenesis, and reduce vascular inflammation. The statins’ cholesterol-independent vascular effects appear to directly restore or improve endothelial function by increasing NO production, promote endothelial repair after arterial injury, and decrease vascular inflammation. Statins improve endothelial function by:
- increasing production of nitric oxide,
- promoting blood flow,
- dampening inflammation,
- antagonizing thrombogenicity, and
- reducing endothelial vasoresponses.
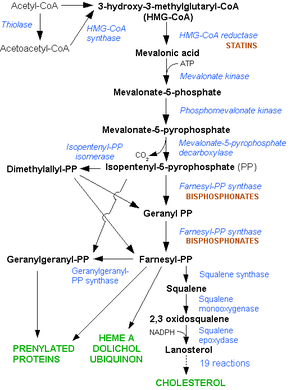
The HMG-CoA reductase pathway, which is blocked by statins via inhibiting the rate limiting enzyme HMG-CoA reductase. (Photo credit: Wikipedia)
We review effects of statins on endothelial cells and endothelial progenitor cells that identifies a novel therapeutic potential of statin drugs.
- Evidence in support of the new “pleiotrophic” non-lipid effects of Statins
- Endothelial cell progenitors leave the bone marrow in response to cytokines or ischemic Injury.
- They proliferate, migrate, and acquire resistance to apoptotic cell death.
Transplanting mice with the bone marrow of a transgenic animal carrying the LacZ reporter gene under control of the Tie2 promoter, which is active in endothelial cells…showed that statin-treated animals accumulate marrow-derived endothelial cells at the site of corneal neovascularization, administering statins is probably safer than giving VEGF to promote angiogenesis or vasculogenesis.
- Akt activation has emerged as an indispensable signaling gateway at the crossroads between angiogenesis and endothelial stem cell recruitment and differentiation
- Placental growth factor, which seems preferentially involved in facilitating postnatal blood vessel formation, is another “vasculogenic factor” that acts very much like the statins
- Increase in endothelial nitric oxide synthase expression and activity is clearly stimulated by statins, which results in Akt activation a multifaceted developmental pathway of stem cell mobilization and differentiation is exploited by statins
Altieri DC. Statins’ benefits begin to sprout. J. Clin. Invest. 108:365–366 (2001). DOI:10.1172/JCI200113556
“Pleiotropic” Effect of statins
Recent studies have shown the restoration of endothelial function before significant reduction of serum cholesterol levels effect of statins on the endothelium were first defined by their ability to enhance endothelial NO production, upregulating endothelial nitric oxide synthase (eNOS) PI3 kinase/Akt signaling, which is a crucial regulator of cell metabolism and apoptosis, appears to mediate statin-induced eNOS upregulation.
The mechanism of eNOS activation by phosphorylation by statins
Statins can also inhibit Rho isoprenylation/activation resulting in enhanced eNOS mRNA stability and increased eNOS expression statins inhibit ox-LDL-induced endothelin-1 (ET-1) expression and the biological function of angiotensin II, and its receptor subtype 1 (AT1), which are both potent vasoconstrictors/mitogens thought to contribute to the development of atherosclerotic lesions.
Vascular inflammation
Statins have been shown to reduce the number of inflammatory cells in atherosclerotic lesions. Inhibitory effects of statins on adhesion molecules such as intercellular adhesion molecule-1 (ICAM-1), vascular cell adhesion molecule (VCAM-1) and E-selectin, which are involved in the adhesion/rolling/extravasation of inflammatory cells.
Statin therapy in humans has also been shown to lower high-sensitivity C-Reactive Protein (hs-CRP), which reflects low-grade systemic/vascular inflammation, in hypercholesterolemic patients. This has been shown to correlate with reductions in the rates of acute major or recurrent coronary events.
