In our recent article on “Amyloidosis” we discussed about its causes and forms. One such form is familial amyloidosis (ATTR) a most common form of inherited amyloidoses.
ATTR is caused by a mutation in the transthyretin (TTR) gene that produces abnormal transthyretin protein which deposits as amyloid fibrils.
Symptoms of disease are usually:
Neuropathy (numbness and tingling in the arms and legs, dizziness upon standing, and alternating constipation and diarrhea), and
Cardiomyopathy and occur in mid to late life.
The standard treatment is liver transplantation since the transthyretin protein which causes familial amyloidosis is made in the liver, replacing this organ removes the source of mutant protein production. A new liver will make only normal transthyretin.
It is known that the transthyretin amyloidoses (ATTR) are invariably fatal diseases characterized by above mentioned progressive neuropathy and/or cardiomyopathy. The early onset TTR amyloidoses are caused by inherited TTR mutations that weaken the tetramers’ ability to stick together, producing monomers which are more likely to aggregate into amyloids and other aggregate structures.
Transthyretin (TTR) is a tetrameric protein involved in the transport of thyroxine and the vitamin A–retinol-binding protein complex. Mutations within TTR that cause autosomal dominant forms of disease facilitate tetramer dissociation, monomer misfolding, and aggregation, although wild-type TTR can also form amyloid fibrils in elderly patients. Because tetramer dissociation is the rate-limiting step in TTR amyloidogenesis, targeted therapies have focused on small molecules that kinetically stabilize the tetramer, inhibiting TTR amyloid fibril formation.
Scientists from The Scripps Research Institute and Pfizer Inc. have published a new study showing how this type of amyloidosis can be inhibited. A new drug called tafamidis (Vyndaqel®)) has been developed to treat this deadly nerve disease caused by transthyretin (TTR) amyloid fibril formation, or the accumulation of abnormal assemblies of the TTR protein. Researchers have designed tafamidis to grab either of those thyroxine-binding sites, in a way that bridges the seam and helps keep the tetramer from coming apart.
Tafamidis binds to the natural, functional TTR structure (mutant and wild type), in a way that prevents it from deviating from this natural, functional form into the amyloid state. TTR’s natural, functional form is a “tetramer”made from four copies of the protein. Amyloidosis occurs when these tetramers come apart and the individual TTR proteins (“monomers”) undergo shape changes enabling them to misassemble into dysfunctional amyloid aggregates. Included in the TTR aggregate distribution are amyloid fibrils—protein stacks made from millions of TTR monomers—although researchers suspect that smaller, shorter-lived pre-amyloid aggregates do more direct damage to nerve cells and nerve fibers.
Tafamidis treats ATTR by reducing the rate of amyloid formation, and clinical trials have shown that it delays the typical progression of nerve destruction in polyneuropathy patients.
This drug is approved for use in Europe and currently under review by the US Food and Drug Administration (FDA), and it is the first medication approved by a major regulatory agency to treat an amyloid disease, a class of conditions that include Alzheimer’s.
Source:
http://www.scripps.edu/newsandviews/e_20120604/tafamidis.html
http://www.pnas.org/content/suppl/2012/05/29/1121005109.DCSupplemental/sapp.pdf
Curated by: Dr. Venkat S. Karra, Ph.D.
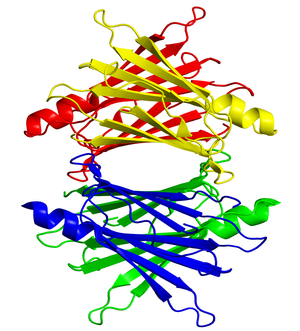

t would have on retinoids, also important in vit A metabolism.
The scientist at Scripps is a brilliant young man who presented at the 1st International TTR Symposium in Strasbourg maybe 10 years ago.
You know, this genetic disease occurs in Portugal, which there is an eminent researcher on the topic, in Sweden, and in Japan, and of course, Brazil, where they do a lot of transplants.
Yves Ingenbleek wanted to have a 2nd Congress 7 years ago, but I couldn’t put it together then.
I’m not sure that the amyloid related to diabetes (pancreatic islets), and the amyloid related to alzheimer’s are related, except in the microscopic polarization.
I have to check this out. There is a famous scientist in New York as well.
Thank you for your comment.