Re-endothelialization
Accelerated re-endothelialization after angioplasty/de-endothelialization is known to inhibit neointimal hyperplasia, which leads to luminal narrowing or restenosis at the injured site. Re-endothelialization has been shown to be promoted by vascular endothelial growth factor (VEGF), hepatocyte growth factor (HGF), estrogen, prostacyclin, blockade of TNFα, and now Statins.
Ii M, Losordo DW. Statins and the endothelium. Vascular Pharmacology 2007; 46: 1–9.
Altieri DC. Statins’ benefits begin to sprout. J. Clin. Invest. 108:365–366 (2001). DOI:10.1172/JCI200113556
Further observations
- Statins exert cholesterol-independent effects on the endothelium, which lead to the improvement of endothelial function.
- Statins exert biphasic, dose dependent effects on angiogenesis. At low doses, statins induce angiogenesis, whereas angiogenesis is inhibited at higher doses. These biphasic activities of statins on endothelial cell biology can be explained by the properties of the biosynthetic pathways that originate from mevalonic acid.
- It appears that low concentrations of statins (such as those achieved in vivo) induce pro-angiogenic effects through activating PI3 kinase/Akt signaling leading to eNOS phosphorylation and NO production.
- High (supra-physiologic) concentrations of statins will inhibit the synthesis of the non-sterol products mevalonate, leading to decreases in protein prenylation, inhibition of cell growth, or apoptosis.
The sum-up of two factors: the loss of the vascular relaxation directly dependent of the endothelium (flow – dependent) and the NO dependent are the main reason for endothelial dysfunction and play a very important role in the pathogenesis of heart failure.
- Endothelial dysfunction on vascular peripheral levels contributes to the increased peripheral resistance in patients with heart failure. Endothelial dysfunction, as a pathophysiology disorder, is present early.
- Statins’ benefits begin to sprout in the initiation of the atherosclerotic process.
- The injury of eNOS activity seems to occur with impaired coronary vasodilatation in response to acetylcholine in patients with hypertension, hypercholesterolemia, diabetes, smokers.
Summary of Key Points
Mechanisms which are essential for the impairment of eNOS activity for the appearance of endothelial dysfunction are:
• dysfunctional signal transduction receptor – endothelial cell;
• decreased bioavailability of the substrate L- arginine;
• altered expression of gene NOS3 and stability of mARN; polymorphism NOS3;
• altered eNOS activity;
• increased destruction of NO;
• changes in the balance between NO derived endothelium and the hyperpolarizing factor (EDHF);
• decreased sensitivity of atherosclerotic smooth muscle to NO.
Effects other than those due to lowering LDL levels and independent of the LDL level
• improved endothelial function
• diminish vascular inflammation
• improve ventricular function of heart failure
• antithrombotic effect
• reduce the rate of vascular events
• antioxidant effect
Statins improve endothelial function through the following mechanisms:
• enhanced endothelial NO production by decrease of cholesterol, by up regulating posttranscriptional mRNA of eNOS and by antioxidative effects (reduction of reactive oxygen species, increase of super oxide elimination and decrease of oxidized LDL);
• reduced production of endothelin-1, endothelial vasoconstrictor factor;
• diminish the affinity for AT1 receptors ;
• stimulation of angiogenesis through proliferation, migration and survival of the circulating endothelial progenitor cells
Statins decrease the swell of the vascular wall by:
• decreasing the level of C – Reactive Protein
• decreasing the synthesis of proinflammatory cytokines (IL-1, IL-6, IL-8, TNF α)
• diminishing the leukocyte adhesion to endothelial cells inhibiting macrophage growth and smooth muscle cell migration and proliferation
Suciu M. The Role Of Nitric Oxide (No) And Statins In Endothelial Dysfunction And Atherosclerosis. Farmacia 2009; 57 (2): 131-139
Relevant observations
ECs treated with rosuvastatin increase eNOS activation. The increased NO production is involved in modulating S-nitrosylation and translation of proteins.
Bin Huang, Fu An Li, Chien Hsing Wu, Danny Ling Wang. The role of nitric oxide on rosuvastatin-mediated S-nitrosylation and translational proteomes in human umbilical vein endothelial cells. Proteome Science 2012, 10:43. doi:10.1186/1477-5956-10-43
Emerging evidence from both clinical trials and basic science studies suggest that statins have anti-inflammatory properties, which may additionally lead to clinical efficacy. Measurement of markers of inflammation such as high sensitivity C-Reactive Protein in addition to lipid parameters may help identify those patients who will benefit most from statin therapy.
Blake GJ and Ridker PM. Are statins anti-inflammatory? Curr Control Trials Cardiovasc Med 2000, 1:161–165.
Most favorable and unexpected findings were:
- new indications for TDZs as stimulators of eNOS, in addition to the new indication for atherosclerosis besides the classic indication in pharmacology books, being in the reduction of insulin resistance.
- new indications for beta blockers as NO stimulant, nebivolol, a case in point, thus, fulfilling two indications in one drug along the direction of the study to identify eNOS agonists. Nebivolol is a vasodilator, thus functions as an antihypertensive.
Aviva Lev-Ari. Cardiovascular Disease (CVD) and the Role of agent alternatives in endothelial Nitric Oxide Synthase (eNOS) Activation and Nitric Oxide Production. July 19, 2012 pharmaceuticalintelligence.com
References
Heeba G, Hassan MK, Khalifa, M; Malinski T. Adverse Balance of Nitric Oxide/ Peroxynitrite in the Dysfunctional Endothelium Can be Reversed by Statins. Journal of Cardiovascular Pharmacology. 2007; 50(4):391-398.
Tandon VR, Gupta BM, Tandon R. Non-lipid Actions of Statins. JK Science 2004; 6(3): 124-126.
Sacks FM. Do statins play a role in the early management of the acute coronary syndrome? European Heart Journal Supplements (2004) 6 (Supplement A), A32–A36.
Alonso D, Radomski MW. Nitric oxide, platelet function, myocardial infarction and reperfusion therapies. Heart Fail Rev 2003; 8:47–54.
Cardiovascular Disease (CVD) and the Role of agent alternatives in endothelial Nitric Oxide Synthase (eNOS) Activation and Nitric Oxide Production. PharmaceuticalIntelligence.WordPress.com
Nitric oxide and signalling pathways. PharmaceuticalIntelligence.WordPress.com
Rationale of NO use in hypertension and heart failure. PharmaceuticalIntelligence.WordPress.com
LH Bernstein. Mitochondria: Origin from oxygen free environment, role in aerobic glycolysis, metabolic adaptation in cancer (Warburg effect). PharmaIntell.Wordpress.com
R Saxena. Mitochondria: More than just the powerhouse of the cell. PharmaIntell.WordPress.com
Bernstein LH. Expanding the Genetic Alphabet and linking the genome to the metabolome. PharmaIntell.wordpress.com. luly 24, 2012.
R saxena. β Integrin emerges as an important player in mitochondrial dysfunction associated Gastric Cancer. PharmaIntell.wordpress.com 2012
Related articles
- Interaction of Nitric Oxide and Prostacyclin in Vascular Endothelium (pharmaceuticalintelligence.com)
- Angiogenesis Impairment in Diabetes: Role of Methylglyoxal-Induced Receptor for Advanced Glycation Endproducts, Autophagy and Vascular Endothelial Growth Factor Receptor 2 (plosone.org)
- Endothelial Dysfunction, Diminished Availability of cEPCs, Increasing CVD Risk for Macrovascular Disease – Therapeutic Potential of cEPCs (pharmaceuticalintelligence.com)
- Inhibition of Proliferation, Migration and Proteolysis Contribute to Corticosterone-Mediated Inhibition of Angiogenesis (plosone.org)
- Nitric Oxide has a ubiquitous role in the regulation of glycolysis -with a concomitant influence on mitochondrial function (pharmaceuticalintelligence.com)
- Nitric Oxide Covalent Modifications: A Putative Therapeutic Target? (pharmaceuticalintelligence.com)
- Discovery of nitric oxide and its role in vascular biology (pharmaceuticalintelligence.com)
Interesting observations of statins especially effect of statins of ICAM1, ET-1 expression in vascular endothelia. I would also be interested in further information of statins effect on the intracellular isoprenyl pools, as you have mentioned that statins may affect Rho-dependent GTPases and further studies on the mechanism by which statins increase eNOS activity? Interesting posting.