Please review two of my posts on this website
Post # 1: 4/30/2012
Resident-cell-based Therapy in Human Ischaemic Heart Disease: Evolution in the PROMISE of Thymosin beta4 for Cardiac Repair
http://pharmaceuticalintelligence.com/2012/04/30/93/
Post #2: 5/29/2012
Lev-Ari, A. (2012b).Triple Antihypertensive Combination Therapy Significantly Lowers Blood Pressure in Hard-to-Treat Patients with Hypertension and Diabetes
http://pharmaceuticalintelligence.com/2012/05/29/445/
The thymosine beta4 report is very interesting. I’m straining to get all of this knowledge integrated and to comprehended. I am not surprised that they have found a basis for cardiac remodeling, to some extent, but the rapidity of these repeted developments is astounding in the last 8 years.
In the case of amyloid, it is unlikely to succeed. In the case of longstanding ischemic heart disease, we have known that there is remodeling of arterial circulation. There may also be associated generalized atherosclerosis (carotid, peripheral vascular disease, and not generally recognized, superior mesenteric artery disease). This contributes to a more difficult general circulatory problem, especially with loss of renal nephron mass, and with CHF.
Nevertheless, the scar from replacement of significant myocardial mass is compromizing function. Activating the repair as reported is terrific. This could be very intrigue for post-cocksackie myocarditis.
A change in our orientation to these discoveries takes some time to be realized. I spent 2 years in orthopedic pathology during the Vietnam war. The average person thinks about bone fragility and may view the tissue as a rigid tubular structure. We have a reasonable comprehension of the process of bone removal (osteoclast), bone accretion (osteoblast), and bone maintenance and remodeling (osteocyte). As in serious inflammatory diseases, the rates of loss are greater than the rates of repair. This is completely disordered in Paget’s disease.
Among so very many talents of your, you are born for Scientific Syndication of Research Results and carrying the subject matter straight to potential therapeutics – Yes this is exactly what I had in my own mind when I launched the platform – we, myself and all our contributors are both popularizing Science and are creating informational value added. So very much enjoying all your selection of topics to post about. Many thanks to you, Aviva
Thank you, Dr. Aviva. Its indeed a good feeling and encouraging to hear such kind words.
This particular topic is always very important and interesting as majority of, if not all, health complications in my little understanding boil down to deficient/ defective / faulty assimilation of food and /or elimination of so-called ‘effete’ materials. And one such disorders is this one, e.g., take Alzheimer’s, it is primarily, per current understanding, because of accumulation of amyloid plaques but not because of over production of it.
Understanding diseases from this perspective makes it more interesting. At the end of the day every thing boils down to the proper functioning of the system at a cellular / tissue level and apply that wealth of knowledge to prevent diseases in prenatal stage itself in order continue to have a Healthy World….because Health is the Mother of All Prosperity.
May I suggest to you to please curate a new post on the mechanism of action of There are two types of medications that help treat the symptoms of Alzheimer’s disease: acetylcholinesterase inhibitors and glutamate pathway modifiers. NAMENDA is the only glutamate pathway modifier and it works differently than acetylcholinesterase inhibitors, like ARICEPT.
http://www.namenda.com/Whats-Is-Combination-Therapy/Benefits.aspx
It will be complimentary and rather important to carry the pathophysiology, genetics and cell biology to the stage of available pharmacological treatment.
Please select few PDF to review and to curate from
http://www.bu.edu/alzresearch/team/faculty/abraham/
Prof. Carmela Abraham is a member of our Group Leaders in Pharmaceutical Business Intelligence on LinkedIn.
I will create a NEW category for Alzheimer’s disease and all your post and future posts on this topic will come under the Parent AZD category.
Thank you for joining forces with me, the privilege is fully mine, Aviva
functioning of the system at a cellular / tissue level and apply that wealth of knowledge to prevent diseases in prenatal stage itself
That may be expecting a lot because many of these diseases are not predictable in the prenatal state. The vast majority are secondary effects, which drives the interest in gene modifications.
Yes exactly. Each time we conduct a camp or otherwise, I come across many children who are suffering with chronic diseases like diabetes, heart troubles etc. My understanding is that that is because of the defective genetics from their parents (parents with chronic secondary effects continued in one or more generations before).
Taking into account the advancements in the understanding and genome technology may be in the near future one would be able to prevent such defective genes entering into their offsprings or get them repaired (remodify the modified genes) before planning to conceive.
This may sound like as simple as getting the vehicle repaired before hitting the highway, But we never know. With the developments in our understanding and the way the secrets of life unfolding I am very optimistic that it could become a reality one day and may be in the fifty years or so down the road we will see an absolutely healthy world.
[…] How to deal with the most common form of inherited amyloidoses?(pharmaceuticalintelligence.com) […]