Dr. Williams,
Thank you for your comment. I would like to invite you to continue the research I started in 2006. I have THREE more posts and I am done.
Please find below, the trove of pearls, and many very creative thoughts.
Please start with
http://pharmaceuticalintelligence.com/2012/04/30/93/
Curator: Cardiovascular Pharmacological Therapy
Lev-Ari, A., (2012W). Endothelin Receptors in Cardiovascular Diseases: The Role of eNOS Stimulation
http://pharmaceuticalintelligence.com/2012/10/04/endothelin-receptors-in-cardiovascular-diseases-the-role-of-enos-stimulation/
Lev-Ari, A., (2012V). Inhibition of ET-1, ETA and ETA-ETB, Induction of NO production, stimulation of eNOS and Treatment Regime with PPAR-gamma agonists (TZD): cEPCs Endogenous Augmentation for Cardiovascular Risk Reduction – A Bibliography
http://pharmaceuticalintelligence.com/2012/10/04/inhibition-of-et-1-eta-and-eta-etb-induction-of-no-production-and-stimulation-of-enos-and-treatment-regime-with-ppar-gamma-agonists-tzd-cepcs-endogenous-augmentation-for-cardiovascular-risk-reduc/
Lev-Ari, A., (2012U). Cardiovascular Outcomes: Function of circulating Endothelial Progenitor Cells (cEPCs): Exploring Pharmaco-therapy targeted at Endogenous Augmentation of cEPCs
http://pharmaceuticalintelligence.com/2012/08/28/cardiovascular-outcomes-function-of-circulating-endothelial-progenitor-cells-cepcs-exploring-pharmaco-therapy-targeted-at-endogenous-augmentation-of-cepcs/
Lev-Ari, A., (2012T). Endothelial Dysfunction, Diminished Availability of cEPCs, Increasing CVD Risk for Macrovascular Disease – Therapeutic Potential of cEPCs
http://pharmaceuticalintelligence.com/2012/08/27/endothelial-dysfunction-diminished-availability-of-cepcs-increasing-cvd-risk-for-macrovascular-disease-therapeutic-potential-of-cepcs/
Lev-Ari, A., (2012S). Vascular Medicine and Biology: CLASSIFICATION OF FAST ACTING THERAPY FOR PATIENTS AT HIGH RISK FOR MACROVASCULAR EVENTS Macrovascular Disease – Therapeutic Potential of cEPCs
http://pharmaceuticalintelligence.com/2012/08/24/vascular-medicine-and-biology-classification-of-fast-acting-therapy-for-patients-at-high-risk-for-macrovascular-events-macrovascular-disease-therapeutic-potential-of-cepcs/
Lev-Ari, A., (2012O). Biosimilars: Intellectual Property Creation and Protection by Pioneer and by Biosimilar Manufacturers
http://pharmaceuticalintelligence.com/2012/07/30/biosimilars-intellectual-property-creation-and-protection-by-pioneer-and-by-biosimilar-manufacturers/
Lev-Ari, A., (2012P). Biosimilars: Financials 2012 vs. 2008
http://pharmaceuticalintelligence.com/2012/07/30/biosimilars-financials-2012-vs-2008/
Lev-Ari, A., (2012Q). Biosimilars: CMC Issues and Regulatory Requirements
http://pharmaceuticalintelligence.com/2012/07/29/biosimilars-cmc-issues-and-regulatory-requirements/
Lev-Ari, A. (2012L).. Cardiovascular Disease (CVD) and the Role of agent alternatives in endothelial Nitric Oxide Synthase (eNOS) Activation and Nitric Oxide Production
http://pharmaceuticalintelligence.com/2012/07/19/cardiovascular-disease-cvd-and-the-role-of-agent-alternatives-in-endothelial-nitric-oxide-synthase-enos-activation-and-nitric-oxide-production/
Lev-Ari, A. (2012a). Resident-cell-based Therapy in Human Ischaemic Heart Disease: Evolution in the PROMISE of Thymosin beta4 for Cardiac Repair
http://pharmaceuticalintelligence.com/2012/04/30/93/
Lev-Ari, A. (2012b). Triple Antihypertensive Combination Therapy Significantly Lowers Blood Pressure in Hard-to-Treat Patients with Hypertension and Diabetes
http://pharmaceuticalintelligence.com/2012/05/29/445/
Lev-Ari, A. (2012h). Macrovascular Disease – Therapeutic Potential of cEPCs: Reduction Methods for CV Risk
http://pharmaceuticalintelligence.com/2012/07/02/macrovascular-disease-therapeutic-potential-of-cepcs-reduction-methods-for-cv-risk/
Lev-Ari, A. (2012j) Mitochondria Dysfunction and Cardiovascular Disease – Mitochondria: More than just the “powerhouse of the cell”
http://pharmaceuticalintelligence.com/2012/07/09/mitochondria-more-than-just-the-powerhouse-of-the-cell/
Lev-Ari, A. (2012i). Bystolic’s generic Nebivolol – positive effect on circulating Endothelial Proginetor Cells endogenous augmentation
http://pharmaceuticalintelligence.com/2012/07/16/bystolics-generic-nebivolol-positive-effect-on-circulating-endothilial-progrnetor-cells-endogenous-augmentation/
Lev-Ari, A. & Abourjaily, P. (2006a) “An Investigation of the Potential of circulating Endothelial Progenitor Cells (cEPC) as a Therapeutic Target for Pharmacologic Therapy Design for Cardiovascular Risk Reduction.”Part I: Macrovascular Disease – Therapeutic Potential of cEPCs – Reduction methods for CV risk. Part II: (2006b) Therapeutic Strategy for cEPCs Endogenous Augmentation: A Concept-based Treatment Protocol for a Combined Three Drug Regimen. Part III: (2006c) Biomarker for Therapeutic Targets of Cardiovascular Risk Reduction by cEPCs Endogenous Augmentation USING NEW COMBINATION DRUG THERAPY OF THREE DRUG CLASSES AND SEVERAL DRUG INDICATIONS. Northeastern University, Boston, MA 02115
Heart Vasculature (2007) Regeneration and Protection of Coronary Artery Endothelium and Smooth Muscle: A Concept-based Pharmacological Therapy of a Combined Three Drug Regimen. Bouve College of Health Sciences, Northeastern University, Boston, MA 02115
Dr. Larry,
Thank you for all the clarity you have beautifully expressed following a marvelous synthesis of this area seeded with complex biochemical interactions and dependencies.
Just completed reading this post edifying the Public on the virtues of Statin as one generic drug that is implicated with a very wide array of benefits as a therapeutic agent for multiple indications, chiefly, in the amelioration of negative metabolic effects on cardiovascular disease. In the modern history of Pharmacology, there was only one other agent, that in addition to its original indication, it was implicated with a wealth of benefits as a therapeutic agent, again exerting favorable effects on the Cardiovascular system, it was named in the late 20s, Aspirin, the ASA of the NSAID class.
Long term use of Statins were found to have negative non reversible effects on the muscular skeletal system.
Long term use of Aspirin is documented to have the potential of causing internal bleeding and in cases of patients with evolving yet undiagnosed blood discreasia or diagnosed coagulation disorder with sudden necessity for an unplanned emergency surgical intervention – it is the 14 days call of non use needed to clear this compound from the system – in some cases this condition is been violated.
From Statins and Aspirin, both generics, at the lowest cost and at the widest population in use, the medical trade offs are been brought to bear in addressing the prevalence of search for treatment for Cardiovascular diseases.
In the case of both drugs, the benefits out weight the risk. Both, the enterocoated 81mg Aspirin, and the Statins, are part of the AHA Protocol of Treatment, early stage of cardiovascular disorder.
The distinction made above about the high dose vs low dose of Statins, and the opposite effects obtained in a dose-dependent regimen, one would assume the it is only, the long term use of the high dose (60mg-80mg) of daily use, would bring about the risk on muscular function distraction, as a Physician told me “my patient is now in a wheel chair, it took 20 years of Statins use, he was not on any other Med.” Though the 81mg Aspirin pill recognized for it cardio protective effects, the magic bullet is the 325mg not for the long term, some time in use on a long term basis.
In Long Term Post Acute setting, 35%-45% of patients have standing orders of BID 1000mg Tylenol for pain caused by underlying conditions of inflammation causing chronic discomfort, often PRN analgesia offered in addition to the standing order. In the elderly population, mitigation of chronic pain, may have a favorable indirect cardiovascular effect for a dysfunction treated by several antihypertensive and other cardio regulation agents.
Let’s encourage all to excercise and follow the guidelines of the Health diet.
[…] Statins’ nonlipid effects on vascular endothelium through eNOS activation (pharmaceuticalintelligence.com) […]
[…] Statins’ Nonlipid Effects on Vascular Endothelium through eNOS Activation Curator, Author,Writer, Reporter: Larry Bernstein, MD, FCAP […]
[…] Statins’ nonlipid effects on vascular endothelium through eNOS activation (pharmaceuticalintelligence.com) […]
[…] http://pharmaceuticalintelligence.com/2012/10/08/statins-nonlipid-effects-on-vascular-endothelium-th… […]
[…] http://pharmaceuticalintelligence.com/2012/10/08/statins-nonlipid-effects-on-vascular-endothelium-th… […]
[…] Statins’ nonlipid effects on vascular endothelium through eNOS activation (pharmaceuticalintelligence.com) […]
[…] Statins’ nonlipid effects on vascular endothelium through eNOS activation (pharmaceuticalintelligence.com) […]
[…] Statins’ nonlipid effects on vascular endothelium through eNOS activation (pharmaceuticalintelligence.com) […]
[…] Statins’ nonlipid effects on vascular endothelium through eNOS activation (pharmaceuticalintelligence.com) […]
[…] Statins’ nonlipid effects on vascular endothelium through eNOS activation (pharmaceuticalintelligence.com) […]
[…] Statins’ nonlipid effects on vascular endothelium through eNOS activation […]
PUT IT IN CONTEXT OF CANCER CELL MOVEMENT
The contraction of skeletal muscle is triggered by nerve impulses, which stimulate the release of Ca2+ from the sarcoplasmic reticuluma specialized network of internal membranes, similar to the endoplasmic reticulum, that stores high concentrations of Ca2+ ions. The release of Ca2+ from the sarcoplasmic reticulum increases the concentration of Ca2+ in the cytosol from approximately 10-7 to 10-5 M. The increased Ca2+ concentration signals muscle contraction via the action of two accessory proteins bound to the actin filaments: tropomyosin and troponin (Figure 11.25). Tropomyosin is a fibrous protein that binds lengthwise along the groove of actin filaments. In striated muscle, each tropomyosin molecule is bound to troponin, which is a complex of three polypeptides: troponin C (Ca2+-binding), troponin I (inhibitory), and troponin T (tropomyosin-binding). When the concentration of Ca2+ is low, the complex of the troponins with tropomyosin blocks the interaction of actin and myosin, so the muscle does not contract. At high concentrations, Ca2+ binding to troponin C shifts the position of the complex, relieving this inhibition and allowing contraction to proceed.
Figure 11.25
Association of tropomyosin and troponins with actin filaments. (A) Tropomyosin binds lengthwise along actin filaments and, in striated muscle, is associated with a complex of three troponins: troponin I (TnI), troponin C (TnC), and troponin T (TnT). In (more ) Contractile Assemblies of Actin and Myosin in Nonmuscle Cells
Contractile assemblies of actin and myosin, resembling small-scale versions of muscle fibers, are present also in nonmuscle cells. As in muscle, the actin filaments in these contractile assemblies are interdigitated with bipolar filaments of myosin II, consisting of 15 to 20 myosin II molecules, which produce contraction by sliding the actin filaments relative to one another (Figure 11.26). The actin filaments in contractile bundles in nonmuscle cells are also associated with tropomyosin, which facilitates their interaction with myosin II, probably by competing with filamin for binding sites on actin.
Figure 11.26
Contractile assemblies in nonmuscle cells. Bipolar filaments of myosin II produce contraction by sliding actin filaments in opposite directions. Two examples of contractile assemblies in nonmuscle cells, stress fibers and adhesion belts, were discussed earlier with respect to attachment of the actin cytoskeleton to regions of cell-substrate and cell-cell contacts (see Figures 11.13 and 11.14). The contraction of stress fibers produces tension across the cell, allowing the cell to pull on a substrate (e.g., the extracellular matrix) to which it is anchored. The contraction of adhesion belts alters the shape of epithelial cell sheets: a process that is particularly important during embryonic development, when sheets of epithelial cells fold into structures such as tubes.
The most dramatic example of actin-myosin contraction in nonmuscle cells, however, is provided by cytokinesisthe division of a cell into two following mitosis (Figure 11.27). Toward the end of mitosis in animal cells, a contractile ring consisting of actin filaments and myosin II assembles just underneath the plasma membrane. Its contraction pulls the plasma membrane progressively inward, constricting the center of the cell and pinching it in two. Interestingly, the thickness of the contractile ring remains constant as it contracts, implying that actin filaments disassemble as contraction proceeds. The ring then disperses completely following cell division.
Figure 11.27
Cytokinesis. Following completion of mitosis (nuclear division), a contractile ring consisting of actin filaments and myosin II divides the cell in two.
http://www.ncbi.nlm.nih.gov/books/NBK9961/
This is good. I don’t recall seeing it in the original comment. I am very aware of the actin myosin troponin connection in heart and in skeletal muscle, and I did know about the nonmuscle work. I won’t deal with it now, and I have been working with Aviral now online for 2 hours.
I have had a considerable background from way back in atomic orbital theory, physical chemistry, organic chemistry, and the equilibrium necessary for cations and anions. Despite the calcium role in contraction, I would not discount hypomagnesemia in having a disease role because of the intracellular-extracellular connection. The description you pasted reminds me also of a lecture given a few years ago by the Nobel Laureate that year on the mechanism of cell division.
I actually consider this amazing blog , âSAME SCIENTIFIC IMPACT: Scientific Publishing –
Open Journals vs. Subscription-based « Pharmaceutical Intelligenceâ, very compelling plus the blog post ended up being a good read.
Many thanks,Annette
I actually consider this amazing blog , âSAME SCIENTIFIC IMPACT: Scientific Publishing –
Open Journals vs. Subscription-based « Pharmaceutical Intelligenceâ, very compelling plus the blog post ended up being a good read.
Many thanks,Annette
I actually consider this amazing blog , âSAME SCIENTIFIC IMPACT: Scientific Publishing –
Open Journals vs. Subscription-based « Pharmaceutical Intelligenceâ, very compelling plus the blog post ended up being a good read.
Many thanks,Annette